-
表皮生长因子受体酪氨酸激酶抑制剂(epidermal growth factor receptor tyrosine kinase inhibitors,EGFR-TKIs)为治疗晚期非小细胞肺癌(non-small cell lung cancer, NSCLC)提供了一种新的治疗方法[1]。EGFR-TKIs是目前治疗包含外显子19缺失或外显子21 L858R突变EGFR突变的晚期NSCLC的一线治疗的最佳方案[2]。与一线含铂双药方案和二线单药多西他赛或培美曲塞相比,应用第一代(吉非替尼、埃克替尼)和第二代EGFR-TKIs(阿法替尼)治疗EGFR突变的病人,在临床观察中发现病人的客观反应率(objective response rate,ORR)、无进展生存期(progression-free survival,PFS)和总体生存期(overall survival,OS)均取得较好疗效, 在不同病人间存在显著的个体差异性,一部分病人经靶向治疗后病人的PFS可能会持续数年,而另一部分人可能在数周内病情进展,甚至疾病的病程呈爆发性加快进展[3-4]。出现以上情况的主要生物学机制可能为病人存在与EGFR共同突变的相关的基因,该病人存在对第一代、第二代EGFR-TKIs的具有天然耐药基因[5-7]。PIK3CA是EGFR信号通路的下游关键传导基因,对细胞的存活、增殖和生长有重要作用。在临床前模型研究中发现的EGFR- TKIs耐药性与体内多个基因突变相关,在本研究中将观察PIK3CA与肺癌EGFR-TKIs靶向治疗的相关性。
-
选择2015年9月至2018年6月就诊于我院门诊或病房经电子支气管镜活检、CT或B超引导下经皮肺穿刺、电子胸腔镜活检、胸腔穿刺胸腔积液病理或细胞学确诊为NSCLC病人。病理均由2位病理医师确诊,均为初始治疗,治疗未行化疗及放疗。经本实验室通过PCR-ARMS法在中晚期NSCLC病人的肿瘤病理组织中检测EGFR基因突变情况。同意将EGFR-TKIs治疗作为一线方案的病人被纳入研究,最终纳入62例NSCLC病人。男21例,女41例,年龄49~83岁。依据TNM分期,其中Ⅲb期16例,Ⅳ期46例;根据病理类型分类,鳞癌6例,腺癌56例;有淋巴结转移者共有50例,无淋巴结转移者共有12例;吸烟16例,不吸烟46例。
-
所使用的主要仪器设备Leica-ASP200S全自动脱水机,Leica染色机Autostainer XL,Leica-EG1150H全自动包埋机,Leica RM2235石蜡切片机,Sartorlus PB-10酸度计,Olympus BX41生物显微镜,无水乙醇、二甲苯等购自上海麦克林生化试剂有限公司。
-
(1) 切片脱蜡至水:经二甲苯Ⅰ、Ⅱ、Ⅲ,3次,每次5 min,要确保二甲苯足量有效;无水乙醇2次,每次5 min;95%乙醇2次,每次5 min;蒸馏水2次,每次5 min。(2)采用柠檬酸抗原修复法热修复后冷却至室温,PBS洗3次,每次5 min。(3)3%过氧化氢封闭15 min:30%过氧化氢与蒸馏水1:9配置,新鲜配置,现配现用;(4)PBS洗3次,每次5 min;(5)15%牛血清白蛋白封闭30 min;(6)封闭后甩掉封闭液,加一抗孵育,4 ℃过夜:(7)室温平衡30 min;(8)PBS洗3次,每次5 min;(9)二抗孵育,37 ℃,1 h;(10)PBS洗3次,每次5 min;(11)DAB显色;(12)显色完毕置于蒸馏水,梯度浓度乙醇脱水,二甲苯透明,中性树胶封片。
-
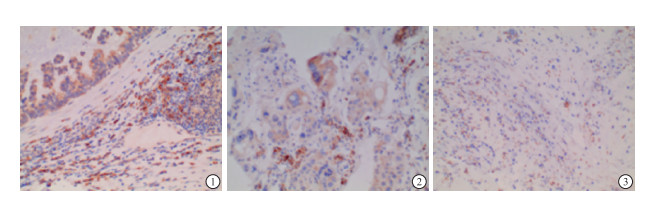
PIK3CA的表达产物位于细胞质和(或)细胞核中。细胞核和(或)细胞质的棕色染色为PIK3CA阳性染色,半定量积分法测定PIK3CA的表达。根据染色强度计算:0分, 无染色; 1分, 低染色(淡黄色); 2分, 中染色(棕黄色); 3分, 强染色(棕褐色)。所有标本在低倍镜下选择阳性染色集中区域,随机选择5个高倍镜(×400)视野。计算100个肿瘤细胞,每个样本按照标准A和标准B计算最终得分。A+B < 3分,定义为PIK3CA蛋白表达显著减少或缺失(即表达失活或失活); A+B>为3分,定义为PIK3CA蛋白阳性表达[8]。所有切片的染色结果由两名病理医生联合作出判定。
-
所有病人均首先应用PCR-ARMS法进行EGFR基因常见突变位点检测,所有经检测阳性病人,依据NCCN肺癌治疗指南给予病人应用埃克替尼,并收集病人的病理资料,肿瘤基因型,EGFR-TKIs的类型及剂量,给予病人建立随访档案。为进一步分析协同突变基因对于埃克替尼治疗疗效的影响。采用实体瘤反应评价标准(RECIST)(1.1版)进行疗效评价。PFS和OS按给予埃克替尼治疗时间计算。根据中国慈善总会病人的购药及赠药记录收集和管理数据,结果的数据截止日期是2018年12月31日。并对EGFR基因型和共存PIK3CA突变进行比对,给予病人进行PFS的数据采集。
-
采用χ2检验和生存分析。
-
62例肺癌组织标本作为实验组,PIK3CA阳性表达者总数30例,阳性信号定位于细胞质,呈棕黄色或棕褐色,阳性率达48.38%,阴性表达者32例(51.62%)(见图 1)。
-
PIK3CA表达情况与性别、年龄、TNM分期、淋巴结转移、脑转移、吸烟状态、病理类型及EGFR突变位点差异均无统计学意义(P>0.05)(见表 1)。
分组 PIK3CA 阳性率/% χ2 P + - 性别 男
女19
1116
1654.28
40.741.120 > 0.05 年龄 ≥60岁
< 60岁16
1421
1143.24
56.000.792 > 0.05 TNM分期 Ⅲb期
Ⅳ期7
239
2343.75
50.000.186 > 0.05 淋巴结转移 是
否25
525
750.00
41.660.269 > 0.05 脑转移 是
否9
2110
2247.36
48.830.011 > 0.05 吸烟 是
否9
217
2556.25
45.650.534 > 0.05 病理类型 鳞癌
腺癌4
262
3066.66
46.120.889 > 0.05 突变位点 19
2119
1118
1251.35
47.820.071 > 0.05 表 1 PIK3CA阳性表达情况与其临床资料比较
-
在PIK3CA表达阳性病人应用埃克替尼后中位疾病PFS 10.5个月(95%CI:5.6~15.4);PIK3CA表达阴性的病人应用埃克替尼后中位疾病PFS 17.0个月(95%CI:10.1~23.8)。PIK3CA阳性表达较PIK3CA阴性表达的EGFR-TKIs耐药率明显升高,2组生存曲线差异有统计学意义(χ2=7.16,P < 0.05)。
-
PIK3CA是一种体细胞突变的癌基因,现有研究[9]表明, PIK3CA基因是定位于染色体3q26.3的癌基因, 是1994年由VOLINIA等利用原位杂交技术检测发现的。位于EGFR下游, 是PI3K-Akt-mTOR通路的核心分子之一,其编码蛋白是PI3Ks催化亚单位p110。DE LAURENTIIS等[10-13]研究发现致癌基因中PIK3CA突变频率较高,在乳腺癌、结肠癌、卵巢癌和子宫内膜癌等都存在该基因的突变;VERENA等[14]利用焦磷酸测序试验显示,PIK3CA第9、20号外显子存在2个基因突变;MARLENA等[15]通过体外细胞实验,证实PF-4989216调控PI3K下游既而产生细胞凋亡、抑制细胞增殖、分化。突变的PIK3CA基因促进小细胞肺癌转移瘤的生长, NSCLC组织中PIK3CA表达上调可能预示淋巴结转移、术后复发[16];WANG等[17]研究PIK3CA与EGFR基因共表达频率高,NSCLC PIK3CA基因突变预后不良,结合PIK3CA突变与EGFR野生亚群诊断价值,提示PIK3CA基因突变可能决定NSCLC个体治疗策略。
在171例EGFR突变肿瘤病人的研究中,有20例使用至少有限的综合基因组分析平台进行了测序[18]。其中同时存在10% PIK3CA突变、5% PTEN突变等。EGFR突变型/PIK3CA表达阴性肿瘤对EGFR-TKIs的应答率、PFS、EGFR-T790m TKI耐药率和生存率均高于EGFR突变型/PIK3CA表达阴性肿瘤。在临床样本收集中显示在肺腺癌EGFR突变的病人中存在大量的并发突变,在世界癌症肺腺癌基因组图谱数据显示PIK3CA突变是常见的并发突发[19]。在先期的临床模型中,PIK3CA基因突变/激活/超表达可以下游通路参与由表皮生长因子受体通路的调节,改变机体对EGFR-TKIs治疗疗效影响[20]。这种突变如何产生目前尚不清楚,可能由于肿瘤细胞自身的肿瘤异质性或单个活检标本的采样差异性[21]。但病人出现中位疾病PFS的时间更短,这一影响需给予进一步研究,拟在进一步扩大样本,给予病人联合化疗及靶向治疗,以改善病人的中位疾病PFS。
本研究中,62例EGFR突变型的NSCLC肿瘤组织标本中, PIK3CA表达阳性为30例, 阳性率为48.38%。这与MACHADO-RUGOLO等[22]研究结果一致,在研究中发现除了p53和血管内皮生长因子外,在检测PIK3CA表达时,发现根据免疫组化检测的蛋白过表达与NGS检测的蛋白突变相结合的两种检测方法时,两种标本检测一致病例的比例很低,其在检测NSCLC中均发现类似现象,这与本研究中检测表达阳性率较高相一致。我们的数据表明, 应用免疫组化检测PIK3CA蛋白质含量可能是作为测序检测突变率较低的基因的有效补充工具。
不同病人由于遗传、临床表型和药代动力学的差异,相同疾病的不同病人观察到临床疗效的显著差异。目前的指南和药物批准辅助诊断倾向于对NSCLC中EGFR突变进行有限的单基因分析,这限制了我们对最常见的同时发生的肿瘤抑制因子和/或癌基因突变如何影响EGFR-TKIs单药治疗的临床结果的认识。在既往的临床研究中发现PIK3CA及PTEN表达与晚期NSCLC病人对于埃克替尼治疗具有预见性, 与晚期NSCLC病人对于EGFR-TKIs一/二线药物疗效具有预见性。因此,在本研究中探索在晚期EGFR突变NSCLC中进行EGFR突变检测及PIK3CA蛋白表达联合应用,将共突变谱与EGFR的反应/耐药性关联起来, 在本实验中观察在EGFR突变阳性的且PIK3CA蛋白高表达,病人对EGFR-TKIs药物疗效减弱。
本研究结果表明在中晚期EGFR突变型NSCLC中2组病人PIK3CA表达与其性别、年龄、淋巴结转移、TNM分期差异无统计学意义。当随着检测技术的不断创新,以及奥希替尼[23]、克唑替尼[24]靶向药物的临床应用病人数量增加,以及细胞毒性T淋巴细胞相关抗原4及程序性细胞死亡蛋白l免疫治疗应用于临床[25],在临床上多个基因位点的联合检测将越来越普及。
总之,我们的小样本研究强调EGFR突变肿瘤具有明显的瘤内和瘤内异质性,同时存在重要的癌症相关基因突变。一些最常见伴随改变, 包括PIK3CA基因突变/激活/超表达, 可能改变EGFR-TKIs治疗病人的临床结果。本研究从蛋白表达水平,在EGFR突变的肺癌病人,检测PIK3CA在不同标本中表达的个体差异性,为目前的单一基因分析方法在致癌基因驱动的肺癌中增加预测和/或预后信息。同时突变,特别是PIK3CA突变,在EGFR突变的肺癌中很常见,可能会改变临床结果,全面的分子分析能为单基因分析鉴定增加临床相关信息。在未来的临床治疗中通过对治疗前干预,为病人提供更好的用药指导,以进一步提高EGFR-TKIs疗效。
PIK3CA对埃克替尼治疗EGFR基因突变的非小细胞肺癌病人的疗效预测
Predicting the efficacy of taking icotinib in the non-small cell lung cancer patients with EGFR gene mutation by PIK3CA
-
摘要:
目的探讨PIK3CA对埃克替尼治疗EGFR基因突变的非小细胞癌(NSCLC)病人疗效预测价值。 方法首先应用ARMS法对确诊肺腺癌标本给予EGFR基因突变检测。对于EGFR检测阳性肺腺癌标本应用免疫组化蛋白定性法进行PIK3CA表达状态分析。对EGFR检测阳性者给予埃克替尼治疗,观察PIK3CA高表达组与低表达组埃克替尼治疗后的无进展生存期(PFS)。 结果在62例EGFR突变的肺癌病人中,48.38%同时存在PIK3CA高表达。在PIK3CA表达阳性病人应用埃克替尼后中位疾病PFS 10.5个月(95% CI:5.6~15.4);PIK3CA表达阴性的病人应用埃克替尼后中位疾病PFS 17.0个月(95% CI:10.1~23.8)。PIK3CA低表达病人用埃克替尼治疗的应答率、中位PFS、EGFR-TKIs耐药率均有较高的趋势(χ2=7.16,P < 0.05)。 结论在EGFR突变的接受埃克替尼治疗的NSCLC病人,检测PIK3CA表达状态有助于鉴别出EGFR-TKIs治疗有效较低的病人,提前进行干预,从而延长病人中位PFS。 Abstract:ObjectiveTo investigate the usage of PIK3CA in predicting the efficacy of taking icotinib in the non-small cell lung cancer (NSCLC) patients with EGFR gene mutation. MethodsFirst, the ARMS method was used to detect the mutation of EGFR gene in the specimens of lung adenocarcinoma.Then, for EGFR-positive lung adenocarcinoma specimens, immunohistochemistry was used to analyze the expression status of PIK3CA.After that, icotinib was given to EGFR-positive patients, and progression-free survival (PFS) was observed for patients with high-expression PIK3CA and low-expression PIK3CA. ResultsAmong 62 lung cancer patients with EGFR mutation, 48.38% had high expression of PIK3CA.After applying icotinib in PIK3CA-positive patients, the median disease progression-free survival period reached 10.5 months(95% CI:5.6-15.4), and the median disease progression-free survival period for PIK3CA-negative patients taking icotinib was 17.0 months (95% CI:10.1-23.8).The response rate, median PFS, and EGFR-TKIs resistance rates of patients with low expression of PIK3CA increased significantly after taking icotinib, and the differences were significant(χ2=7.16, P < 0.05). ConclusionsFor the NSCLC patients who have received EGFR-TKIs treatment, the detection of the expression status of PIK3CA can help to identify the patients with low efficacy, so early intervention can be adopted to extend the PFS. -
Key words:
- non-small cell lung cancer /
- PIK3CA /
- icotinib
-
表 1 PIK3CA阳性表达情况与其临床资料比较
分组 PIK3CA 阳性率/% χ2 P + - 性别 男
女19
1116
1654.28
40.741.120 > 0.05 年龄 ≥60岁
< 60岁16
1421
1143.24
56.000.792 > 0.05 TNM分期 Ⅲb期
Ⅳ期7
239
2343.75
50.000.186 > 0.05 淋巴结转移 是
否25
525
750.00
41.660.269 > 0.05 脑转移 是
否9
2110
2247.36
48.830.011 > 0.05 吸烟 是
否9
217
2556.25
45.650.534 > 0.05 病理类型 鳞癌
腺癌4
262
3066.66
46.120.889 > 0.05 突变位点 19
2119
1118
1251.35
47.820.071 > 0.05 -
[1] 钟锐, 邬麟, 江美林, 等.EGFR-TKIs获得性耐药的机制及其治疗的研究进展[J].肿瘤药学, 2017, 7(6):641. doi: 10.3969/j.issn.2095-1264.2017.06.01 [2] MAEMONDO M, INOUE A, KOBAYASHI K, et al.Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR[J].N Engl J Med, 2010, 362(25):2380. doi: 10.1056/NEJMoa0909530 [3] MOK TS, WU YL, THONGPRASERT S, et al.Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma[J].N Engl J Med, 2009, 361(10):947. doi: 10.1056/NEJMoa0810699 [4] MITSUDOMI T, MORITA S, YATABE Y.Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor(WJTOG3405):an open label, randomised phase 3 trial[J].Lancet Oncol, 2010, 11(2):121. doi: 10.1016/S1470-2045(09)70364-X [5] COSTA DB, KOBAYASHI SS.Whacking a molecule:clinical activity and mechanisms of resistance to third generation EGFR inhibitors in EGFR mutated lung cancers with EGFR-T790M[J].Transl Lung Cancer Res, 2015, 4(6):809. [6] KOBAYASHI S, BOGGON TJ, DAYARAM T, et al.EGFR mutation and resistance of non-small-cell lung cancer to gefitinib[J].N Engl J Med, 2005, 352(8):786. doi: 10.1056/NEJMoa044238 [7] NGUYEN KS, KOBAYASHI S, COSTA DB.Acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors in non-small-cell lung cancers dependent on the epidermal growth factor receptor pathway[J].Clin Lung Cancer, 2009, 10(4):281. doi: 10.3816/CLC.2009.n.039 [8] 倪琛琛, 于敏, 张志红.EGFR与PI3K/AKT信号通路相关蛋白在非小细胞肺癌组织中的表达及其意义[J].安徽医科大学学报, 2011, 46(12):1264. doi: 10.3969/j.issn.1000-1492.2011.12.012 [9] BAEHMAN KE, ARGANI P, SAMUELS Y, et al.The PIK3CA gene is mutated with high frequency in human breast cancers[J].Cancer Biol Ther, 2004, 3(8):772. doi: 10.4161/cbt.3.8.994 [10] DE LAURENTⅡS A, PARDO OE, PALAMIDESSI A, et al.The catalytic class Ⅰ(A) PI3K isoforms play divergent roles in breast cancer cell migration[J].Cell Signal, 2011, 23(3):529. doi: 10.1016/j.cellsig.2010.10.021 [11] BABA Y, NOSHO K, SHIMA K, et al.Phosphorylated AKT expression is associated with PIK3CA mutation, low stage, and favorable outcome in 717 colorectal cancers[J].Cancer, 2011, 117(7):1399. doi: 10.1002/cncr.25630 [12] DE MARCO C, RINALDO N, BRUNI P, et al.Multiple genetic alterations within the PI3K pathway are responsible for AKT activation in patients with ovarian carcinoma[J].PLoS One, 2013, 8(2):e55362. doi: 10.1371/journal.pone.0055362 [13] WEIGELT B, WARNE PH, LAMBROS MB, et al.PI3K pathway dependencies in endometrioid endometrial cancer cell lines[J].Clin Cancer Res, 2013, 19(13):3533. doi: 10.1158/1078-0432.CCR-12-3815 [14] VERENA S, JESSICA LU, JAN D, et al.Identification of uncommon PIK3CA mutations in lung cancer by using pyrosequencing[J].Diagn Mol Pathol, 2013, 22(1):22. doi: 10.1097/PDM.0b013e31825f5f93 [15] MARLENA W, SANGITA M, PRAMOD P, et al.targeting small cell lung cancer harboring mutation with PIK3CA a selective oral PI3K inhibitor PF-4989216[J].Clin Cancer Res, 2014, 20(3):631. doi: 10.1158/1078-0432.CCR-13-1663 [16] ZHAO Q, ZHANG B, SHAO YF, et al.Correlation between the expression levels of miR-1 and PIK3CA in non-small cell lung cancer and their relationship with clinical characteristics and prognosis[J].Future Oncol, 2014, 10(1):49. doi: 10.2217/fon.13.242 [17] WANG L, HU HC, PAN YJ, et al.PIK3CA mutations frequently coexist with EGFR/KRAS mutations in non-small cell lung cancer and suggest poor prognosis in EGFR/KRAS wildtype subgroup[J].PLoS One, 2014, 9(2):e88291. doi: 10.1371/journal.pone.0088291 [18] VANDERLAAN PA, RANGACHARI D, MOCKUS SM, et al.Mutations in TP53, PIK3CA, PTEN and other genes in EGFR mutatedlung cancers:correlation with clinical outcomes[J].Lung Cancer, 2017, 106(4):17. [19] COLLISSON EA, CAMPBELL JD.Brooks AN.Cancer genome atlas research network.comprehensive molecular profiling of lung adenocarcinoma[J].Nature, 2014, 511(7):543. [20] COSTA DB, HALMOS B, KUMAR A, et al.BIM mediates EGFR tyrosine kinaseinhibitor-induced apoptosis in lung cancers with oncogenic EGFR mutations[J].PLoS Med, 2007, 4(10):1669. [21] MCGRANAHAN N, FAVERO F, DE BRUIN EC, et al.Clonal status of actionable driver events and the timing of mutationalprocesses in cancer evolution[J].Sci Transl Med, 2015, 283(7):54. [22] MACHADO-RUGOLO J, FABRO AT, ASCHERI D, et al.Usefulness of complementary next-generation sequencing and quantitative immunohistochemistry panels for predicting brain metastases and selecting treatment outcomes of non-small cell lung cancer[J].Hum Pathol, 2019, 83(1):177. [23] SULLIVAN I, PLANCHARD D.Osimertinib in the treatment of patients with epidermal growth factor receptor T790M mutation-positive metastatic non-small cell lung cancer:clinical trial evidence and experience[J].Ther Adv Respir Dis, 2016, 10(6):54. [24] DURUISSEAUX M, BESSE B, CADRANEL J, et al.Overall survival with crizotinib and next-generation ALK inhibitors in ALK-positive non-small-cell lung cancer(IFCT-1302 CLINALK):a French nationwide cohort retrospective study[J].Oncotarget, 2017, 8(13):21903. [25] HELLMANN MD, NATHANSON T, RIZVI H, et al.Genomic features of response to combination immunotherapy in patients with advanced non-small-cell lung cancer[J].Cancer Cell, 2018, 33(5):843. doi: 10.1016/j.ccell.2018.03.018 -




 下载:
下载: