-
肝脏手术特别是肝移植手术中常出现肝缺血/再灌注(I/R)的操作过程,这可导致肺脏等多器官的损伤,严重者出现功能衰竭。肝脏手术I/R所致急性肺损伤的发病机制目前尚不清楚。山莨菪碱为M胆碱受体拮抗剂,研究[1]证实其对多个器官I/R损伤具有良好的保护作用。本研究制作70%肝脏I/R损伤模型,以山莨菪碱进行预处理,旨在探讨山莨菪碱预处理对大鼠肝I/R诱发肺损伤的影响,为临床防治研究提供理论支持。
-
本实验选择健康清洁成年雄性SD大鼠48只,体质量210~280 g,由西安交通大学医学部动物实验中心提供。盐酸消旋山莨菪碱注射液(天津金耀氨基酸有限公司,批号:1409051);SP试剂盒购自北京中山生物技术有限公司;丙二醛(MDA)、髓过氧化物酶(MPO)与超氧化物歧化酶(SOD)试剂盒购自南京生物建成工程研究所;图像采集系统、光学显微镜等由西安交通大学医学院病理学实验室提供。
-
48只大鼠按随机数字表法平均分成3组,分别为假手术组(S组)、肝脏缺血再灌注组(IR组)和山莨菪碱预处理组(A组),每组16只。S组:大鼠开腹后分离肝门不做结扎;IR组:大鼠开腹后结扎肝门,60 min后恢复肝脏血流,开放血流3 h后处死大鼠;A组:制模过程同IR组,阻断入肝血流前30 min,自尾静脉注射稀释成1 mL的山莨菪碱(约2 mg/kg),其余同IR组。S组和IR组自尾静脉注射等剂量的0.9%氯化钠溶液。
按NAUTA等[2]的方法制作70%肝脏I/R模型:动物于术前禁食12 h,自由饮水,采用腹腔内注射10%水合氯醛(400 mg/kg)麻醉大鼠,2~4 min后以钳夹四肢无反应作为麻醉成功标志,然后将大鼠仰卧位固定于操作台上。经阴茎背静脉注入稀释肝素(200 U/kg),使其肝素化。自腹下部沿正中线纵行向上剪开皮肤,直至胸腹交界处。逐层打开腹腔,分离肝门及肝十二指肠韧带,寻找到肝门,IR组和A组用小动脉夹夹闭肝左叶和肝中叶部分肝门,30 min后松开小动脉夹肝脏恢复血流,然后逐层关闭腹腔,3 h后处死大鼠。S组只解剖,肝门不作血管夹闭。
-
大鼠处死后,取右肺上叶置于10%中性甲醛溶液浸泡固定,待行HE染色和免疫组织化学(免疫组化)。取右肺下叶组织,称重为湿重,然后放入70 ℃烤箱中烘烤48 h后,称重为干重,计算肺组织湿重/干重(W/D)比值。取部分右肺组织用滤纸吸干,置于-70 ℃冰箱保存,待进一步检测MDA含量、MPO和SOD活性。
-
取部分肺组织经过脱蜡,水化,染色,脱水,透明,封片,在100倍和400倍光镜下观察肺组织病理形态学变化,采集图像。
-
取部分右上叶肺组织标本,石蜡包埋,制成3 μm厚的切片。SP法免疫组化染色按SP试剂盒操作说明实施。随机抽取上述切片,以PBS代替HO-1和iNOS多克隆抗体作为阴性对照(其余步骤不变)。全自动图像分析系统对阳性染色进行分析,测定HO-1和iNOS阳性表达的吸光度值,随机测定5个高倍视野,计算其平均值作为该切片的代表值。
-
取部分肺组织50 mg用冷0.9%氯化钠溶液制成肺组织匀浆,4℃ 3 000 r/min离心10 min,上清液-80 ℃保存,检测前再次离心取上清液;用试剂盒分别采用硫代巴比妥酸法测定MDA含量、黄嘌呤氧化酶法测定SOD活性和过氧化氢还原法测定MPO活性,严格按照试剂盒说明书操作步骤进行检测。
-
采用单因素方差分析和q检验。
-
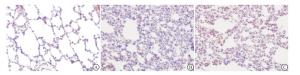
S组双肺组织表面光滑,呈淡粉红色,打开胸腔后很快萎缩;IR组肺脏体积增大颜色变深,组织包膜下可见点状、片状出血灶,肺泡腔有渗出液;A组肺组织轻度增大,肺组织可见点状、片状出血灶,但较IR组轻。
-
S组肺组织结构完整,未见明显的异常,肺泡腔完整,由单层肺泡上皮细胞组成;IR组肺组织血管充血,肺泡腔破裂且大小不一,可见红色渗出液,肺间质增厚水肿,有大量炎性细胞浸润;A组肺组织可见充血水肿,肺间质增厚有炎性细胞浸润,但较IR组轻(见图 1)。
-
光镜下,IR组、A组大鼠肺组织肺泡上皮细胞、肺间质炎性细胞及血管内皮细胞等细胞内HO-1的表达呈阳性(呈棕黄色),A组表达最强,S组表达最弱(呈弱阳性)。与S组比较,IR组和A组肺组织HO-1蛋白表达均明显增加(P < 0.01);与IR组比较,A组肺组织HO-1蛋白表达明显增加(P < 0.01)(见表 1、图 2)。
分组 n HO-1 iNOS S组 16 0.036±0.008 0.021±0.007 IR组 16 0.165±0.013** 0.136±0.012** A组 16 0.202±0.016**△△ 0.078±0.014**△△ F — 745.46 407.98 P — < 0.01 < 0.01 MS组内 — 0.000 2 0.000 1 q检验:与S组比较**P < 0.01;与IR组比较△△P < 0.01 表 1 3组大鼠肺组织HO-1和iNOS蛋白表达的比较(x ±s)
-
光镜下,IR组和A组大鼠肺组织中肺泡间质的血管平滑肌细胞、单核-巨噬细胞、支气管上皮细胞、血管内皮细胞等胞内iNOS的表达呈阳性(呈棕黄色),IR组表达最强,S组表达最弱(呈弱阳性)。与S组比较,IR组和A组肺组织iNOS蛋白表达均明显增加(P < 0.01);与IR组比较,A组肺组织iNOS蛋白表达明显降低(P < 0.01)(见表 1、图 3)。
-
与S组比较,IR组和A组肺组织W/D比值、MDA含量、MPO活性均明显增高(P < 0.01),SOD活性均明显降低(P < 0.01);与IR组相比较,A组肺组织W/D比值、MDA含量、MPO活性均明显降低(P < 0.01),SOD活性明显增高(P < 0.01)(见表 2)。
分组 n W/D比值 MDA/(nmol/mg) MPO/(U/g) SOD/(U/mg) S组 16 2.99±0.32 1.22±0.25 1.36±0.80 94.52±8.11 IR组 16 6.16±0.82** 2.96±0.44** 5.12±1.28** 68.45±9.82** A组 16 4.42±0.78**△△ 2.06±0.35**△△ 3.18±1.12**△△ 81.33±9.22**△△ F — 87.46 96 48.04 32.99 P — < 0.01 < 0.01 < 0.01 < 0.01 MS组内 — 0.461 0.126 1.178 82.404 q检验:与S组比较**P < 0.01;与IR组比较△△P < 0.01 表 2 3组肺组织W/D比值、MDA含量、MPO和SOD活性的比较(x ±s)
-
肝脏I/R损伤在临床上十分常见,创伤、休克、肝叶切除、肝移植等过程中都有可能发生, 不仅引起肝脏本身的损害,还可导致远隔器官如肺、肾、小肠等器官的功能受损,肺脏是其中最易受累的器官之一。我们的前期研究[3]发现,肝I/R损伤能加重大鼠肺损伤,上调肺组织中的HO-1及iNOS表达。本实验研究结果显示,与S组比较,IR组和A组肺组织HE染色病理改变显著加重,肺叶W/D比显著升高,肺组织MDA、MPO表达显著升高,而SOD显著降低,表明肝I/R过程中造成了肺损伤。
山莨菪碱是提取自茄科植物山莨菪中的一种生物碱,与阿托品等莨菪烷类药物同属于M胆碱受体拮抗剂[4]。主要用于治疗胃肠道痉挛收缩所引起的腹痛、感染中毒性休克等[5]。近年来研究[6]发现山莨菪碱对生命重要器官脑、肝、肾、心等器官的I/R损伤具有良好的保护作用,但其机制尚不清楚。研究[7-8]证实山莨菪碱对大鼠肝脏I/R损伤有保护作用,其机制可能是山莨菪碱能改善再灌注后的肝脏微循环障碍、减少氧自由基的生成。山莨菪碱可能通过抑制细胞因子肿瘤坏死因子-α、白细胞介素-6、白细胞介素-1等的释放,抑制中性粒细胞聚集和氧自由基的产生、稳定细胞膜和溶酶体膜、改善和疏通微循环等作用,对重要器官I/R损伤发挥保护作用[9]。实验过程中我们发现,山莨菪碱对I/R的肝脏具有保护作用。山莨菪碱是否对肝脏I/R损伤过程中肺损伤具有保护作用,目前尚少报道。因此,本实验首先建立70%肝脏I/R损伤模型,探讨山莨菪碱预处理对大鼠肝I/R诱发肺损伤的保护作用。
本研究参照文献[7-8]确定山莨菪碱剂量为2 mg/kg进行预处理,结果表明,与IR组比较,A组肺组织HE染色病理改变显著减轻,肺叶W/D比显著降低,肺组织MDA、MPO表达显著降低,而SOD表达显著升高,表明山莨菪碱预处理对肝脏I/R后肺损伤具有一定的保护作用。
HO-1又称热休克蛋白32 (HSP32),是血红素的限速酶和关键酶,有研究[10]表明HO-1活性的上调,可产生抗炎、抗氧化、抗凋亡等生物学效应,对肝脏I/R损伤起重要的保护作用[10]。iNOS主要存在炎性细胞中,是一种重要炎性介质,病理状态下iNOS受炎性介质诱导生成增加,可产生大量NO,然而病理情况下大量的NO参与介导许多病理过程如肝脏I/R损伤、炎症及休克等,导致器官损伤[11]。本实验研究发现,与S组比较,IR组和A组的HO-1和iNOS蛋白表达显著增加;与IR组比较,A组的HO-1蛋白表达显著增加,而iNOS蛋白表达显著降低。表明肝脏I/R损伤后肺组织中HO-1和iNOS蛋白表达都显著增加,而山莨菪碱预处理可以显著增加HO-1蛋白表达和减少iNOS蛋白表达,这可能与山莨菪碱预处理能减轻肝脏I/R后肺损伤的机制有关。
综上所述,山莨菪碱预处理可减轻大鼠肝I/R肺损伤,其机制可能与上调肺组织中的HO-1及下调iNOS蛋白表达有关。但肝脏I/R肺损伤是个多环节构成、多因素参与的复杂过程,是否存在其他方面的因素,有待我们进一步研究。
山莨菪碱预处理对大鼠肝缺血再灌注诱发肺损伤的影响
Effect of anisodamine pretreatment on the lung injury induced by hepatic ischemia-reperfusion in rats
-
摘要:
目的探讨山莨菪碱预处理对大鼠肝缺血再灌注诱发肺损伤的影响。 方法48只SD健康雄性大鼠,采用随机数字表法,分为假手术组(S组)、肝缺血再灌注组(IR组)和山莨菪碱预处理组(A组),各16只。S组只做解剖肝门,IR组和A组制备70%大部分肝缺血再灌注损伤模型。A组于阻断肝血流前30 min,静脉注射山莨菪碱2 mg/kg,S组和IR组静脉注射等剂量的0.9%氯化钠溶液。大鼠于再灌注后3 h处死,取肺脏组织,HE染色,光镜下观察病理学结果。计算肺湿/干重(W/D)比值。采用硫代巴比妥酸法测定丙二醛(MDA),黄嘌呤氧化酶法测定超氧化物歧化酶(SOD),氧化氢还原法测定髓过氧化物酶(MPO)的表达;采用免疫组织化学法测定肺组织血红素氧合酶-1(HO-1)和诱导型一氧化氮合酶(iNOS)蛋白的表达。 结果肺组织HE染色病理改变,IR组和A组肺损伤较S组重,A组肺损伤较IR组轻;与S组比较,IR组和A组肺组织W/D比值、MDA含量、MPO活性、HO-1和iNOS蛋白表达均明显增高(P < 0.01),SOD活性明显降低(P < 0.01);与IR组比较,A组肺组织W/D比值、MDA含量、MPO活性、iNOS蛋白表达均明显降低(P < 0.01),肺组织HO-1蛋白表达和SOD活性均明显升高(P < 0.01)。 结论山莨菪碱预处理可减轻大鼠肝缺血再灌注诱发的肺损伤。 Abstract:Objective To investigate the effects of anisodamine pretreatment on the lung injury induced by hepatic ischemia-reperfusion in rats. Methods Forty-eight healthy male Sprague-Dawley rats were randomly divided into the sham operation group(group S), hepatic ischemia-reperfusion group(group IR) and anisodamine group(group A) using a random number table(16 rats each group).The hepatic portal in group S was dissected, and the ischemia-reperfusion model of 70% liver in group IR and group A were established.The group A was injected with 2 mg/kg anisodamine before 30 min of blocking liver blood flow and the group S and group IR were injected with the same volume of 0.9% sodium chloride solution.The rats were sacrificed after 3 h of reperfusion, the lung tissue was harvested, stained using HE, and the result of which was observed under light microscope.The ratio of wet to dry(W/D) of lung weight was calculated.The levels of malondialdehyde(MDA), superoxide dismutase(SOD) and myeloperoxidase(MPO) in lung tissue were detected using thiobaituricacid, xanthinoxidase and Hydrogen oxide reduction method, respectively.The expression levels of heme oxygenase-1(HO-1) and inducible nitric oxide synthase(iNOS) in lung tissue were determined by immunohistochemistry. Results The Results of HE staining in lung tissue showed that the lung injury in IR and A groups were more serious than that in group S, and the lung injury in group A was lighter than that in group IR.Compared with the group S, the ratio of W/D, MDA content, MPO activity and HO-1 and iNOS protein expression level significantly increased(P < 0.01), and the SOD activity significantly decreased in IR and A groups(P < 0.01).Compared with the group IR, the ratio of W/D, MDA content, MPO activity and iNOS protein expression level significantly decreased(P < 0.01), and the HO-1 protein level and SOD activity in lung tissue significantly increased in group A(P < 0.01). Conclusions Anisodamine pretreatment can alleviate the lung injury induced by hepatic ischemia-reperfusion in rats. -
Key words:
- hepatic ischemia-reperfusion /
- anisodamine /
- pretreatment /
- lung injury
-
表 1 3组大鼠肺组织HO-1和iNOS蛋白表达的比较(x ±s)
分组 n HO-1 iNOS S组 16 0.036±0.008 0.021±0.007 IR组 16 0.165±0.013** 0.136±0.012** A组 16 0.202±0.016**△△ 0.078±0.014**△△ F — 745.46 407.98 P — < 0.01 < 0.01 MS组内 — 0.000 2 0.000 1 q检验:与S组比较**P < 0.01;与IR组比较△△P < 0.01 表 2 3组肺组织W/D比值、MDA含量、MPO和SOD活性的比较(x ±s)
分组 n W/D比值 MDA/(nmol/mg) MPO/(U/g) SOD/(U/mg) S组 16 2.99±0.32 1.22±0.25 1.36±0.80 94.52±8.11 IR组 16 6.16±0.82** 2.96±0.44** 5.12±1.28** 68.45±9.82** A组 16 4.42±0.78**△△ 2.06±0.35**△△ 3.18±1.12**△△ 81.33±9.22**△△ F — 87.46 96 48.04 32.99 P — < 0.01 < 0.01 < 0.01 < 0.01 MS组内 — 0.461 0.126 1.178 82.404 q检验:与S组比较**P < 0.01;与IR组比较△△P < 0.01 -
[1] 曹锋生, 韩继媛.抗胆碱能药防治缺血-再灌注损伤的研究进展[J].医药导报, 2006, 25(9):931. doi: 10.3870/j.issn.1004-0781.2006.09.037 [2] NAUTA RJ, TSIMOYIANNIS E, URIBE M, et al.Oxygen-derived free radical in hepatic ischemia and reperfusion injury in the rat[J].Surg Gynecol Obstet, 1990, 17:120. [3] 谭敬, 周荣胜, 刘庆波, 等.肝缺血再灌注损伤对大鼠肺组织HO-1和iNOS表达的影响[J].山西医科大学学报, 2015, 46(9):861. [4] ZHANG WW, XU ZP, CUI YY, et al.Peripheral cholinoceptor antagonist anisodamine counteracts cholinergic adverse effects and facilitates cognitive amelioration of rivastigmine[J].J Neural Transm, 2009, 116(12):1643. doi: 10.1007/s00702-009-0297-7 [5] 孙凯, 杨丽敏.山莨菪碱的药理和临床研究进展[J].世界临床药物, 2010, 31(3):182. [6] 尹文, 虎晓岷, 袁静, 等.山莨菪碱对多器官功能障碍综合征保护机制的实验研究[J].中国医师杂志, 2004, 6(1):8. doi: 10.3760/cma.j.issn.1008-1372.2004.01.003 [7] 董满库, 崔彦, 陈昌玮, 等.山莨菪碱对大鼠肝脏缺血再灌注损伤保护作用的实验研究[J].中国普外基础与临床杂志, 2001, 8(5):295. doi: 10.3969/j.issn.1007-9424.2001.05.004 [8] 董满库, 崔彦, 周立艳, 等.山莨菪碱对肝脏缺血再灌注后氧自由基的影响[J].世界华人消化杂志, 2003, 11(1):82. doi: 10.3969/j.issn.1009-3079.2003.01.022 [9] 夏春秋, 仲崇俊, 张亚年.山莨菪碱对肺缺血(再灌注损伤的保护机制[J].中国微循环, 2006, 10(3):221. [10] 杨定华, 张华, 黄毓, 等.HO-1的表达对肝脏缺血再灌注损伤的保护作用[J].南方医科大学学报, 2009, 29(11):2329. doi: 10.3321/j.issn:1673-4254.2009.11.024 [11] MATSUI T, NAGAFUJI T, KUMANISHI T, et al.Role of nitric oxide in pathogenesis underlying ischemic cerebral damage[J].Cellular Molecular Neurob, 1999, 19(1):177. -





 下载:
下载: