-
结肠癌是一种常见的消化道恶性肿瘤,死亡率仅次于肺癌、肝癌及胃癌,居第4位,对人类的生命健康造成极大危害[1]。随着医疗水平的不断提升,结肠癌在诊断及治疗方面已取得明显进步,但是药物治疗不仅疗效不理想,而且还带来明显的不良反应及结肠癌细胞的转移[2]。因此,开发更有效的化疗药物已成为抗肿瘤治疗研究的热点。萝卜硫素是一种从十字花科植物中提取的异硫氰酸盐,研究[3-6]发现其具有抗肿瘤、抗氧化、抗炎、免疫调节等药理活性,然而萝卜硫素抗结肠癌的作用机制报道甚少。转化生长因子β1(TGF-β1)信号通路是细胞内重要的信号传导途径,可介导结肠癌细胞的增殖、分化、凋亡及迁移等生物学过程,抑制TGF -β1/Smad信号通路活化可以降低结肠癌细胞增殖、并诱导其凋亡[7-10]。本实验探讨萝卜硫素对结肠癌HT-29细胞增殖的抑制作用及其对细胞中TGF-β1/Smad信号通路的影响,以期为其开发应用提供理论依据。
-
结肠癌HT-29细胞购自武汉巴菲尔生物有限公司;胎牛血清、RPMI-1640培养基及0.25%胰蛋白酶-EDTA购自英国Abcam公司;萝卜硫素(批号H2011265418,纯度≥98%)购于山东绿叶制药有限公司;鼠抗人p-Smad3、Smad4、Cyclin D1及c-Myc抗体由美国CST公司提供,HRP标记山鼠抗兔IgG由上海谷歌生物有限公司提供;HRP发光试剂盒购自Millipore公司;TGF-β1 ELISA试剂盒购自武汉华美生物有限公司;CCK8试剂盒购自北京智杰方远科技有限公司。
-
BIO-RAD-550型酶标仪(美国Bio-Rad公司),BBS-V800型单人超净台(山东鑫贝西公司),SPX-250型细胞培养箱(美国Thermo公司),GE-100型凝胶电泳仪(北京六一仪器厂)。
-
采用RPMI-1640完全培养基培养,条件为37 ℃、5%CO2浓度的恒湿培养箱,用0.25%胰蛋白酶-EDTA消化传代培养。当细胞生长状态良好时,取对数生长期细胞用于后续实验。
-
取对数生长期细胞消化计数,以5×104/mL细胞密度接种于96孔板中,培养过夜。实验组按照不同浓度10、20、40 μmol/L加入萝卜硫素溶液,对照组加入等体积培养基,药物干预24、48、72 h后弃去培养基,每孔加入100 μL含10 μL CCK8试剂的培养液,继续培养2 h,在450 nm波长处检测每孔吸光度(OD)值。
-
取对数生长期HT-29细胞,消化、重悬、计数后,以1×105/mL的密度接种于6孔板,培养24 h后,加入10、20、40 μmol/L萝卜硫素干预细胞24 h,收集细胞,按照试剂盒操作说明书分别加入Annexin-V FITC和PI处理细胞,用流式细胞仪检测细胞凋亡情况。
-
收集“1.5”项各孔细胞培养液,1 000 r/min离心5 min,取上清液。按照ELISA试剂盒说明书操作,检测各组上清液中TGF β1水平。
-
收集“1.5”项各孔细胞,采用RIPA裂解液冰上充分裂解细胞,并测定细胞匀浆中蛋白浓度,每个样品取50 μg蛋白上样电泳。待蛋白完全分离后进行蛋白转膜实验,然后用5%脱脂奶粉封闭1 h,TBST洗膜3次后将对应条带置于p-Smad3、Smad4、Cyclin D1及c-Myc抗体溶液(1: 3 000稀释)中,4 ℃摇床反应过夜。次日洗膜后将条带置于HRP标记IgG溶液中室温反应1 h,洗膜3次,HRP试剂显影后进行蛋白条带灰度值分析。
-
采用方差分析和q检验。
-
10、20、40 μmol/L萝卜硫素组细胞OD值相对于对照组明显降低(P < 0.01);同一浓度萝卜硫素处理48 h和72 h,细胞OD值相比于24 h也降低(P < 0.05~P < 0.01)。40 μmol/L萝卜硫素干预细胞72 h后,细胞抑制率高达76.25%,而低浓度萝卜硫素对HT-29细胞增殖抑制率表现出时间依赖性(P < 0.05)(见表 1)。
分组 n 24 h OD值 48 h OD值 72 h OD值 F P MS组内 对照组 3 1.4251±0.018 4 1.635 1±0.021 5 1.485 7±0.030 5 60.74 < 0.01 0.001 10 μmol/L萝卜硫素 3 1.014 3±0.016 5++ 0.951 7±0.021 6++* 0.627 1±0.020 4++**# 336.64 < 0.01 0.000 20 μmol/L萝卜硫素 3 0.540 6±0.012 6++▲▲ 0.485 1±0.013 5++*▲▲ 0.395 1±0.016 2++**#▲▲ 80.53 < 0.01 0.000 40 μmol/L萝卜硫素 3 0.385 1±0.009 4++▲▲■ 0.410 5±0.011 8++*▲▲■ 0.352 8±0.012 7++**#▲▲ 19.91 0.01 0.000 F — 3 120.49 3 043.56 1 894.27 — — — P — < 0.01 < 0.01 < 0.01 — — — MS组内 — 0.000 0.000 0.000 — — — q检验:与24 h比较*P < 0.05,** P < 0.01;与48 h比较##P < 0.01;与对照组比较++P < 0.01;与萝卜硫素10 μmol/L比较▲▲P < 0.01;与萝卜硫素20 μmol/L比较■■P < 0.01 表 1 各组间HT-29细胞OD值比较(x±s)
-
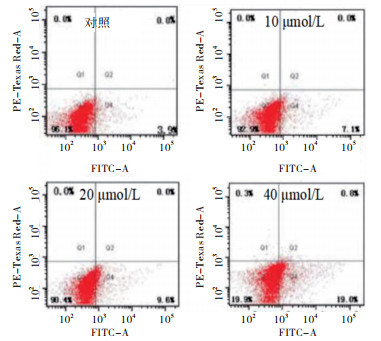
10、20、40 μmol/L萝卜硫素干预HT-29细胞24 h,细胞凋亡率均高于对照组(P < 0.05~P < 0.01)(见图 1、表 2)。
分组 n 细胞凋亡率/% 对照组 3 3.85±0.61 10 μmol/L萝卜硫素 3 7.35±0.84* 20 μmol/L萝卜硫素 3 10.57±1.16**# 40 μmol/L萝卜硫素 3 19.53±1.84**##▲ F — 93.35 P — < 0.01 MS组内 — 1.452 q检验:与对照组比较*P < 0.05,** P < 0.01;与10 μmol/L萝卜硫素比较#P < 0.05,##P < 0.01;与20 μmol/L萝卜硫素比较▲P < 0.05 表 2 各组细胞凋亡率比较(x±s)
-
ELISA结果表明,药物处理HT-29细胞24 h,10、20、40 μmol/L萝卜硫素组细胞培养液中TGF-β1水平均低于对照组(P < 0.05~P < 0.01)(见表 3)。
分组 n TGF β1/β-actin 对照组 3 1 315.26±40.17 10 μmol/L萝卜硫素 3 1 141.39±26.37* 20 μmol/L萝卜硫素 3 892.54±20.38**# 30 μmol/L萝卜硫素 3 627.43±15.27**##▲ F — 364.63 P — < 0.01 MS组内 — 739.381 q检验:与对照组比较*P < 0.05,** P < 0.01;与10 μmol/L萝卜硫素比较#P < 0.05,##P < 0.01;与20 μmol/L萝卜硫素比较▲P < 0.05 表 3 各组细胞TGF β1水平比较(x±s)
-
与对照组相比,10、20、40 μmol/L萝卜硫素均能抑制p-Smad3、Smad4、Cyclin D1及c-Myc蛋白表达(P < 0.05~P < 0.01)(见图 2、表 4)。
分组 n p-Smad3/β-actin Smad4/β-actin Cyclin D1/β-actin c-Myc/β-actin 对照组 3 0.41±0.05 0.57±0.04 0.36±0.05 0.63±0.04 10 μmol/L萝卜硫素 3 0.24±0.03* 0.34±0.05* 0.22±0.04* 0.31±0.05* 20 μmol/L萝卜硫素 3 0.15±0.03** 0.21±0.03** 0.14±0.03** 0.16±0.03** 30 μmol/L萝卜硫素 3 0.07±0.02**##▲ 0.17±0.03**#▲ 0.06±0.03**#▲ 0.13±0.02**#▲ F — 54.36 66.08 33.29 116.50 P — < 0.01 < 0.01 < 0.01 < 0.01 MS组内 — 0.001 0.001 0.001 0.001 q检验:与对照组比较*P < 0.05,** P < 0.01;与10 μmol/L萝卜硫素比较#P < 0.05,##P < 0.01;与20 μmol/L萝卜硫素比较▲P < 0.05 表 4 各组p-Smad3、Smad4、Cyclin D1及c-Myc蛋白表达比较(x±s)
-
结肠癌属于常见的恶性肿瘤,其发病率及死亡率较高,对人类健康威胁极大,统计发现全球每年约有50万病人死于结肠癌[11]。当前对结肠癌的诊疗水平有了大幅度提升,但是其预后效果并不理想。天然中药的有效单体成分在多种肿瘤治疗中均表现出明显的治疗效果[12-13]。本研究发现,萝卜硫素对结肠癌HT-29细胞的增殖具有明显抑制作用,并能诱导其凋亡,进一步研究发现该作用可能与萝卜硫素下调TGF β1表达,抑制TGF β1/Smad信号传导相关。
采用ELISA分析HT-29细胞培养液上清中TGF β1蛋白水平,结果显示萝卜硫素对TGF-β1表达具有抑制作用。研究[7-10]发现TGF-β1/Smad信号通路在调控结肠癌细胞的增殖方面具有重要意义,可通过抑制TGF-β1蛋白表达阻滞TGFβ1/Smad信号通路的信号传导,从而降低肿瘤细胞的增殖能力。由此提示,萝卜硫素对结肠癌HT-29细胞的增殖抑制作用可能与其阻滞结肠癌HT-29细胞中TGF-β1/Smad信号传导相关。TGF-β1/Smad信号通路在细胞内发挥着重要的作用,p-Smad3、Smad4为TGF-β1/Smad通路中重要的信号分子,其中p-Smad3、Smad4蛋白表达量的改变对肿瘤细胞的增殖、凋亡等生物学行为具有重要的调控作用[14-15]。本实验结果发现HT-29细胞经萝卜硫素干预后,p-Smad3、Smad4表达水平随之降低。相关报道[14]提出TGF-β1/Smad信号通路通过调控Cyclin D1及c-Myc蛋白表达进一步调节肿瘤细胞的增殖。研究[14-16]发现对TGF-β1/Smad信号通路进行抑制,能够显著下调Cyclin D1及c-Myc蛋白表达,进而抑制癌细胞增殖。此外,TGF-β1/Smad信号传导受到抑制时,Cyclin D1及c-Myc蛋白表达也随之下降,并与p-Smad3、Smad4蛋白表达的抑制具有相似的趋势。研究[17-18]发现萝卜硫素可通过降低TGF-β1/Smad信号传导活性进而抑制肝纤维化及肌肉纤维化。
综上,萝卜硫素可能通过调节TGF-β1/Smad信号通路分子TGF-β1、p-Smad3、Smad4蛋白表达,阻滞TGF-β1/Smad信号传导,进而下调Cyclin D1及c-Myc蛋白表达,从而抑制结肠癌HT-29细胞增殖。
萝卜硫素调控TGF-β1/Smad信号通路抑制结肠癌HT-29细胞增殖的机制研究
Sulforaphane inhibits proliferation of colon cancer HT-29 cells via regulating TGF β1/Smad signaling pathway
-
摘要:
目的研究萝卜硫素对结肠癌HT-29细胞增殖的影响及其可能的作用机制。 方法采用CCK8分析萝卜硫素对HT-29细胞增殖的影响,流式细胞术检测HT-29细胞凋亡,ELISA检测细胞培养液中转化生长因子β1(TGF-β1)的水平,Western blotting检测细胞中p-Smad3、Smad4、Cyclin D1及c-Myc蛋白表达。 结果CCK8结果显示,随着药物浓度和作用时间的增加,萝卜硫素对HT-29细胞的增殖抑制作用增强(P < 0.05~P < 0.01)。10、20、40 μmol/L萝卜硫素干预HT-29细胞24 h,细胞凋亡率均高于对照组(P < 0.05~P < 0.01)。ELISA结果表明,药物处理HT-29细胞24 h,10、20、40 μmol/L萝卜硫素组细胞培养液中TGF β1水平均低于对照组(P < 0.05~P < 0.01)。与对照组相比,10、20、40 μmol/L萝卜硫素均能抑制p-Smad3、Smad4、Cyclin D1及c-Myc蛋白表达(P < 0.05~P < 0.01)。 结论萝卜硫素可抑制结肠癌HT-29细胞的增殖,其作用机制可能与调控TGF-β1/Smad信号通路有关。 -
关键词:
- 结肠肿瘤 /
- 萝卜硫素 /
- 增殖 /
- TGF β1/Smad信号通路
Abstract:ObjectiveTo study the effect of sulforaphane on the proliferation of colon cancer HT-29 cells and its potential mechanism. MethodsCCK8 was used to detect the effect of sulforaphane on the proliferation of HT-29 cells.Flow cytometry was applied to determine the apoptosis of HT-29 cells.ELISA was performed to measure the levels of transforming growth factor β1 (TGF-β1).Western blotting was carried out to analyze the expression of p-Smad3, Smad4, Cyclin D1 and c-Myc protein. ResultsCCK8 results showed that the inhibitory effect of sulforaphane on the proliferation of HT-29 cells increased with the increase of drug concentration and time (P < 0.05 to P < 0.01).The apoptosis rate of HT-29 cells treated with 10, 20, 40 μmol/L sulforaphane was higher than that in control group (P < 0.05 to P < 0.01).The results of ELISA showed that the level of TGF-β1 in cell culture medium in 10, 20, 40 μmol/L sulforaphane group was lower than that in control group (P < 0.05 to P < 0.01).The expression of p-Smad3, Smad4, Cyclin D1 and c-Myc protein in HT-29 cells after treatment with 10, 20, 40 μmol/L sulforaphane was decreased compared with control group (P < 0.05 to P < 0.01). ConclusionsSulforaphane can inhibit the proliferation of colon cancer HT-29 cells, the mechanism of which might be related to the regulation of TGF-β1/Smad signaling pathway. -
Key words:
- colon neoplasms /
- sulforaphane /
- proliferation /
- TGF-β1/Smad signaling pathway
-
表 1 各组间HT-29细胞OD值比较(x±s)
分组 n 24 h OD值 48 h OD值 72 h OD值 F P MS组内 对照组 3 1.4251±0.018 4 1.635 1±0.021 5 1.485 7±0.030 5 60.74 < 0.01 0.001 10 μmol/L萝卜硫素 3 1.014 3±0.016 5++ 0.951 7±0.021 6++* 0.627 1±0.020 4++**# 336.64 < 0.01 0.000 20 μmol/L萝卜硫素 3 0.540 6±0.012 6++▲▲ 0.485 1±0.013 5++*▲▲ 0.395 1±0.016 2++**#▲▲ 80.53 < 0.01 0.000 40 μmol/L萝卜硫素 3 0.385 1±0.009 4++▲▲■ 0.410 5±0.011 8++*▲▲■ 0.352 8±0.012 7++**#▲▲ 19.91 0.01 0.000 F — 3 120.49 3 043.56 1 894.27 — — — P — < 0.01 < 0.01 < 0.01 — — — MS组内 — 0.000 0.000 0.000 — — — q检验:与24 h比较*P < 0.05,** P < 0.01;与48 h比较##P < 0.01;与对照组比较++P < 0.01;与萝卜硫素10 μmol/L比较▲▲P < 0.01;与萝卜硫素20 μmol/L比较■■P < 0.01 表 2 各组细胞凋亡率比较(x±s)
分组 n 细胞凋亡率/% 对照组 3 3.85±0.61 10 μmol/L萝卜硫素 3 7.35±0.84* 20 μmol/L萝卜硫素 3 10.57±1.16**# 40 μmol/L萝卜硫素 3 19.53±1.84**##▲ F — 93.35 P — < 0.01 MS组内 — 1.452 q检验:与对照组比较*P < 0.05,** P < 0.01;与10 μmol/L萝卜硫素比较#P < 0.05,##P < 0.01;与20 μmol/L萝卜硫素比较▲P < 0.05 表 3 各组细胞TGF β1水平比较(x±s)
分组 n TGF β1/β-actin 对照组 3 1 315.26±40.17 10 μmol/L萝卜硫素 3 1 141.39±26.37* 20 μmol/L萝卜硫素 3 892.54±20.38**# 30 μmol/L萝卜硫素 3 627.43±15.27**##▲ F — 364.63 P — < 0.01 MS组内 — 739.381 q检验:与对照组比较*P < 0.05,** P < 0.01;与10 μmol/L萝卜硫素比较#P < 0.05,##P < 0.01;与20 μmol/L萝卜硫素比较▲P < 0.05 表 4 各组p-Smad3、Smad4、Cyclin D1及c-Myc蛋白表达比较(x±s)
分组 n p-Smad3/β-actin Smad4/β-actin Cyclin D1/β-actin c-Myc/β-actin 对照组 3 0.41±0.05 0.57±0.04 0.36±0.05 0.63±0.04 10 μmol/L萝卜硫素 3 0.24±0.03* 0.34±0.05* 0.22±0.04* 0.31±0.05* 20 μmol/L萝卜硫素 3 0.15±0.03** 0.21±0.03** 0.14±0.03** 0.16±0.03** 30 μmol/L萝卜硫素 3 0.07±0.02**##▲ 0.17±0.03**#▲ 0.06±0.03**#▲ 0.13±0.02**#▲ F — 54.36 66.08 33.29 116.50 P — < 0.01 < 0.01 < 0.01 < 0.01 MS组内 — 0.001 0.001 0.001 0.001 q检验:与对照组比较*P < 0.05,** P < 0.01;与10 μmol/L萝卜硫素比较#P < 0.05,##P < 0.01;与20 μmol/L萝卜硫素比较▲P < 0.05 -
[1] GREMILLION CL, SAVAGE M, COHEN EB.Radiographic findings and clinical factors in dogs with surgically confirmed or presumed colonictorsion[J].Vet Radiol Ultrasound, 2018, 59(3):272. doi: 10.1111/vru.12595 [2] WANG M, WU G, HOU X, et al.LIN28B promotes colon cancer migration and recurrence[J].PLoS One, 2014, 9(10):e109169. doi: 10.1371/journal.pone.0109169 [3] TORTORELLA SM, ROYCE SG, LICCIARDI PV, et al.Dietarysulforaphane in cancer chemoprevention:the role of epigenetic regulation and HDAC inhibition[J].Antioxid Redox Signal, 2015, 22(16):1382. doi: 10.1089/ars.2014.6097 [4] NALLASAMY P, SI H, BABU PV, et al.Sulforaphane reduces vascular inflammation in mice and prevents TNF-α-induced monocyte adhesion to primary endothelial cells through interfering with the NF-κB pathway[J].J Nutr Biochem, 2014, 25(8):824. doi: 10.1016/j.jnutbio.2014.03.011 [5] BAI Y, CHEN Q, SUN YP, et al.Sulforaphane protection against the development of doxorubicin-induced chronic heart failure is associated with Nrf2 upregulation[J].Cardiovasc Ther, 2019, 35(5):53. [6] Al-HARBI NO, NADEEM A, AHMAD SF, et al.Sulforaphane treatment reverses corticosteroid resistance in a mixed granulocytic mouse model of asthma by upregulation of antioxidants and attenuation of Th17 immune responses in the airways[J].Eur J Pharmacol, 2019, 855(5):276. [7] HAWINKELS LJ, PAAUWE M, VERSPAGET HW, et al.Interaction with colon cancer cells hyperactivates TGF-β signaling in cancer-associated fibroblasts[J].Oncogene, 2014, 33(1):97. [8] CHEN S, ZHU J, ZUO S, et al.1, 25(OH)2D3 attenuates TGF-β1/β2-induced increased migration and invasion via inhibiting epithelial-mesenchymal transition in colon cancer cells[J].Biochem Biophys Res Commun, 2015, 468(1/2):130. [9] GONZALEZ-ZUBELDIA I, DOTOR J, REDRADO M, et al.Co-migration of colon cancer cells and CAFs induced by TGFβ enhances liver metastasis[J].Cell Tissue Res, 2015, 359(3):829. doi: 10.1007/s00441-014-2075-6 [10] SUN X, LAN C.Study on the expression of Runx3 and TGF-β1 protein in the colonic tissue from rats with irritable bowel syndrome[J].Asian Pac J Trop Med, 2011, 4(2):88. doi: 10.1016/S1995-7645(11)60043-6 [11] ORANGIO GR.The economics of colon cancer[J].Surg Oncol Clin N Am, 2018, 27(2):327. [12] WU K, YANG QJ, MU YQ, et al.Berberine inhibits the proliferation of colon cancer cells by inactivating Wnt/β-catenin signaling[J].Int J Oncol, 2012, 41(23):292. [13] HE BC, GAO JL, LUO XJ, et al.Ginsenoside Rg3 inhibits colorectal tumor growth through the down-regulation of Wnt/β-catenin signaling[J].Int J Oncol, 2011, 38(11):437. [14] CHEN L, YANG T, LU DW, et al.Central role of dysregulation of TGF-β/Smad in CKD progression and potential targets of its treatment[J].Biomed Pharmacother, 2018, 101(4):670. [15] KIM KO, SAMPSON ER, MAYNARD RD, et al.Ski inhibits TGF-beta/phospho-Smad3 signaling and accelerates hypertrophic differentiation in chondrocytes[J].J Cell Biochem, 2012, 113(6):2156. [16] ZHENG L, SUZUKI H, NAKAJO Y, et al.Regulation of c-MYC transcriptional activity by transforming growth factor-beta 1-stimulated clone 22[J].Cancer Sci, 2018, 109(2):395. [17] OH CJ, KIM JY, MIN AK, et al.Sulforaphaneattenuates hepatic fibrosis via NF-E2-related factor 2-mediated inhibition of transforming growth factor-β/Smadsignaling[J].Free Radic Biol Med, 2012, 52(3):671. doi: 10.1016/j.freeradbiomed.2011.11.012 [18] SUN C, LI S, LI D.Sulforaphane mitigates muscle fibrosis in mdx mice via Nrf2-mediated inhibition of TGF-β/Smad signaling[J].J Appl Physiol, 2016, 120(4):377. -





 下载:
下载: