-
鼻咽癌是我国东南地区发病率较高的恶性肿瘤之一,有研究[1-2]显示中国目前鼻咽癌的发病率和死亡率仍处于世界较高水平,由于鼻咽癌病人在接受放化疗治疗后,预后较差,易复发等原因,导致部分鼻咽癌病人的5年存活率不高[3-6]。因此,为了提高鼻咽癌治疗效果,寻找新的治疗策略以提高鼻咽癌病人的生存率值得研究。糖酵解抑制剂3-溴丙酮酸(3-bromopyruvate,3-BrPA)是一种小分子强烷化剂,研究[7-9]表明3-BrPA具有良好的体外抗肿瘤作用,能有效抑制肝癌、胰腺癌和胃癌的生长。本课题组前期研究[10-12]发现,3-BrPA能促进鼻咽癌、乳腺癌细胞的凋亡以及增强肝癌细胞对顺铂的敏感性。研究[13-14]显示,抑制肿瘤细胞糖酵解过程,使其能量大量缺失能够增强抗癌药物的疗效。本课题组前期在肝癌细胞中对3-BrPA的增敏作用进行了初步探讨,但3-BrPA能否在不同肿瘤细胞中发挥良好的化疗药物增敏作用仍需继续探索,在鼻咽癌细胞中,3-BrPA对顺铂诱导的鼻咽癌细胞凋亡的影响及其作用机制有待进一步探究。因此,本研究以人鼻咽癌细胞HNE1为研究对象,探讨3-BrPA对顺铂诱导的鼻咽癌细胞凋亡的敏感性及其作用机制,以期为鼻咽癌的临床治疗提供参考依据。
-
RPMI 1640培养基:HyClone公司;胰蛋白酶:Gibco公司;胎牛血清:杭州四季青生物工程材料有限公司;ATP检测试剂盒、线粒体膜电位检测试剂盒(JC-1)、青霉素-链霉素溶液:碧云天生物技术有限公司;二甲基亚砜(DMSO):Amresco公司;3-BrPA:Sigma公司;顺铂:齐鲁制药有限公司;AnnexinⅤ-FITC/PI双染试剂盒:上海贝博生物科技有限公司;Mcl-1、Bcl-2、Bax、Bak抗体:Proteintech公司;山羊抗兔IgG:Abcam公司。
-
鼻咽癌细胞株HNE1购自北京北纳创联生物技术研究院,由蚌埠医学院科研中心生化药理实验室冻存。将HNE1细胞置于含10%胎牛血清的RPMI 1640培养液内,培养于37 ℃、5% CO2、饱和湿度的培养箱中。
-
取对数生长期的HNE1细胞,以每孔6 000细胞接种于96孔板,培养24 h后换液加入不同浓度3-BrPA(20、40、80、160、320 μmol/L)、顺铂(2、4、8、16、32 μmol/L)以及80 μmol/L 3-BrPA联合顺铂(2、4、8、16、32 μmol/L)处理,同时设阴性对照组和空白对照组,每个处理3个复孔,分别培养24、48、72 h后,加入15 μL 20 μg/mL MTT溶液,继续培养4 h,吸去培养液,每孔加入150 μL DMSO,用酶标仪在490 nm处测定吸光度值(A490 nm)。计算细胞的存活率。细胞存活率(%)=(处理组A490 nm-空白组A490 nm)/(对照组A490 nm-空白组A490 nm)×100%。
-
取对数生长期的HNE1细胞,以每孔5 000细胞接种于6孔板,24 h后使用8 μmol/L3-BrPA,0.8 μmol/L顺铂以及8 μmol/L3-BrPA+0.8 μmol/L顺铂处理HNE1细胞,继续培养5~7 d,观察到细胞集落形成后,去除培养基,PBS洗涤2次,4%多聚甲醛-20 ℃固定20 min,结晶紫染色5 min,弃染色液,清洗干净,室温干燥并拍照。
-
实验分为对照组、80 μmol/L 3-BrPA组、8 μmol/L顺铂组、80 μmol/L 3-BrPA+8 μmol/L顺铂组。取对数生长期的HNE1细胞,以每孔1.5×105细胞接种于12孔板,经给药换液,继续培养24 h,收集细胞,1 000 r/min离心8 min,弃上清液,加入PBS洗1次,弃上清,按照试剂说明书将PI和AnnexinⅤ-FITC加入细胞中孵育20 min,1 h内用流式细胞仪检测细胞的凋亡情况。
-
取对数生长期的HNE1细胞,以每孔2×105细胞接种于6孔板,经给药换液(实验分组同1.5),继续培养24 h,吸除培养液,每孔加入0.5 mL新配制的JC-1染色工作液(按照JC-1检测试剂盒说明书进行操作),于37 ℃孵育30 min,再用预冷的JC-1染色缓冲液洗涤2次,冰上保存。荧光显微镜观察、拍照。
-
取对数生长期的HNE1细胞,以每孔2×105细胞接种于6孔板,经给药换液(实验分组同1.5),继续培养24 h,吸除培养液,PBS洗涤细胞2次,4%多聚甲醛室温固定20 min,每孔加0.5 mL DAPI染色液,避光孵育5 min,弃上清,PBS洗涤2次,加2 mL PBS。用荧光显微镜观察、拍照。
-
取对数生长期的HNE1细胞,以每孔2×105细胞接种于6孔板,每组3个复孔,经给药换液(实验分组同1.5),继续培养24 h后收集细胞。根据ATP检测试剂盒说明书进行操作,以酶标仪测定化学发光强度。用实验组的化学发光强度与对照组的化学发光强度的比值确定各组细胞内ATP水平。
-
取对数生长期的HNE1细胞,以每孔2.5×105细胞接种于6孔板,经给药换液(实验分组同1.5),继续培养24 h。收集细胞,冰上裂解30 min,4 ℃、12 000 r/min离心30 min,提取细胞总蛋白,BCA蛋白定量法测定各组蛋白浓度,加入SDS-PAGE蛋白上样缓冲液,100 ℃煮沸5 min。每组取50 μg蛋白,10% SDS-PAGE电泳(80 V,30 min;120 V,100 min);转膜(200 mA,2~3 h)至PVDF膜;5%脱脂牛奶室温封闭4 h(或4 ℃过夜);一抗室温孵育4 h(或4 ℃过夜);TPBS洗涤3次;二抗室温孵育2 h;TPBS洗涤3次;ECL发光试剂盒发光,凝胶成像系统获取图像。
-
采用方差分析和q检验。
-
3-BrPA(20、40、80、160、320 μmol/L)、顺铂(2、4、8、16、32 μmol/L)以及80 μmol/L 3-BrPA联合不同浓度顺铂(2、4、8、16、32 μmol/L)都能明显抑制HNE1细胞的体外增殖活性,与对照组比较,差异有统计学意义(P < 0.01)(见表 1、2)。
分组 存活率/% F P MS组内 24 h 48 h 72 h 顺铂/(μmol/L) 0 100.0±1.9 100.0±1.1 100.0±1.4 0.00 >0.05 2.239 2 93.9±1.2** 85.5±1.1** 73.6±1.1** 240.62 < 0.01 1.293 4 85.2±1.2** 65.7±1.4** 56.2±1.3** 383.13 < 0.01 1.718 8 74.4±1.3** 51.4±1.4** 37.2±1.4** 545.40 < 0.01 1.938 16 59.3±1.4** 39.5±1.8** 24.0±1.3** 394.72 < 0.01 2.376 32 39.5±1.4** 26.5±1.4** 11.0±1.2** 327.77 < 0.01 1.858 3-BrPA/(μmol/L) 0 100.0±2.0 100.0±1.1 100.0±1.4 0.00 >0.05 2.482 20 91.9±1.6** 85.2±2.0** 79.3±1.0** 46.02 < 0.01 2.578 40 85.7±1.3** 75.5±2.1** 68.1±1.4** 88.31 < 0.01 2.639 80 75.1±1.3** 59.0±1.2** 46.1±2.2** 245.14 < 0.01 2.571 160 51.3±2.5** 35.2±2.0** 20.5±2.1** 148.23 < 0.01 4.804 320 31.0±1.9** 20.1±1.2** 11.4±1.8** 100.36 < 0.01 2.867 q检验:与相应0 μmol/L组比较**P < 0.01 表 1 3-BrPA、顺铂对HNE1细胞增殖的影响(ni=3;x±s)
分组 存活率/% F P MS组内 对照组 100.0±1.6 80 μmol/L 3-BrPA+顺铂/(μmol/L) 2 59.0±2.2** 4 48.0±2.0** 8 37.1±2.3** 537.58 < 0.01 5.070 16 30.0±3.1** 32 12.3±1.9** q检验:与对照组比较**P < 0.01 表 2 3-BrPA联用顺铂对HNE1细胞增殖的影响(ni=3;x±s)
-
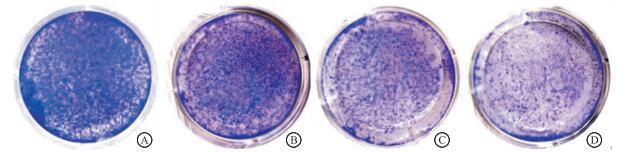
3-BrPA+顺铂组细胞集落数明显低于3-BrPA、顺铂单独处理组(P < 0.01);3-BrPA、顺铂单独处理组细胞集落数明显低于对照组(P < 0.01)(见图 1、表 3)。
分组 集落数 F P MS组内 对照组 438.0±5.3 3-BrPA 275.7±6.4** 3 236.16 < 0.01 22.167 顺铂 192.0±3.6** 3-BrPA+顺铂 69.7±2.5**##¤¤ q检验:与对照组比较**P < 0.01;与3-BrPA组比较##P < 0.01;与顺铂组比较¤¤P < 0.01 表 3 3-BrPA、顺铂及二者联用对HNE1细胞集落形成能力的影响(ni=3;x±s)
-
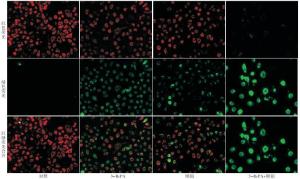
3-BrPA联用顺铂处理细胞24 h,与3-BrPA、顺铂单独处理组相比,红色荧光向绿色荧光转变明显(见图 2)。
-
3-BrPA联用顺铂处理细胞24 h,与3-BrPA、顺铂单独处理组相比,核碎裂及核固缩明显增多(见图 3)。
-
3-BrPA联用顺铂处理HNE1细胞24 h,细胞凋亡率明显高于3-BrPA、顺铂单独处理组(P < 0.01);3-BrPA、顺铂单独处理组细胞凋亡率明显高于对照组(P < 0.01)(见图 4、表 4)。
分组 细胞凋亡率/% F P MS组内 对照组 6.0±0.2 3-BrPA 15.6±0.3** 977.59 < 0.01 0.589 顺铂 14.6±0.6** 3-BrPA+顺铂 38.4±1.4**##¤¤ q检验:与对照组比较**P < 0.01;与3-BrPA组比较##P < 0.01;与顺铂组比较¤¤P < 0.01 表 4 3-BrPA、顺铂及二者联用对HNE1细胞凋亡的影响(ni=3;x±s)
-
3-BrPA联用顺铂处理细胞24 h,细胞内ATP水平明显低于3-BrPA、顺铂单独处理组,3-BrPA、顺铂单独处理组明显低于对照组(P < 0.01)(见表 5)。
分组 ATP水平/% F P MS组内 对照组 100.0±4.1 3-BrPA 71.0±2.7** 190.14 < 0.01 9.655 顺铂 86.8±2.1** 3-BrPA+顺铂 42.5±3.2**##¤¤ q检验:与对照组比较**P < 0.01;与3-BrPA组比较##P < 0.01;与顺铂组比较¤¤P < 0.01 表 5 3-BrPA、顺铂及二者联用对HNE1细胞ATP水平的影响(ni=3;x±s)
-
与对照组和3-BrPA、顺铂单独处理组相比,3-BrPA+顺铂组抗凋亡蛋白Mcl-1和Bcl-2蛋白表达下调,促凋亡蛋白Bax和Bak蛋白表达上调,差异有统计学意义(P < 0.05~P < 0.01)(见图 5、表 6)。
分组 相对表达量 Bcl-2 Mcl-1 Bax Bak 对照组 0.98±0.05 1.02±0.04 0.39±0.07 0.24±0.05 3-BrPA 0.73±0.01** 0.63±0.05** 0.75±0.04** 0.58±0.03* 顺铂 0.66±0.04** 0.70±0.06** 0.79±0.07** 0.74±0.04** 3-BrPA+顺铂 0.47±0.03**##¤¤ 0.21±0.02**##¤¤ 0.97±0.02**##¤ 1.01±0.02**##¤¤ F 99.51 166.75 61.28 206.08 P < 0.01 < 0.01 < 0.01 < 0.01 MS组内 0.001 0.002 0.003 0.002 q检验:与对照组比较*P < 0.05,**P < 0.01;与3-BrPA组比较##P < 0.01;与顺铂组比较¤P < 0.05,¤¤P < 0.01 表 6 3-BrPA、顺铂及二者联用对HNE1细胞凋亡相关蛋白表达的影响(ni=3;x±s)
-
肿瘤在形成和生长的过程中,其各种生物代谢过程与正常组织明显不同[15-17],能量代谢异常是肿瘤的典型特征之一[18-19]。3-BrPA是一种糖酵解抑制剂,能在体外抑制多种肿瘤细胞的生长[20-22],它不仅可以靶向糖酵解过程,还可以抑制肿瘤细胞中线粒体氧化磷酸化和ATP的产生[23-25]。代谢改变被广泛认为是癌症的标志之一,其中恶性肿瘤细胞表现出Warburg效应,这是加速ATP生成的主要糖酵解表型现象[26-27]。越来越多的证据表明,3-BrPA能通过靶向多种代谢分子,产生显著的肿瘤特异性细胞毒性,主要通过其抑制ATP的生成[28]。由于肿瘤细胞增殖需要消耗大量能量来完成蛋白质和RNA的合成、DNA复制和细胞质分裂等[29],因此,肿瘤细胞可能通过各种方式对ATP的消耗敏感。本研究结果显示,3-BrPA联用顺铂处理HNE1细胞后,其体外增殖明显受抑制,且细胞内ATP水平明显降低。
Bcl-2家族蛋白通过线粒体途径调节细胞凋亡[30]。Bcl-2蛋白作为细胞凋亡关键的抑制因子之一,使其成为一些药物的有效作用靶点。Mcl-1与Bcl-2在结构和功能上相似,能保护线粒体膜的完整性,在调节肿瘤细胞凋亡中起重要作用,Mcl-1已被证明是调节细胞凋亡的重要分子之一[31]。Bax和Bak蛋白主要通过调控各种凋亡诱导因子来发挥调节肿瘤细胞凋亡的作用[32]。本研究Western blotting结果表明,3-BrPA联用顺铂处理HNE1细胞,相对于3-BrPA、顺铂单独处理组,Bcl-2和Mcl-1蛋白表达下调,Bax和Bak蛋白表达上调,提示3-BrPA增强HNE1细胞对顺铂诱导凋亡的敏感性,其机制可能与调节凋亡相关蛋白的表达有关。
综上所述,3-BrPA能诱导HNE1细胞凋亡,并能增强HNE1细胞对顺铂的敏感性,其机制可能与降低细胞内ATP水平以及下调Mcl-1和Bcl-2表达,上调Bak和Bax蛋白的表达有关。
3-溴丙酮酸增强人鼻咽癌细胞对顺铂敏感性的作用及机制研究
Study on the effects of 3-bromopyruvate on the sensitivity of human nasopharyngeal carcinoma cells to cisplatin and its mechanism
-
摘要:
目的探讨3-溴丙酮酸(3-BrPA)增强人鼻咽癌细胞对顺铂敏感性的作用及作用机制。 方法MTT法检测3-BrPA和顺铂对鼻咽癌HNE1细胞增殖的影响。集落克隆形成实验观察3-BrPA和顺铂对HNE1细胞克隆形成能力的影响。线粒体膜电位检测试剂盒分析细胞早期凋亡情况。DAPI荧光染色法检测细胞核形态变化。AnnexinⅤ-FITC/PI双染法检测细胞凋亡率的变化。ATP检测试剂盒测定细胞内ATP水平的变化。Western blotting检测Bcl-2、Bax、Mcl-1、Bak蛋白的表达。 结果3-BrPA(20、40、80、160、320 μmol/L)、顺铂(2、4、8、16、32 μmol/L)以及80 μmol/L 3-BrPA联合不同浓度顺铂都能明显抑制HNE1细胞的体外增殖活性,与对照组比较,差异有统计学意义(P < 0.01)。8 μmol/L 3-BrPA+0.8 μmol/L顺铂组细胞集落数明显低于3-BrPA、顺铂单独处理组及对照组(P < 0.01)。80 μmol/L 3-BrPA+8 μmol/L顺铂组细胞红色荧光向绿色荧光转变明显;细胞核碎裂及核固缩明显增加;细胞凋亡率明显高于3-BrPA、顺铂单独处理组及对照组(P < 0.01);细胞内ATP水平明显低于3-BrPA、顺铂单独处理组及对照组(P < 0.01);Bcl-2、Mcl-1蛋白的表达降低,Bax、Bak蛋白的表达增高,与3-BrPA、顺铂单独处理组及对照组比较,差异有统计学意义(P < 0.05~P < 0.01)。 结论3-BrPA能诱导HNE1细胞凋亡,并能增强HNE1细胞对顺铂的敏感性,其机制可能与降低细胞内ATP水平以及下调Mcl-1和Bcl-2表达、上调Bak和Bax蛋白的表达有关。 Abstract:ObjectiveTo investigate the effect of 3-bromopyruvate (3-BrPA) on enhancing the sensitivity of human nasopharyngeal carcinoma cells to cisplatin and its mechanism. MethodsThe effects of 3-BrPA and cisplatin on the proliferation of nasopharyngeal carcinoma HNE1 cells were detected by MTT assay.The effects of 3-BrPA and cisplatin on the colony forming ability of HNE1 cells were observed using colony formation assay.The early apoptosis was analyzed by mitochondrial membrane potential detection kit.The nuclear morphological changes were detected by DAPI fluorescence staining.The apoptosis rate was detected by Annexin Ⅴ-FITC/PI double staining.The changes of intracellular ATP level were measured by ATP detection kit.The protein expressions of Bcl-2, Bax, Mcl-1 and Bak were analyzed by Western blotting. Results3-BrPA (20, 40, 80, 160 and 320 μmol/L), cisplatin (2, 4, 8, 16 and 32 μmol/L) and 80 μmol/L 3-BrPA combined with different concentrations of cisplatin significantly inhibited the proliferation of HNE1 cells in vitro, which was statistically significant compared with control group (P < 0.01).The combination of 8 μmol/L 3-BrPA and 0.8 μmol/L cisplatin significantly decreased the colony formation of HNE1 cells compared with that of 3-BrPA or cisplatin alone and control group (P < 0.01).The transformation of red fluorescence to green fluorescence was obvious, and nuclear fragmentation and nuclear pyknosis increased significantly in 80 μmol/L 3-BrPA combined with 8 μmol/L cisplatin group.The apoptosis rate and intracellular ATP level in 80 μmol/L 3-BrPA combined with 8 μmol/L cisplatin group were significantly higher and lower than those in 3-BrPA or cisplatin alone group and control group, respectively (P < 0.01).The expression levels of Bcl-2 and Mcl-1 protein were significantly downregulated, and the expression levels of Bax and Bak protein were significantly upregulated in 80 μmol/L 3-BrPA combined with 8 μmol/L cisplatin group, which was statistically significant compared with 3-BrPA or cisplatin alone group and control group (P < 0.05 to P < 0.01). Conclusions3-BrPA can induce apoptosis in HNE1 cells and enhance the sensitivity of HNE1 cells to cisplatin.The mechanism of which may be related to the decrease of intracellular ATP level, down-regulation of Mcl-1 and Bcl-2 protein, and up-regulation of Bak and Bax protein. -
Key words:
- nasopharyngeal neoplasms /
- 3-bromopyruvate /
- apoptosis
-
表 1 3-BrPA、顺铂对HNE1细胞增殖的影响(ni=3;x±s)
分组 存活率/% F P MS组内 24 h 48 h 72 h 顺铂/(μmol/L) 0 100.0±1.9 100.0±1.1 100.0±1.4 0.00 >0.05 2.239 2 93.9±1.2** 85.5±1.1** 73.6±1.1** 240.62 < 0.01 1.293 4 85.2±1.2** 65.7±1.4** 56.2±1.3** 383.13 < 0.01 1.718 8 74.4±1.3** 51.4±1.4** 37.2±1.4** 545.40 < 0.01 1.938 16 59.3±1.4** 39.5±1.8** 24.0±1.3** 394.72 < 0.01 2.376 32 39.5±1.4** 26.5±1.4** 11.0±1.2** 327.77 < 0.01 1.858 3-BrPA/(μmol/L) 0 100.0±2.0 100.0±1.1 100.0±1.4 0.00 >0.05 2.482 20 91.9±1.6** 85.2±2.0** 79.3±1.0** 46.02 < 0.01 2.578 40 85.7±1.3** 75.5±2.1** 68.1±1.4** 88.31 < 0.01 2.639 80 75.1±1.3** 59.0±1.2** 46.1±2.2** 245.14 < 0.01 2.571 160 51.3±2.5** 35.2±2.0** 20.5±2.1** 148.23 < 0.01 4.804 320 31.0±1.9** 20.1±1.2** 11.4±1.8** 100.36 < 0.01 2.867 q检验:与相应0 μmol/L组比较**P < 0.01 表 2 3-BrPA联用顺铂对HNE1细胞增殖的影响(ni=3;x±s)
分组 存活率/% F P MS组内 对照组 100.0±1.6 80 μmol/L 3-BrPA+顺铂/(μmol/L) 2 59.0±2.2** 4 48.0±2.0** 8 37.1±2.3** 537.58 < 0.01 5.070 16 30.0±3.1** 32 12.3±1.9** q检验:与对照组比较**P < 0.01 表 3 3-BrPA、顺铂及二者联用对HNE1细胞集落形成能力的影响(ni=3;x±s)
分组 集落数 F P MS组内 对照组 438.0±5.3 3-BrPA 275.7±6.4** 3 236.16 < 0.01 22.167 顺铂 192.0±3.6** 3-BrPA+顺铂 69.7±2.5**##¤¤ q检验:与对照组比较**P < 0.01;与3-BrPA组比较##P < 0.01;与顺铂组比较¤¤P < 0.01 表 4 3-BrPA、顺铂及二者联用对HNE1细胞凋亡的影响(ni=3;x±s)
分组 细胞凋亡率/% F P MS组内 对照组 6.0±0.2 3-BrPA 15.6±0.3** 977.59 < 0.01 0.589 顺铂 14.6±0.6** 3-BrPA+顺铂 38.4±1.4**##¤¤ q检验:与对照组比较**P < 0.01;与3-BrPA组比较##P < 0.01;与顺铂组比较¤¤P < 0.01 表 5 3-BrPA、顺铂及二者联用对HNE1细胞ATP水平的影响(ni=3;x±s)
分组 ATP水平/% F P MS组内 对照组 100.0±4.1 3-BrPA 71.0±2.7** 190.14 < 0.01 9.655 顺铂 86.8±2.1** 3-BrPA+顺铂 42.5±3.2**##¤¤ q检验:与对照组比较**P < 0.01;与3-BrPA组比较##P < 0.01;与顺铂组比较¤¤P < 0.01 表 6 3-BrPA、顺铂及二者联用对HNE1细胞凋亡相关蛋白表达的影响(ni=3;x±s)
分组 相对表达量 Bcl-2 Mcl-1 Bax Bak 对照组 0.98±0.05 1.02±0.04 0.39±0.07 0.24±0.05 3-BrPA 0.73±0.01** 0.63±0.05** 0.75±0.04** 0.58±0.03* 顺铂 0.66±0.04** 0.70±0.06** 0.79±0.07** 0.74±0.04** 3-BrPA+顺铂 0.47±0.03**##¤¤ 0.21±0.02**##¤¤ 0.97±0.02**##¤ 1.01±0.02**##¤¤ F 99.51 166.75 61.28 206.08 P < 0.01 < 0.01 < 0.01 < 0.01 MS组内 0.001 0.002 0.003 0.002 q检验:与对照组比较*P < 0.05,**P < 0.01;与3-BrPA组比较##P < 0.01;与顺铂组比较¤P < 0.05,¤¤P < 0.01 -
[1] TANG LL, CHEN WQ, XUE WQ, et al.Global trends in incidence and mortality of nasopharyngeal carcinoma[J].Cancer Letters, 2016, 374(1):22. doi: 10.1016/j.canlet.2016.01.040 [2] CHEN YP, CHAN ATC, LE QT, et al.Nasopharyngeal carcinoma[J].Lancet, 2019, 394(10192):64. doi: 10.1016/S0140-6736(19)30956-0 [3] LEE AW, MA BB, WT NG, et al.Management of nasopharyngeal carcinoma:current practice and future perspective[J].J Clin Oncol, 2015, 33(29):56. [4] LEEAW, TUNG SY, DT CHUA, et al.Randomized trial of radiotherapy plus concurrent-adjuvant chemotherapy vs radiotherapy alone for regionally advanced nasopharyngeal carcinoma[J].J Natl Cancer Inst, 2010, 102(15):88. [5] 梁雷锋, 林展.复发鼻咽癌治疗的新进展[J].中国临床新医学, 2019, 12(3):342. [6] NG WT, TUNG SY, LEE V, et al.Concurrent-adjuvant chemoradiation therapy for stage Ⅲ-IVB nasopharyngeal carcinoma-exploration for achieving optimal 10-year therapeutic ratio[J].Int J Radiat Oncol Biol Phys, 2018, 101(5):78. [7] 郭兴裕.糖酵解抑制剂对胃癌细胞MGC-803的影响及其机制研究[D].桂林: 广西医科大学, 2017. [8] 张海丽, 曾常茜, 郑学仿.3-溴丙酮酸对人肝癌HepG-2细胞增殖和凋亡的影响[J].广西医科大学学报, 2010, 27(2):183. [9] YANG Y, ZHU G, DONG B, et al.The NQO1/PKLR axis promotes lymph node metastasis and breast cancer progression by modulating glycolytic reprogramming[J].Cancer Letters, 2019, 453:170. doi: 10.1016/j.canlet.2019.03.054 [10] 邹雪.3-溴丙酮酸诱导鼻咽癌细胞死亡的作用及其机制[D].蚌埠: 蚌埠医学院, 2016. [11] 赵素容, 张媛媛, 吴成柱, 等.3-溴丙酮酸增强肝癌细胞对顺铂敏感性的作用[J].南方医科大学学报, 2014, 34(1):25. [12] 李其响.3-溴丙酮酸对乳腺癌细胞的细胞毒性依赖于MCT1表达[D].蚌埠: 蚌埠医学院, 2018. [13] SZCZUKA I, GAMIAN A, TERLECKI G.3-Bromopyruvate as a potential pharmaceutical in the light of experimental data[J].Postepy Hig Med Dosw, 2017, 71:88. [14] IHRLUND LS, HERNLUND E, KHAN O, et al.3-Bromopyruvate as inhibitor of tumour cell energy metabolism and chemopotentiator of platinum drugs[J].Mol Oncol, 2008, 2(1):94. [15] DEVORKIN L, PAVEY N, CARLETON G, et al.Autophagy regulation of metabolism is required for CD8(+) T cell anti-tumor immunity[J].Cell Rep, 2019, 27(2):502. [16] JAGUST P, LUXAN-DELGADO B DE, PAREJO-ALONSO B, et al.Metabolism-based therapeutic strategies targeting cancer stem cells[J].Front Pharmacol, 2019, 10:203. doi: 10.3389/fphar.2019.00203 [17] MARTINEZ-OUTSCHOOM UE, PESTELL RG, HOWELL A, et al.Energy transfer in "parasitic" cancer metabolism[J].Cell Cycle, 2011, 10(24):4208. doi: 10.4161/cc.10.24.18487 [18] WARD PS, THOMPSON CB.Metabolic reprogramming:acancer hallmark even warburg did not anticipate[J].Cancer Cell, 2012, 21(3):297. doi: 10.1016/j.ccr.2012.02.014 [19] LIU Y, FANG S, SUN Q, et al.Anthelmintic drug ivermectin inhibits angiogenesis, growth and survival of glioblastoma through inducing mitochondrial dysfunction and oxidative stress[J].Biochem Biophys Res Commun, 2016, 480(3):415. [20] ISHIGURO Y, KOBAYASHI M, IDENO M, et al.Valproate sensitizes human glioblastoma cells to 3-bromopyruvate-induced cytotoxicity[J].Int J Pharm, 2018, 551(1-2):97. doi: 10.1016/j.ijpharm.2018.08.039 [21] CHEN Y, WEI L, ZHANG X, et al.3-Bromopyruvate sensitizes human breast cancer cells to TRAIL-induced apoptosis via the phosphorylated AMPK-mediated upregulation of DR5[J].Oncol Rep, 2018, 40(5):2435. [22] ZOU X, ZHANG M, SUN Y, et al.Inhibitory effects of 3-bromopyruvate in human nasopharyngeal carcinoma cells[J].Oncol Rep, 2015, 34(4):1895. [23] PULASKI L, JATCZAK-PAWLIK I, SOBALSKA-KWAPIS M, et al.3-Bromopyruvate induces expression of antioxidant genes[J].Free Radic Res, 2019, 53(2):170. doi: 10.1080/10715762.2018.1541176 [24] LIS P, DYLAG M, NIEDZWIECKA K, et al.The HK2 dependent "warburg effect" and mitochondrial oxidative phosphorylation in cancer:targets for effective therapy with 3-bromopyruvate[J].Molecules, 2016, 21(12):1730. doi: 10.3390/molecules21121730 [25] TRAN Q, LEE H, PARK J, et al.Targeting cancer metabolism-revisiting the warburg effects[J].Toxicol Res, 2016, 32(3):177. doi: 10.5487/TR.2016.32.3.177 [26] YADAV S, PANDEY SK, KUMAR A, et al.Antitumor and chemosensitizing action of 3-bromopyruvate:implication of deregulated metabolism[J].Chem Biol Interact, 2017, 270:73. doi: 10.1016/j.cbi.2017.04.015 [27] LEBELO MT, JOUBERT AM, VISAGIE MH.Warburg effect and its role in tumourigenesis[J].Arch Pharm Res, 2019, 42(10):833. doi: 10.1007/s12272-019-01185-2 [28] FAN T, SUN G, SUN X, et al.Tumor energy metabolism and potential of 3-bromopyruvate as an inhibitor of aerobic glycolysis:implications in tumor treatment[J].Cancers (Basel), 2019, 11(3):317. doi: 10.3390/cancers11030317 [29] STAGG J, SMYTH MJ.Extracellular adenosine triphosphate and adenosine in cancer[J].Oncogene, 2010, 29(39):5346. doi: 10.1038/onc.2010.292 [30] SCHENK RL, STRASSER A, DEWSON G.BCL-2:long and winding path from discovery to therapeutic target[J].Biochem Biophys Res Commun, 2017, 482(3):459. [31] WENG JR, BAI LY, CHIU SJ, et al.Divaricoside exerts antitumor effects, in part, by modulating Mcl-1 in human oral squamous cell carcinoma cells[J].Comput Struct Biotechnol J, 2019, 17:151. doi: 10.1016/j.csbj.2019.01.004 [32] PENA-BLANCO A, GARCIA-SAEZ AJ.Bax, Bak and beyond-mitochondrial performance in apoptosis[J].The FEBS Journal, 2018, 285(3):416. doi: 10.1111/febs.14186 -





 下载:
下载: