-
食管癌是世界范围内常见的消化道肿瘤,其预后较差,5年生存率为19%,晚期食管癌仅为0.9%[1]。食管癌的发病率和组织学亚型表现出较大地理差异,最常见的组织学亚型为食管鳞癌,其在东亚和非洲部分地区发病率最高,而在西方国家,食管腺癌则为最常见的亚型[2]。流行病学分析[3]结果显示,烟酒史、胃部疾病、进食速度过快、热烫食品等均为其致病因素,男性发病率高于女性,且随着年龄增长,发病率明显增加,有超过60%的病人为老年人,且有约20%的食管癌病人在确诊时已经超过75岁[4]。虽然食管癌的治疗仍以手术为主,但对于老年病人,其身体机能以及术前合并症等对于外科医生仍是巨大的挑战,老年病人手术相关的死亡率也较高,因此,寻求适合于老年食管癌病人的治疗方式具有重要的临床意义。近些年的研究[5-6]表明,放、化疗对食管癌病人尤其是老年食管癌病人的临床疗效和生活质量的提高具有明显作用,与手术治疗相比,放、化疗对局部食管癌的侵袭性更小。本研究对老年食管癌病人采用替吉奥同步化疗联合放疗和单纯放疗进行治疗,评价其近、远期疗效和安全性。现作报道。
-
回顾性分析2015年12月至2017月5月我科收治的老年食管癌病人70例,所有病人均经病理组织学确诊为食管癌,其中男47例,女23例,年龄70~85岁;肿瘤位置:颈段3例,胸上段19例,胸中段41例,胸下段7例;病变长度2~10 cm。纳入标准:(1)年龄≥70周岁;(2)未接受过系统性抗肿瘤治疗;(3)病人均拒绝接受手术治疗;(4)病理组织学确诊为食管癌,病理类型为鳞癌;(5)根据实体瘤疗效评价标准1.1版本,具有可测量的病灶;(6)ECOG体能状态≤1,且重要器官功能无严重异常;(7)研究开始前癌细胞无转移;(8)充足的骨髓器官功能。排除标准:(1)对本研究中药物如替吉奥等不能耐受;(2)既往接受过针对不可切除的、局部晚期复发性或转移性食管鳞癌的全身治疗;(3)在研究治疗开始前4周内曾接受针对食管鳞癌的姑息性放疗;(4)有其他严重疾病或未控的全身疾病史;(5)有其他恶性肿瘤史者;(6)有活动性自身免疫性疾病或有自身免疫性疾病史;(7)重度慢性或活动性感染;(8)严重营养不良者;(9)药物滥用、酒精成瘾、精神异常者。其中35例接受同步化疗,35例接受单纯放疗,2组病人性别、年龄、肿瘤位置、病变长度差异均无统计学意义(P>0.05),具有可比性(见表 1)。
分组 男 女 年龄/岁 肿瘤位置 病变长度/cm 颈段 胸上段 胸中段 胸下段 同步化疗组 26(74.3) 9(25.7) 74.71±3.15 2(5.7) 10(28.57) 18(51.43) 5(14.29) 4.73±1.67 单纯放疗组 21(60.0) 14(40.0) 76.29±4.07 1(2.86) 9(25.71) 23(65.71) 2(5.71) 5.0±2.18 t >0.05△ 1.82 2.28△ 0.58 P >0.05 >0.05 >0.05 >0.05 △示χ2值 表 1 2组病人一般资料比较(ni=35;x±s)
-
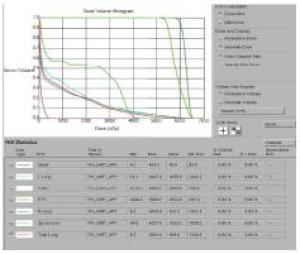
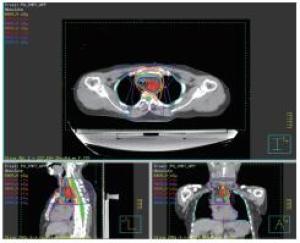
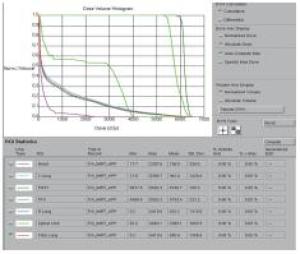
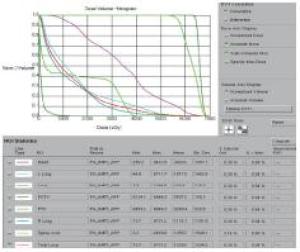
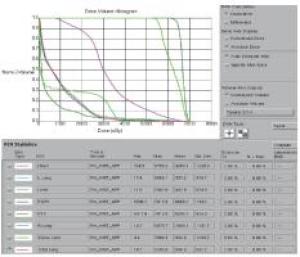
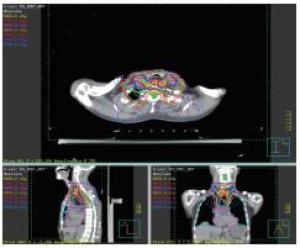
2组病人均全程采用三维适形调强放射治疗。病人仰卧位,根据颈部曲度选择合适头枕,双手上举交叉抱肘,以热塑体模固定,行CT模拟定位机扫描,扫描范围由下颌骨下缘至肝脏右叶下缘水平,层厚5 mm,扫描图像传输至Pinnacle治疗计划系统。由放疗科医师及物理师来确定肿瘤靶区(gross target volume,GTV)、临床靶区(clinical target volume,CTV)、计划靶区(planning target volume,PTV)以及危及器官(organ at risk,OAR)。GTV根据CT、食管镜及食道造影等勾画,包括食管病变及转移的淋巴结;CTV在GTV基础上前后左右各外扩0.5~0.8 cm,上下两端外扩2.5~3.0 cm;PTV将CTV前后左右均匀外扩0.5 cm,上下外扩1.0 cm;OAR包括脊髓、心脏、两侧肺组织以及肝脏。采用5~7个照射野进行适形调强治疗,95%等剂量线覆盖PTV,通过剂量体积直方图(dose volume histograms,DVH)和等剂量曲线进行优化(见图 1~8)。处方剂量:GTV为60~64 Gy,每次2 Gy,每天1次,每周5次,放疗周期为6~6.5周。OAR限量:双肺V20 < 30%,V30 < 20%,脊髓最大照射剂量 < 45 Gy,心脏V40 < 30%,V30 < 40%,肝脏V30 < 40%。所有病人首次放疗前均行兆伏级锥形CT(cone beam CT,CBCT)验证后,开始放疗。同步化疗组同时采用替吉奥(规格:20 mg/25 mg;齐鲁制药有限公司及山东新时代药业有限公司生产)单药化疗,体表面积 < 1.25 m2,每次40 mg;体表面积1.25~1.5 m2,每次50 mg;体表面积≥1.5 m2,每次60 mg,于放疗开始后口服,2次/天,21 d为1个周期,化疗1~2个周期)。
-
(1) 近期疗效:根据实体瘤疗效评价标准[7],按照完全缓解、部分缓解、稳定和进展评价近期疗效。(2)远期疗效:随访2年,采用电话、门诊或住院复查等方式进行随访,其中第1年每隔3个月随访1次,共计随访4次;第2年每隔3~4个月随访1次,共随访3~4次。比较2组的中位随访时间和总生存率。
-
按照美国国立癌症研究所常规毒性判定标准[8](4.0)进行判定,出现任何程度的不良反应均需立即进行评价,必要时应给予对症处理,也可终止治疗。
-
采用t检验、χ2检验、秩和检验和Fisher′s确切概率法。
-
2组近期疗效差异无统计学意义(P>0.05)(见表 2)。以完全缓解率和部分缓解计算总有效率,同步化疗组总有效率为85.71%(30/35),高于单纯放疗组的60.0%(21/35)(χ2=5.85,P < 0.05)。
分组 n 完全缓解 部分缓解 稳定 进展 uc P 同步化疗组 35 16(45.71) 14(40.00) 3(8.57) 2(5.71) 单纯放疗组 35 12(34.29) 9(25.71) 8(22.86) 6(17.14) 1.82 >0.05 合计 70 28(40.00) 23(32.86) 11(15.71) 8(11.43) 表 2 2组病人近期疗效比较[n;百分率(%)]
-
2组均未出现因不良反应致死或终止治疗的病人。主要不良反应均为骨髓抑制(白细胞减少、中性粒细胞减少、血小板减少)、消化道反应(恶心、呕吐)和放射性食管炎等,经升白细胞药物、升血小板药物、止吐药物处理后均缓解,病人均能较好耐受。2组Ⅲ~Ⅳ级骨髓抑制及放射性食管炎发生率差异均无统计学意义(P>0.05)(见表 3)。
分组 n 骨髓抑制 放射性食管炎 同步化疗组 35 2(5.71) 11(41.43) 单纯放疗组 35 0(0.00) 9(25.71) χ2 — — 0.28 P — >0.05* >0.05 *示Fisher′s确切概率法 表 3 2组病人Ⅲ~Ⅳ级不良反应比较[n;百分率(%)]
-
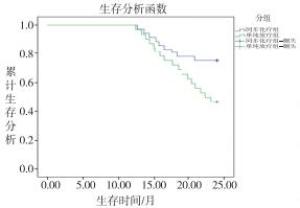
随访2年,同步化疗组和单纯放疗组分别有1例和3例病人失访,2组2年生存率分别为73.53%(25/34)和46.88%(15/32),差异有统计学意义(χ2=4.91,P < 0.05)。同步化疗组和单纯放疗组病人的中位生存时间分别为24.0个月和18.0个月,绘制Kaplan-Meier生存曲线,经Log-rankχ2分析,2组差异有统计学意义(χ2=4.98,P < 0.05)(见图 9)。
-
我国是食管癌病死率最高的国家之一,确诊时多数病例为中晚期,外科手术治疗为治疗食管癌的主要方法,但即使是早期病变的切除,术后的复发率仍然很高。由于食管黏膜下独特的淋巴管系统,食管癌早期侵入黏膜肌层时已有转移,临床研究中也观察到,大多数局部中晚期食管癌病人中已经发生了局部区域的淋巴转移[9-10]。我国国内食管癌的发病年龄集中于老年病人,就诊时分期主要在T3、T4期。而由于老年病人的脏器功能减退、基础疾病较多等原因,进行手术治疗的可能性较低,放、化疗联合治疗是主要的治疗方法[11]。
2003年,SWISHER等[12]对局部晚期食管癌病人进行化疗、放化疗以及手术治疗的长期疗效的对比研究结果,确定了将5-氟尿嘧啶和顺铂的同步放化疗作为了治疗局部晚期食管癌的非手术治疗的标准治疗方法。近些年,为了降低食管癌病人不良反应发生率、延长病人生存期、降低食管癌复发率,不同药物的同步放化疗研究成为了临床医师的关注热点。CHEN等[13]将436例ⅡA~ⅣA期食管鳞癌病人作为研究对象并随机分为2组,分别采用同步放化疗标准治疗方法(顺铂联合5-氟尿嘧啶)和5-氟尿嘧啶联合紫杉醇的同步放化疗治疗方法,放疗剂量为61.2 Gy/34F,结果显示,5-氟尿嘧啶联合紫杉醇的同步放化疗治疗食管癌可延长病人生存率。SATAKE等[14]对DCF疗法(多西他赛、顺铂和5-氟尿嘧啶)治疗食管鳞癌的研究,结果也显示病人的中位生存期达到26.0个月,3年的生存率为40.4%,证明DCF同步放化疗具有良好的临床疗效,使病人的生存期得以有效延长,且提高了病人的生存率。
替吉奥胶囊是最早在日本上市生产的由替加氟、吉美嘧啶和奥替拉西钾组成的复方制剂,用于治疗不能切除的局部晚期或转移性胃癌,其疗效已经过多年临床经验证实并获得临床医师的广泛认可[15-16]。日本学者[17]将替吉奥联合顺铂化疗用于治疗食管癌及颈、胸、腹腔动脉淋巴结转移的病人,取得良好的临床疗效。在我国,佐志刚等[18]采用替吉奥化疗联合三维适形放射治疗56例中晚期食管癌病人,结果亦显示,替吉奥联合三维适形放射治疗食管癌提高了近期疗效。赵世杰[19]采用同样的方法对老年食管癌病人进行治疗,结果显示,同步放化疗治疗食管癌的临床疗效较好,且不良反应均可以耐受。本研究结果亦显示,同步化疗组治疗总有效率高于单纯放疗组;2组均未出现因不良反应致死或终止治疗病人,Ⅲ~Ⅳ级骨髓抑制及放射性食管炎发生率差异无统计学意义;随访2年,同步化疗组中位生存时间和2年生存率均高于单纯放疗组。
综上,替吉奥同步化疗联合放疗治疗老年食管癌近期疗效较好,可增加中位生存时间和生存率,且未增加不良反应,老年病人可以较好耐受,适合临床应用。
替吉奥同步化疗联合放疗治疗老年食管癌的临床疗效分析
Clinical effect analysis of tegafur/gimeracil/oteracil concurrent chemotherapy combined with radiotherapy in the treatment of esophageal cancer in elderly patient
-
摘要:
目的观察替吉奥同步化疗联合放疗治疗老年食管癌的临床疗效及不良反应。 方法回顾性分析老年食管癌病人70例临床资料,其中接受单纯放疗和同步化疗病人各35例。2组均全程采用三维适形调强放疗,同步化疗组同时采用替吉奥胶囊单药化疗。按照实体瘤评价标准(完全缓解、部分缓解、疾病稳定和疾病进展)评价近期疗效,并采用NCI-CTC对不良反应进行评定。随访2年,比较2组的中位生存时间和总生存率。 结果同步化疗组治疗总有效率为85.71%(30/35),高于单纯放疗组的60.0%(21/35)(χ2=5.85,P < 0.05)。2组均未出现因不良反应致死或终止治疗的病人,病人均能较好耐受,2组Ⅲ~Ⅳ级骨髓抑制及放射性食管炎发生率差异均无统计学意义(P>0.05)。随访2年,同步化疗组和单纯放疗组分别有1例和3例病人失访,同步化疗组中位生存时间和2年生存率分别为24.0个月和73.53%(25/34),均高于单纯放疗组的18.0个月和46.88%(15/32)(P < 0.05)。 结论替吉奥同步化疗联合放疗治疗老年食管癌近期疗效较好,不良反应较小,可提高老年病人生存率。 Abstract:ObjectiveTo observe the clinical efficacy and side effects of tegafur/gimeracil/oteracil concurrent chemotherapy combined with radiotherapy in the treatment of esophageal cancer in elderly patient. MethodsThe clinical data of elderly patients with esophageal cancer were retrospectively analyzed, which included 35 patients treated with radiotherapy alone and 35 cases treated with concurrent chemotherapy.The two groups were treated with three-dimensional conformal intensity modulated radiotherapy.The concurrent chemotherapy group was additionally treated with single agent chemotherapy with tegafur/gimeracil/oteracil.The short-term efficacy was evaluated according to the criteria for evaluating solid tumors (complete remission, partial remission, disease stability and disease progression), and the side effects were evaluated using NCI-CTC.The two groups were followed up for 2 years, the median overall survival time and overall survival rate were compared between two groups. ResultsThe total effective rate in concurrent chemotherapy group (85.71%) was higher than that in chemotherapy alone group (60.0%) (P < 0.05).Two groups did not appear because of the adverse reactions to death or termination of the treatment of patients, patients could tolerate, and the differences of the incidence rates of grade Ⅲ ~ Ⅳ bone marrow suppression and radiation esophagitis between two groups were not statistically significant (P>0.05).During two years of following up, one case in concurrent chemotherapy and 3 cases in chemotherapy alone group losed to follow up.The time of median survival time and 2-year survival rate in concurrent chemotherapy group (24 months and 73.53%) were higher than those in chemotherapy alone group (18 months and 46.88%), respectively (P < 0.05). ConclusionsThe tegafur/gimeracil/oteracil concurrent chemotherapy combined with radiotherapy in the treatment of esophageal cancer in elderly patient has good short-term effects and less side effects, which can significantly improve the overall survival rate of elderly patients. -
Key words:
- esophageal neoplasms /
- elderly patient /
- chemotherapy /
- radiotherapy /
- tegafur/gimeracil/oteracil
-
表 1 2组病人一般资料比较(ni=35;x±s)
分组 男 女 年龄/岁 肿瘤位置 病变长度/cm 颈段 胸上段 胸中段 胸下段 同步化疗组 26(74.3) 9(25.7) 74.71±3.15 2(5.7) 10(28.57) 18(51.43) 5(14.29) 4.73±1.67 单纯放疗组 21(60.0) 14(40.0) 76.29±4.07 1(2.86) 9(25.71) 23(65.71) 2(5.71) 5.0±2.18 t >0.05△ 1.82 2.28△ 0.58 P >0.05 >0.05 >0.05 >0.05 △示χ2值 表 2 2组病人近期疗效比较[n;百分率(%)]
分组 n 完全缓解 部分缓解 稳定 进展 uc P 同步化疗组 35 16(45.71) 14(40.00) 3(8.57) 2(5.71) 单纯放疗组 35 12(34.29) 9(25.71) 8(22.86) 6(17.14) 1.82 >0.05 合计 70 28(40.00) 23(32.86) 11(15.71) 8(11.43) 表 3 2组病人Ⅲ~Ⅳ级不良反应比较[n;百分率(%)]
分组 n 骨髓抑制 放射性食管炎 同步化疗组 35 2(5.71) 11(41.43) 单纯放疗组 35 0(0.00) 9(25.71) χ2 — — 0.28 P — >0.05* >0.05 *示Fisher′s确切概率法 -
[1] TESTA U, CASTELLI G, PELOSI E.Esophageal cancer:Genomic and molecular characterization, stem cell compartment and clonal evolution[J].Medicines (Basel), 2017, 4(3):67. [2] WANG K, JOHNSON A, ALI SM, et al.Comprehensive genomic profiling of advanced esophageal squamous cell carcinomas and esophageal adenocarcinomas reveals similarities and differences[J].Oncologist, 2015, 20(10):1132. [3] 安尼瓦尔·买买提, 陈海林, 依不拉音·依不拉音, 等.2015年喀什地区食管癌流行病学分析[J].国际肿瘤学杂志, 2017, 44(3):161. [4] XU YP, ZHENG YD, SUN XJ, et al.Concurrent radiotherapy with gefitinib in elderly patients with esophageal squamous cell carcinoma:Preliminary results of a phase Ⅱ study[J].Oncotarget, 2015, 6(35):38429. [5] SUZUKI G, YAMAZAKI H, AIBE N, et al.Definitive radiotherapy for older patients aged ≥ 75 years with localized esophageal cancer[J].In Vivo, 2019, 33(3):925. [6] GARANT A, WHITAKER TJ, SPEARS GM, et al.A comparison of patient-reported health-related quality of life during proton versus photon chemoradiotherapy for esophageal cancer[J].Prac Radio Oncol, 2019, 9(6):410. [7] MIN SJ, JANG HJ, KIM JH.Comparison of the RECIST and PERCIST criteria in solid tumors:a pooled analysis and review[J].Oncotarget, 2016, 7(19):27848. [8] JANMAAT VT, STEYERBERG EW, VAN DER GAAST A, et al.Palliative chemotherapy and targeted therapies for esophageal and gastroesophageal junction cancer[J].Cochrane Database Syst Rev, 2017, 2017(11):CD004063. [9] OLD OJ, ISABELLE M, BARR H.Staging early esophageal cancer[J].Adv Exp Med Biol, 2016, 908:161. [10] WANG J, YU J, WANG J, et al.Thalidomide combined with chemo-radiotherapy for treating esophageal cancer:A randomized controlled study[J].Oncol Lett, 2019, 18(1):804. [11] XING L, LIANG Y, ZHANG J, et al.Definitive chemoradiotherapy with capecitabine and cisplatin for elder patients with locally advanced squamous cell esophageal cancer[J].J Cancer Res Clin Oncol, 2014, 140(5):867. [12] SWISHER SG, AJANI JA, KOMAKI R, et al.Long-term outcome of phase Ⅱ trial evaluating chemotherapy, chemoradiotherapy, and surgery for locoregionally advanced esophageal cancer[J].Int J Radiat Oncol Biol Phys, 2003, 57(1):120. [13] CHEN Y, ZHU Z, ZHAO W, et al.A randomized phase 3 trial comparing paclitaxel plus 5-fluorouracil versus cisplatin plus 5-fluorouracil in Chemoradiotherapy for locally advanced esophageal carcinoma-the ESO-shanghai 1 trial protocol[J].Radiat Oncol, 2018, 3(1):33. [14] SATAKE H, TAHARA M, MOCHIZUKI S, et al.A prospective, multicenter phase Ⅰ/Ⅱ study of induction chemotherapy with docetaxel, cisplatin and fluorouracil (DCF) followed by chemoradiotherapy in patients with unresectable locally advanced esophageal carcinoma[J].Cancer Chemother Pharmacol, 2016, 78(1):91. [15] YASUMOTO K, KITAKATA H, YAMASHITA K, et al.Combination chemotherapy of TS-1, docetaxel and CDDP produces a remarkable response in a patient with advanced esophageal cancer[J].Gan To Kagaku Ryoho, 2006, 33(7):965. [16] 鲍瑜, 钱江, 宋文灿, 等.阿帕替尼联合替吉奥一线治疗晚期胃癌临床研究[J].中国药业, 2019, 28(11):74. [17] IWASE H, OKEYA M, SHIMADA M, et al.A case of metastatic esophageal cancer responding remarkably to combination chemotherapy of TS-1 and cisplatin[J].Gan To Kagaku Ryoho, 2004, 31(5):755. [18] 佐志刚, 潘东风, 蔡晓军, 等.替吉奥联合三维适形放射治疗56例中晚期食管癌[J].第三军医大学学报, 2010, 32(22):2446. [19] 赵世杰.调强放疗结合替吉奥同步治疗非手术老年食管癌的疗效观察[J].现代诊断与治疗, 2019, 30(6):900. -




 下载:
下载: