-
2019年12月以来,中国湖北省武汉市出现了新型冠状病毒感染的肺炎病人,并迅速蔓延到我国其他地区。2020年1月12日,WHO正式将引起肺炎疫情的新型冠状病毒命名为2019新型冠状病毒(2019 new coronavirus,2019-nCoV),属于β属冠状病毒,其基因组和蝙蝠SARS样冠状病毒(bat-SL-CoVZC45)高度同源,相似度85%以上[1];由其感染引起的肺炎称之为新型冠状病毒肺炎(COVID-19)。临床表现主要有发热、干咳、乏力,重症病人多在发病1周后出现呼吸困难和/或低氧血症,严重者快速进展为急性呼吸窘迫综合征、脓毒症休克、代谢性酸中毒等,实验室检查可出现多种指标的变化,主要表现为发病早期外周血白细胞总数正常或降低,淋巴细胞计数绝对值减少,C反应蛋白(CRP)升高,重症病人常有炎症因子升高并伴有外周血淋巴细胞进行性减少[2]。所以实验室检查对COVID-19的早期筛查和治疗,提高COVID-19的治愈率非常重要。CRP是机体在应激状态下由肝脏合成的一种急性时相反应蛋白,升高常见于细菌感染,在病毒感染时一般不升高或仅有轻微升高。当机体受到感染后,其升高的幅度与感染的程度呈正相关并可早于白细胞升高,故其有较高的敏感性[3]。中性粒细胞淋巴细胞比值(NLR)作为机体炎症反应的生物标志物,可直观地反映机体的炎症状态和全身免疫系统状态,越来越受到炎症相关研究的极大重视。本文旨在研究淋巴细胞(LY)、CRP联合NLR检测在COVID-19早期诊断中的临床应用价值。现作报道。
-
选取2020年1-2月我院收治的53例COVID-19确诊病人进行回顾性分析,其中男30例,女23例;年龄21~87岁。同时选取同期90例已排除新型冠状病毒感染的其他呼吸系统疾病病人作为对照组,其中男44例,女46例;年龄5~86岁。确诊病例和排除病例标准均严格按照国家卫健委发布的新型冠状病毒感染肺炎的诊疗方案(第7版本)执行。
-
血常规检测采用日本希斯美康公司XE-5000型自动血常规分析流水线,由中性粒细胞绝对数和淋巴细胞绝对数计算出NLR值;CRP检测应用罗氏公司生产的Cobas 8000型全自动生化分析仪。采用分析仪原装配套试剂、校准品及质控品,所有标本检测均严格按照试剂盒说明书及相应仪器标准操作程序进行操作,并进行实验室质量控制。
-
以EDTA-K2抗凝管采集受检者静脉血2 mL用于血常规检测。以肝素抗凝管采集受检者静脉血3~5 mL,3 000 r/min,离心5 min,用于CRP的检测。
-
采用独立样本t检验和ROC曲线评价各指标诊断价值。
-
COVID-19组NLR、CRP水平均高于对照组,LY水平低于对照组(P < 0.01)(见表 1)。
分组 n NLR LY/(×109/L) CRP/(mg/L) 对照组 90 3.20±1.60 1.80±0.94 6.96±10.93 COVID-19组 53 6.40±7.16 1.21±0.56 57.93±66.47 t — 3.21 4.70 5.54 P — < 0.01 < 0.01 < 0.01 表 1 2组间各指标检测结果比较(x±s)
-
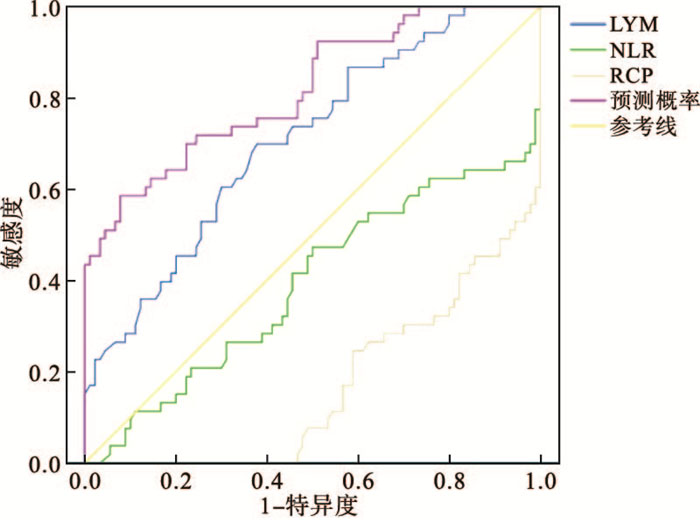
以2019-nCoV核酸检测结果作为金标准,评价NLR、LY、CRP对COVID-19诊断的价值。NLR、LY、CRP单独及NLR+LY+CRP联合用于诊断COVID-19曲线下面积(AUC)依次为:0.610、0.705、0.833、0.817,最佳截断界值(cut-off值)分别为6.23、1.41、11.80,灵敏度分别为31.0%、69.8%、67.9%、58.5%,特异度分别为96.7%、62.2%、80.0%、92.2%,NLR+LY+CRP联合诊断价值高于单独诊断价值(见表 2、图 1)。
指标 AUC cut-off值 灵敏度/% 特异度/% SE P 95%CI NLR 0.610 6.23 34.0 96.7 3.210 < 0.01 0.552~0.712 LY 0.705 1.41 69.8 62.2 4.700 < 0.01 0.625~0.777 CRP 0.833 11.80 67.9 80.0 5.540 < 0.01 0.420~0.590 NLR+LY+CRP 0.817 — 58.5 92.2 — — 0.644~0.790 表 2 各指标对COVID-19诊断价值分析
-
由于COVID-19潜伏期较长,人群普遍易感,并且重症、危重症病人死亡率较高,故加强对COVID-19早诊断、早治疗对提高治愈率、降低死亡率有非常重要的意义,同时也能最大可能地避免医院感染。目前临床诊断COVID-19的“金标准”是2019-nCoV的核酸检测,但其受标本采集、人员操作等因素的影响,可能导致假阴性率过高,同时核酸检测无法做到随到随检,耗时亦较长。而实验室检查结合流行病学调查及影像学检查能给临床早期诊断COVID-19提供有价值的参考。本文通过对我院收治的53例COVID-19确诊病人的实验室检查结果的回顾性分析和研究,旨在探讨NLR、LY、CRP检测在COVID-19早期诊断中的应用价值。
本研究所采用的数据均为病人首诊的检查结果,所以能准确反映病人入院时的状态,也能为临床的早期诊断提供参考。结果发现,COVID-19组的WBC计数正常或者偏低,符合病毒性感染血象变化的一般特点,而LY计数减少[4],这又区别于一般的病毒感染,笔者认为这可能是2019-nCoV可以破坏病人的免疫系统从而导致LY减少,对此周玉平等[5]亦有过报道,由此可以推断LY的减少程度与疾病的严重程度成正相关,这和与其高度同源性的蝙蝠样SARS病毒破坏人体免疫系统非常相似[6-7]。同时在研究过程中还发现有部分病人的WBC升高明显,这有可能是因为病情处于进展期或者病人为重症(危重症)[8]。
CRP作为急性时相反应蛋白,当机体发生感染时,尤其在细菌感染时升高较为明显[9],而在病毒感染时一般不升高或轻微升高,本文研究显示,COVID-19组的CRP水平较对照组明显增高(P < 0.01)。由于CRP受体内某些细胞炎症因子调控,特别是白细胞介素-6(IL-6)水平的影响,当体内IL-6升高,能够刺激肝细胞产生CRP[10],而笔者在对COVID-19病人的实验室结果分析时,发现多数COVID-19病人的IL-6水平均有不同程度的升高。同时CRP在炎症反应中起着积极作用,其与WBC存在正相关,当感染发生后,CRP早于WBC升高,回复正常也较快,故具有极高的敏感性。
NLR是反映机体炎症状态的一个指标,其在炎性反应中具有重要作用,中性粒细胞是天然免疫细胞,具有反应迅速的特点;LY是适应性免疫细胞,具有免疫防御功能。我们在上文中已经讨论过COVID-19病人的白细胞正常或偏低,LY下降,从而导致NLR升高,说明NLR对COVID-19病人和非COVID-19病人的鉴别有一定的临床价值。在结果分析中,我们还发现有些COVID-19病人的白细胞升高,而LY严重下降,NLR升高幅度较大,后经临床资料分析发现这些病人均为临床危重型,这说明NLR也是影响重症发生的重要因素之一,具有良好的预测价值[11],在临床早期鉴别诊断普通型和危重型的过程中具有重要的意义。
采用ROC曲线分析NLR、LY、CRP单独及联合用于诊断COVID-19的AUC依次为:0.610、0.705、0.833、0.817,cut-off值分别为6.23、1.41、11.80,灵敏度分别为31.0%、69.8%、67.9%、58.5%,特异度分别为96.7%、62.2%、80.0%、92.2%。采用NLR、LY、CRP联合检测能够提高COVID-19诊断的灵敏度和准确性,因此,对上述检测指标应联合起来综合分析,结合流行病学史及肺部影像学结果,提高COVID-19的早期诊断水平,从而指导临床及早采取合理的隔离及治疗措施,避免交叉感染,提高疾病治愈率。
淋巴细胞、C反应蛋白联合外周血中性粒细胞淋巴细胞比值检测在新型冠状病毒肺炎早期诊断中的应用价值
Value of lymphocyte, C reactive protein combined with peripheral blood neutrophil to lymphocyte ratio in the early diagnosis of COVID-19
-
摘要:
目的探讨中性粒细胞淋巴细胞比值(NLR)、淋巴细胞(LY)和C反应蛋白(CRP)单独与联合检测在新型冠状病毒肺炎(COVID-19)早期诊断中的临床应用价值。 方法选取对53例COVID-19病人(COVID-19组)以及同期90例其他呼吸系统疾病病人(对照组),回顾性分析和比较2组病人中性粒细胞淋巴细胞比值(NLR)、淋巴细胞(LY)和C反应蛋白(CRP)的表达水平。并利用ROC曲线分析NLR、LY、CRP单独以及NLR+LY+CRP联合检测对COVID-19的早期诊断价值。 结果COVID-19组NLR、CRP水平均高于对照组,LY水平低于对照组(P < 0.01)。NLR、LY、CRP单独及联合用于COVID-19感染诊断的ROC曲线下面积分别为0.610、0.705、0.833、0.817;灵敏度分别为31.0%、69.8%、67.9%、58.5%;特异度分别为96.7%、62.2%、80.0%、92.2%,NLR+LY+CRP联合诊断的价值高于单独诊断价值。 结论NLR、LY、CRP联合诊断可以提高COVID-19早期诊断的特异度和灵敏度,从而指导临床尽早采取合理治疗措施,提高COVID-19的治愈率。 -
关键词:
- 新型冠状病毒肺炎 /
- 淋巴细胞 /
- 中性粒细胞淋巴细胞比值 /
- C反应蛋白 /
- ROC曲线
Abstract:ObjectiveTo investigate the clinical application value of the single and combined detection of the neutrophil to lymphocyte ratio(NLR), lymphocyte(LY) and C reactive protein(CRP) in the early diagnosis of corona virus disease 2019(COVID-19). MethodsFifty-three patients with COVID-19 and 90 patients with other respiratory diseases were divided into the COVID-19 group and control group, respectively.The levels of NLR, LY and CRP between two groups were retrospectively analyzed and compared.The ROC curve was used to analyze the application value of the single and combined detection of NLR, LY and CRP in the early diagnosis of COVID-19. ResultsThe levels of NLR and CRP in COVID-19 group were higher than those in control group, and the level of LY in COVID-19 group was significantly lower than that in control group(P < 0.01).The area under the ROC curve of the single and combined detection of NLR, LY and CRP in the diagnosis of COVID-19 were 0.610, 0.705, 0.833 and 0.817, respectively.The sensitivities and specificities of the single and combined detection of NLR, LY and CRP in the diagnosis of COVID-19 were 31.0%, 69.8%, 67.9%, 58.5% and 96.7%, 62.2%, 80.0%, 92.2%, respectively.The diagnosis value of the combined detection of NLR, LY and CRP were higher than that of the single detection of NLR, LY and CRP. ConclusionsThe combined detection of NLR, LY and CRP can increase the specificity and sensitivity of early diagnosis of COVID-19, which can guide the clinical treatment as soon as possible, and improve the cure rate of COVID-19. -
Key words:
- corona virus disease 2019 /
- lymphocyte /
- neutrophil to lymphocyte ratio /
- C reactive protein /
- ROC curve
-
表 1 2组间各指标检测结果比较(x±s)
分组 n NLR LY/(×109/L) CRP/(mg/L) 对照组 90 3.20±1.60 1.80±0.94 6.96±10.93 COVID-19组 53 6.40±7.16 1.21±0.56 57.93±66.47 t — 3.21 4.70 5.54 P — < 0.01 < 0.01 < 0.01 表 2 各指标对COVID-19诊断价值分析
指标 AUC cut-off值 灵敏度/% 特异度/% SE P 95%CI NLR 0.610 6.23 34.0 96.7 3.210 < 0.01 0.552~0.712 LY 0.705 1.41 69.8 62.2 4.700 < 0.01 0.625~0.777 CRP 0.833 11.80 67.9 80.0 5.540 < 0.01 0.420~0.590 NLR+LY+CRP 0.817 — 58.5 92.2 — — 0.644~0.790 -
[1] ZHU N, ZHANG DY, WANG WL, et al.A novel coronavirus from patients with pneumonia in China, 2019[J].N Engl J Med, 2020, 382:727. [2] 中华人民共和国国家卫生健康委员会.关于印发新型冠状病毒感染的肺炎诊疗方案(试行第七版)的通知[EB/OL].[2020-03-03]. [3] 石亚玲, 区静怡, 陈星, 等.多种炎症指标在新型冠状病毒肺炎的表达水平及临床应用价值[J].中华检验医学杂志, 2020(4):346. [4] GUAN WJ, NI ZY, HU Y, et al.Clinical characteristics of 2019 novel coronavirus infection in China[J].Med Rxiv, 2020.DOI:10.1101/2020.02.06.20020974. [5] 周玉平, 朱传新, 万禧伟, 等.咸宁地区3886例发热、咳嗽患者临床特征与实验室检查结果分析[J/OL].检验医学与临床: 1-91-9[2020-05-27].http://kns.cnki.net/kcms/detail/50.1167.R.20200304.1246.002.html. [6] CUI W, FAN Y, WU W, et al.Expression of lymphocytes and lymphocyte subsets in patients with severe acute respiratory syndrome[J].Clin Infect Dis, 2003, 37(6):857. [7] WONG RS, WU AH, TO K, et al.Haematological manifestations in patients with severe acute respiratory syndrome:retrospective analysis[J].BMJ, 2003, 326(7403):1358. [8] HUANG C, WANG Y, LI X, et al.Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China[J].Lancet, 2020, 395(10223):497. [9] SPROSTON NR, ASHWORTH JJ.Role of c-reactive protein at sites of inflammation and infection[J].Front Immunol, 2018, 9:754. [10] BORAS E, SLEVIN M, ALEXANDER MY, et al.Monomeric C-reactive protein and Notch-3 co-operatively increase angiogenesis through PI3K signalling pathway[J].Cytokine, 2014, 69(2):165. [11] LIU JY, LIU Y, XIANG P, et al.Neutrophil-to-Lymphocyte ratio predicts severe illness patients with 2019 novel coronavirus in the early stage[J].J Transl Med, 2020, 18(1):206. -





 下载:
下载: