-
近年来,我国的癌症发病率和死亡率不断在增加,癌症已成为我国居民的主要死亡原因,肺癌的发病率和死亡率居各类恶性肿瘤之首,其恶性程度高、进展快[1]。目前临床上针对肺癌的治疗手段主要以手术、化疗及放疗为主,但大部分肺癌病人确诊时已是晚期,失去了手术的最佳时机,而传统化疗方案疗效有限,亦存在耐药现象,放疗不良反应较大。因此需探索新的肺癌治疗方式。近年来随着中药提纯技术的提高,应用中药及其有效成分进行综合治疗已成为肿瘤领域的研究热点。华蟾素为干蟾皮经科学方法提取加工制成的水化萃取物,具有清热解毒、利水消肿等作用,对多种肿瘤细胞具有抑制作用,其抗肿瘤的机制尚未完全明确。本研究以肺癌细胞株A549为研究对象,探讨华蟾素抗肺癌的分子机制。现作报道。
-
华蟾素注射液(安徽金蟾生化股份有限公司,5毫升/支,批号041003)。兔抗磷酸化蛋白激酶B(p-AKT)(#4060)、兔抗磷酸化磷脂酰肌醇-3激酶(p-PI3K)(#4228)、磷脂酰肌醇-3激酶(PI3K)(#4292)、兔抗蛋白激酶B(AKT)(#4691)、兔抗磷酸化哺乳动物雷帕霉素靶蛋白(p-mTOR)(#2974)、兔抗哺乳动物雷帕霉素靶蛋白(mTOR)(#2972)、兔抗p-S6(#4858)、兔抗S6(#2217)、兔抗磷酸化PTEN(p-PTEN)(#9551)、兔抗PTEN(#9188)、兔抗肌动蛋白(β-actin)(#8457)抗体均购自Cell Signaling Technology公司;聚偏二氟乙烯(PVDF)膜(EMD Millipore公司,#C2035)。人非小细胞肺癌细胞株A549细胞株(中科院上海生命科学研究院细胞资源中心)。DMEM细胞培养基、胎牛血清、0.25%胰蛋白酶、100 μg/mL青霉素+链霉素均购自美国Invitrogen公司。EdU(Sigma公司),细胞计数试剂盒(CCK-8)试剂盒(碧云天公司)。单人超净台(北京六一仪器厂);HD-3000凝胶成像仪(上海上天精密仪器有限公司);CB15 CO2细胞培养箱(北京六一仪器厂)。
-
人非小细胞肺癌细胞株A549常规复苏,用含10%胎牛血清的DMEM培养液、青霉素(100 μg/mL)及链霉素(100 μg/mL)的培养液进行培养,置于37 ℃、5% CO2恒温培养箱中。取对数生长期的细胞用0.25%胰蛋白酶消化,用细胞计数仪行细胞计数。
-
取对数生长期细胞, 用0.25%胰蛋白酶消化离心后,0.5×104个/孔的密度种植于96孔板中。培养24 h待细胞贴壁后弃去培养液,处理组加入0.2、0.8 μg/mL华蟾素注射液;空白对照组加入等量DMEM培养液。使用CCK-8检测不同浓度华蟾素、不同处理时间(6、12、24、48、72 h)的A549细胞增殖活力。于时间点每孔加入10 μL CCK8试剂并置于37 ℃孵育4 h,用酶标仪测定450 nm波长处各孔的吸光度值。计算均数及标准差,运用GraphPad软件绘制时间依赖生长曲线。
-
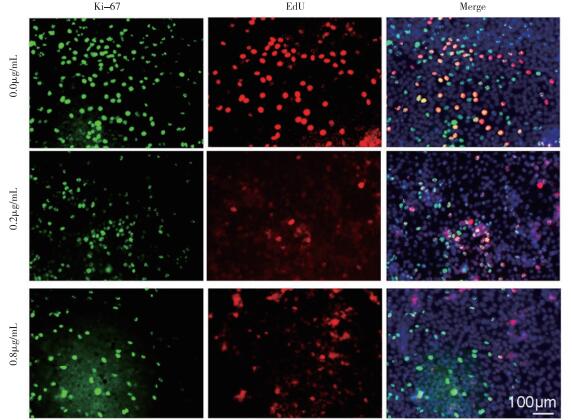
将对数生长期的细胞分为对照组、阴性对照组、实验组,调整细密度为1×105/mL,接种于有无菌盖玻片的24孔板中,37 ℃,5% CO2条件下,培养72 h,根据EdU染色试剂盒说明书,检测细胞增殖。完全培养基稀释EdU溶液为10 mmol/mL,每孔加入100 μL,孵育4 h后弃培养基,PBS清洗2次,多聚甲醛固定24 h,TBST+驴血清破膜30 min,一抗Ki67过夜孵育,PBST清洗3次,每次10 min,二抗荧光显色剂Alexa-488+Apollo+DAPI染色试剂孵育1 h,PBST清洗3次,每次10 min,封片,荧光显微镜下观察。
-
胰酶消化并收集华蟾素处理(对照组为等量DMEM培养液)后72 h的A549细胞,弃培养液,用预冷的PBS清洗2次,加入预冷的含有蛋白酶抑制剂(含1% PMSF、1%氟化钠、1%钒酸钠)的细胞裂解液RIPA,冰浴5~10 min后刮取细胞至离心管,冰上超声裂解2 min,12 000 ×g离心后取上清细胞裂解液裂解细胞,分光光度计测定蛋白浓度,取总蛋白样品进行10% SDS-PAGE电泳并湿转印至PVDF膜上。含5%脱脂奶粉溶液封闭1 h,4 ℃一抗(抗体浓度:p-PI3K、PI3K、p-AKT、AKT、p-mTOR、mTOR、p-S6、S6、p-PTEN、PTEN、β-actin均为1:1 000)孵育过夜。次日TBST洗膜3次(每次8 min)后,用山羊抗兔二抗(1:5 000)孵育1 h,TBST洗膜3次(每次8 min),ECL化学发光显色,Tanon 4500自动化学发光图像分析系统扫描并采用Quantity One软件进行图像灰度分析,计算灰度比值,运用GraphPad软件进行结果绘图。
-
采用方差分析和Dunnett-t检验。
-
CCK8法检测结果示,与对照组比较,0.2 μg/mL华蟾素作用A549细胞株48 h后显示明显抑制作用(P < 0.01),0.8 μg/mL华蟾素处理12 h后均具有不同程度抑制作用(P < 0.05~P < 0.01)(见表 1)。进一步应用EdU及Ki67免疫荧光法检测检测华蟾素对A549细胞株增殖的影响(见图 1),结果显示,与对照组比较,0.2、0.8 μg/mL华蟾素作用72 h后,A549细胞增殖均受到抑制(P < 0.05~P < 0.01)(见表 2)。
华蟾素浓度/(μg/mL) n 6 h 12 h 24 h 48 h 72 h 0.0 3 0.325±0.020 0.515±0.054 0.612±0.081 0.871±0.099 1.045±0.126 0.2 3 0.287±0.049 0.403±0.051 0.528±0.068 0.626±0.078** 0.791±0.086** 0.8 3 0.305±0.041 0.367±0.051* 0.393±0.055** 0.486±0.062** 0.623±0.066** F — 0.74 6.57 7.71 17.25 14.28 P — >0.05 < 0.05 < 0.05 < 0.01 < 0.01 MS组内 — 0.001 0.018 0.037 0.114 0.133 与对照组比较*P < 0.05,**P < 0.01 表 1 CCK8法检测对照组和华蟾素处理组不同时间点细胞增殖情况(x±s)
华蟾素浓度/(μg/mL) n Ki67 EdU Ki67/EdU 0.0 3 39.333±5.163 28.364±5.164 14.182±3.954 0.2 3 28.125±3.842* 14.287±3.606** 7.379±2.518* 0.8 3 11.369±2.054** 8.333±2.517** 2.337±1.082** F — 39.06 20.70 13.74 P — < 0.01 < 0.01 < 0.01 MS组内 — 594.235 317.412 106.000 与对照组比较*P < 0.05,**P < 0.01 表 2 Ki67、EdU检测对照组和华蟾素处理组细胞增殖情况(x±s)
-
PTEN/AKT/mTOR通路活性0.2、0.8 μg/mL华蟾素作用于细胞72 h后,p-PI3K、p-AKT、p-mTOR、p-S6、p-PTEN表达量均较对照组下降(P < 0.05~P < 0.01),而PI3K、AKT、mTOR、S6及PTEN表达量与对照组差异无统计学意义(P < 0.05)(见图 2、表 3)。
华蟾素浓度/(μg/mL) p-PI3K PI3K p-AKT AKT p-mTOR mTOR p-S6 S6 p-PTEN PTEN 0.0 0.261±0.056 0.816±0.082 0.624±0.075 0.959±0.181 0.478±0.056 0.480±0.057 0.754±0.086 0.685±0.077 0.841±0.081 0.576±0.082 0.2 0.137±0.024* 0.804±0.079 0.375±0.059** 0.926 ±0.175 0.286±0.032** 0.522±0.058 0.481±0.059** 0.596±0.084 0.537±0.052* 0.560±0.079 0.8 0.102±0.016** 0.789±0.086 0.224±0.050** 0.915±0.189 0.217±0.036** 0.477±0.064 0.328±0.052** 0.612±0.079 0.414±0.049** 0.619±0.086 F 15.83 0.081 31.57 0.135 27.65 0.415 30.86 0.792 37.27 0.412 P < 0.01 >0.05 < 0.01 >0.05 < 0.01 >0.05 < 0.01 >0.05 < 0.01 >0.05 MS组内 0.021 0.000 0.122 0.004 0.055 0.002 0.140 0.120 0.145 0.003 与对照组比较*P < 0.05,**P < 0.01 表 3 对照组和华蟾素处理组AKT/mTOR信号通路蛋白表达量比较(ni=3;x±s)
-
华蟾素是中华大蟾蜍的干燥表皮的水化萃取物,其主要成分是吲哚生物碱类、嘧啶类以及生物碱等,具有清热解毒、消炎、强心、利水消肿、抗病毒、止痛等作用[2]。华蟾素还具有抗癌功效, 可明显抑制肺癌、膀胱癌、肝癌、乳腺癌、卵巢癌等多种癌细胞的生长[3-4]。本研究结果表明华蟾素可以以时间及剂量依赖方式抑制肺癌A549细胞增殖,与既往研究[5-6]结果一致。
在肿瘤组织中,肿瘤信号转导蛋白磷酸化水平往往表达异常,这与癌细胞增殖、迁移和侵袭性明显相关[7]。PI3K/AKT/mTOR信号通路相关蛋白磷酸化表达增强可影响肿瘤细胞的增殖能力,抑制凋亡发生[8]。PTEN是PI3K/AKT传导通路的负性调控因子,其可去磷酸化PIP3,产生PIP2,从而终止PI3K介导的信号转导[9]。在本研究中,华蟾素可以明显的降低A549细胞中p-PI3K、p-AKT、p-mTOR、p-S6的表达量,而对PI3K、AKT、mTOR、S6的表达量没有明显影响。这说明华蟾素可能是通过抑制肿瘤信号转导蛋白磷酸化而抑制A549增殖。既往研究[10]发现在癌组织中,PTEN主要通过基因突变、杂合性丢失、甲基化等方式失活而造成表达量异常。此外,研究[11-12]表明PTEN的翻译后修饰还包括磷酸化、氧化、和蛋白结合等,其中以磷酸化最为重要。在本研究中,华蟾素明显降低p-PTEN的表达量,这进一步说明华蟾素可能是通过抑制PI3K/AKT/mTOR信号通路上游信号蛋白PTEN的磷酸化,进而进一步抑制PI3K/AKT/mTOR信号通路蛋白磷酸化而发挥抑制肿瘤的作用。
综上,华蟾素可能通过抑制PI3K/AKT/mTOR信号通路蛋白磷酸化,抑制肺癌细胞A549的增殖。
华蟾素对非小细胞肺癌细胞株A549细胞增殖及PTEN/AKT/mTOR信号通路表达的影响
Effect of cinobufacin on the proliferation and expression of PTEN/AKT/mTOR signaling pathway in non-small cell lung cancer cell line A549
-
摘要:
目的观察华蟾素对肺癌细胞A549及PTEN/AKT/mTOR信号通路蛋白表达的影响。 方法在培养肺癌细胞株NCI-A549中分别加入不同浓度的华蟾素,应用细胞计数试剂盒-8检测24、48、72 h后细胞增殖活力,Ki67及EdU检测72 h细胞增殖活力,免疫印迹法检测磷脂酰肌醇-3激酶/蛋白激酶B/哺乳动物雷帕霉素靶蛋白以及磷酸化第10号染色体缺失的磷酸酶和张力蛋白的同源基因(phosphorylated-phosphatase and tensin homolog deleted on chromosome ten,p-PTEN)蛋白表达。 结果0.2 μg/mL华蟾素作用于A549细胞株48 h后显示明显抑制增殖作用(P < 0.01),0.8 μg/mL华蟾素处理12 h后均具有不同程度抑制增殖作用(P < 0.05~P < 0.01)。不同浓度华蟾素作用于细胞72 h后,p-PI3K、p-AKT、p-mTOR、p-S6、p-PTEN表达量均较对照组下降(P < 0.05~P < 0.01),而PI3K、AKT、mTOR、S6及PTEN表达量与对照组差异无统计学意义(P>0.05)。 结论华蟾素可能通过抑制PI3K/AKT/mTOR信号通路蛋白磷酸化,抑制肺癌细胞A549的增殖。 -
关键词:
- 肺肿瘤 /
- 华蟾素 /
- 肺癌细胞株A549 /
- PTEN/AKT/mTOR信号通路 /
- 增殖
Abstract:ObjectiveTo observe the expression changes of PTEN/AKT/mTOR signaling pathway protein after the lung cancer cells A549 treated with cinobufacin. MethodsDifferent concentrations of cinobufacin were added to the cultured lung cancer cell line NCI-A549, and the cell proliferation activity was measured using cell counting kit-8 after 24, 48 and 72 h.Ki67 and EdU were used to detect the proliferation of cells after 72 h of treatment with cinobufacin.Western blotting was used to detect the expression levels of protein kinase B/mammalian rapamycin target protein pathway protein(AKT/mTOR) and phosphorylated-phosphatase and tensin homolog deleted on chromosome ten(P-PTEN). ResultsThe proliferation of A549 cell line inhibited after treatment with 0.2 μg/mL cinobufacin for 48 h(P < 0.01), and the proliferation of A549 cell line was inhibited after treatment with 0.8 μg/mL cinobufacin for 12 h(P < 0.05 to P < 0.01).Compared with the control group, the expression levels of p-PI3K, p-AKT, p-mTOR, p-S6 and p-PTEN in experimential group after 72 h of treatment with different concentrations of cinobufacin decreased(P < 0.05 to P < 0.01), while the differences of the expression levels of PI3K, AKT, mTOR, S6 and PTEN were not statistically significant between the control group and erperimantial group(P>0.05). ConclusionsCinobufacin may inhibit the proliferation of lung cancer cells A549 by inhibiting the phosphorylation of PI3K/AKT/mTOR signaling pathway protein. -
表 1 CCK8法检测对照组和华蟾素处理组不同时间点细胞增殖情况(x±s)
华蟾素浓度/(μg/mL) n 6 h 12 h 24 h 48 h 72 h 0.0 3 0.325±0.020 0.515±0.054 0.612±0.081 0.871±0.099 1.045±0.126 0.2 3 0.287±0.049 0.403±0.051 0.528±0.068 0.626±0.078** 0.791±0.086** 0.8 3 0.305±0.041 0.367±0.051* 0.393±0.055** 0.486±0.062** 0.623±0.066** F — 0.74 6.57 7.71 17.25 14.28 P — >0.05 < 0.05 < 0.05 < 0.01 < 0.01 MS组内 — 0.001 0.018 0.037 0.114 0.133 与对照组比较*P < 0.05,**P < 0.01 表 2 Ki67、EdU检测对照组和华蟾素处理组细胞增殖情况(x±s)
华蟾素浓度/(μg/mL) n Ki67 EdU Ki67/EdU 0.0 3 39.333±5.163 28.364±5.164 14.182±3.954 0.2 3 28.125±3.842* 14.287±3.606** 7.379±2.518* 0.8 3 11.369±2.054** 8.333±2.517** 2.337±1.082** F — 39.06 20.70 13.74 P — < 0.01 < 0.01 < 0.01 MS组内 — 594.235 317.412 106.000 与对照组比较*P < 0.05,**P < 0.01 表 3 对照组和华蟾素处理组AKT/mTOR信号通路蛋白表达量比较(ni=3;x±s)
华蟾素浓度/(μg/mL) p-PI3K PI3K p-AKT AKT p-mTOR mTOR p-S6 S6 p-PTEN PTEN 0.0 0.261±0.056 0.816±0.082 0.624±0.075 0.959±0.181 0.478±0.056 0.480±0.057 0.754±0.086 0.685±0.077 0.841±0.081 0.576±0.082 0.2 0.137±0.024* 0.804±0.079 0.375±0.059** 0.926 ±0.175 0.286±0.032** 0.522±0.058 0.481±0.059** 0.596±0.084 0.537±0.052* 0.560±0.079 0.8 0.102±0.016** 0.789±0.086 0.224±0.050** 0.915±0.189 0.217±0.036** 0.477±0.064 0.328±0.052** 0.612±0.079 0.414±0.049** 0.619±0.086 F 15.83 0.081 31.57 0.135 27.65 0.415 30.86 0.792 37.27 0.412 P < 0.01 >0.05 < 0.01 >0.05 < 0.01 >0.05 < 0.01 >0.05 < 0.01 >0.05 MS组内 0.021 0.000 0.122 0.004 0.055 0.002 0.140 0.120 0.145 0.003 与对照组比较*P < 0.05,**P < 0.01 -
[1] CHEN W, ZHENG R, BAADE PD, et al.Cancer statistics in China, 2015[J].CA Cancer J Clin, 2016, 66(2):115. doi: 10.3322/caac.21338 [2] SHENG X, SUN X, SUN K, et al.Inhibitory effect of bufalin combined with Hedgehog signaling pathway inhibitors on proliferation and invasion and metastasis of liver cancer cells[J].Int J Oncol, 2016, 49(4):1513. doi: 10.3892/ijo.2016.3667 [3] YANG T, SHI R, CHANG L, et al.Huachansu suppresses human bladder cancer cell growth through the Fas/Fasl and TNF-α/TNFR1 pathway in vitro and in vivo[J].J Exp Clin Cancer Res, 2015, 34(1):21. doi: 10.1186/s13046-015-0134-9 [4] 于秀荣, 陈钧泽, 李慧杰, 等.华蟾素抗肿瘤研究[J].中医学报, 2019, 34(6):1174. [5] 熊飞, 施嫣嫣, 沈久成, 等.华蟾素对肺癌NCI-A549移植瘤裸鼠的抑制作用及机制研究[J].中国临床药理学与治疗学, 2018, 23(10):1103. [6] WANG JY, CHEN L, ZHENG Z, et al.Cinobufocini inhibits NF-kappaB and COX-2 activation induced by TNF-α in lung adenocarcinoma cells[J].Oncol Rep, 2012, 27(5):1619. [7] FUMAROLA C, BONELLI MA, PETRONINI PG, et al.Targeting PI3K/AKT/mTOR pathway in non small cell lung cancer[J].Biochem Pharmacol, 2014, 90(3):197. doi: 10.1016/j.bcp.2014.05.011 [8] 葛信艳, 黄玮, 李瑞琴, 等.PI3K/AKt信号转导通路与肿瘤发生相互作用的机制研究[J].中国现代医药杂志, 2017, 19(7):98. [9] CHANG H, CAI Z, ROBERTS TM.The mechanisms underlying PTEN loss in human tumors suggest potential therapeutic opportunities[J].Biomolecules, 2019, 9(11):713. doi: 10.3390/biom9110713 [10] YEHIA L, KEEL E, ENG C.The clinical spectrum of PTEN mutations[J].Ann Rev Med, 2019, 71:103. [11] HO J, BASSI C, STAMBOLIC V.Characterization of nuclear PTEN and its post translational modifications[J].Methods, 2015, 77/78:104. doi: 10.1016/j.ymeth.2015.01.006 [12] ENGELMAN JA, LUO J, CANTLEY LC.The evolution of phosphatidylinositol 3-kinases as regulators of growth and metabolism[J].Nat Rev Genet, 2006, 7(8):606. doi: 10.1038/nrg1879 -




 下载:
下载: