-
结直肠腺瘤是结直肠癌的癌前疾病[1]。结直肠癌主要来自腺瘤和无蒂锯齿状腺瘤的癌变[2]。早期切除是预防结直肠癌的重要手段。切除的标本送病理检查能帮助内镜医生明确后续治疗方案,提出随访建议。目前在肠镜检查中发现息肉将尽量切除并送病理检查,依据肠镜下即时判断息肉组织学类型的光学诊断系统,如NICE分型,国外有较多研究[3-4]探索息肉处理的替代策略,即“切除-丢弃”策略(切除小腺瘤而不送病理检查)和“诊断-离开”策略(直乙结肠的增生性微小息肉(≤0.5 cm)可不处理)。NICE分型适用于放大及非放大NBI内镜,国内外有较多相关研究[5-9]证明其在预判非肿瘤性、肿瘤性结直肠息肉的诊断价值高。
与传统注气肠镜相比,有文献[10-11]表明注水肠镜或注水肠镜联合NBI能提高腺瘤检出率。这可能与注水条件下消除黏膜反光、结直肠息肉病变在水的浮力下更为凸显有关。故注水肠镜可能对非放大窄带成像(NBI)下NICE分型具有一定的积极影响。本研究回顾性分析经我中心日间病房收治并切除的结直肠小息肉病变,比较注水与注气结肠镜下非放大NBI下NICE分型的诊断价值,由此探索更合理高效的日间病房结直肠小息肉治疗策略。
-
回顾性分析2020年1-12月在我中心行肠镜检查发现并于日间病房住院,行内镜下切除并经病理检查证实的449枚结直肠小息肉(包括24枚一并完整切除的结直肠微小息肉)。纳入标准:(1)图像清晰可满足NICE分型需求;(2)直径≤1.0 cm;(3)由同一位资深内镜医师检查并行内镜下治疗的病人。排除标准:(1)肠道准备差(渥太华评分>7分);(2)肠道炎症,如结肠黑变病等;(3)家族性腺瘤性结肠息肉病。
共纳入320例病人,449枚息肉病变,其中盲肠18枚,升结肠73枚,肝曲20枚,横结肠70枚,脾曲9枚,降结肠48枚,乙状结肠179枚,直肠32枚;隆起型息肉(0~Ⅰs)345枚,带蒂息肉(0~Ⅰp)37枚,扁平息肉(0~Ⅱ)67枚;微小息肉(≤5 mm)24枚,小息肉(6~10 mm)296枚。其中注水法肠镜下发现176枚(116例病人)作为注水组,传统注气法肠镜下发现273枚(204例病人)作为注气组。
-
2组病人按照常规行肠道准备:(1)行3 L复方聚乙二醇(PEG)电解质散的分次剂量方案,即于前一天晚8:00-9:00口服1 L,检查当日的检查前4~6 h口服2 L,直至排出清水样便;(2)检查前24 h进食低渣/低纤维饮食,检查当日禁食;(3)肠道准备情况按照渥太华评分量表[12]进行评估,≤7分提示肠道准备合格。
-
病人采取常规左侧卧位,于肛周涂抹肠镜胶,接受肛指检查后,由同一位资深内镜医师使用奥林巴斯CF-H290I型肠镜进行肠镜检查。注气组常规注气,并以轴保持短缩法进镜至盲肠,退镜时充分注气扩张肠腔,发现结直肠息肉病变后分别于白光和非放大NBI下留取图片,仔细观察后尽量吸气以减轻肠腔张力。注水组在通过肛门后以水泵注水代替注气扩张肠腔,并将残余空气吸尽,同样以轴保持短缩法进镜,退镜时大量注水代替注气扩张肠腔,发现结直肠息肉病变后分别于白光和非放大NBI下留取图片,充分观察后尽量吸尽液体。2组病人行小息肉治疗时,由同一位资深内镜医师按照常规肠镜下肠黏膜切除术(EMR)流程进行:(1)黏膜下注射;(2)息肉勒除器完整电切除病灶;(3)金属夹夹闭创面。对于入组病人肠道中的微小息肉,均予息肉勒除器冷切除。
-
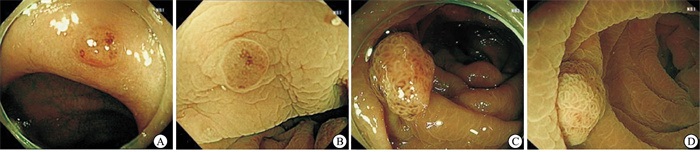
由经验丰富的内镜医生和初学者分别对2组NICE分型进行诊断(见图 1)。以病理诊断为金标准对息肉进行分类。非肿瘤性息肉:炎性息肉、增生性息肉;肿瘤性息肉:管状腺瘤、管状绒毛状腺瘤、锯齿状腺瘤等。
-
采用χ2检验和Kappa一致性检验。
-
注水组结直肠息肉病变176枚,其中扁平息肉35枚,病理证实为肿瘤性息肉132枚(检出率75.00%);注气组273枚中扁平息肉32枚,病理证实为肿瘤性息肉219枚(检出率80.22%)。注水组扁平息肉检出率高于注气组(P < 0.05),2组腺瘤检出率和无蒂锯齿状腺瘤检出率差异均无统计学意义(P>0.05)(见图 2、表 1)。
分组 n 形态 病理类型 腺瘤检出 无蒂锯齿状腺瘤检出 0~Ⅰ 0~Ⅱ 非肿瘤性 肿瘤性 注水组 176 141(80.1) 35(19.9) 44(25.0) 132(75.0) 132(75.0) 4(2.3) 注气组 273 241(88.3) 32(11.7) 54(19.8) 219(80.2) 219(80.2) 7(2.6) χ2 — 5.62 1.71 1.71 0.04 P — < 0.05 >0.05 >0.05 >0.05 表 1 2组结直肠息肉检出情况[n;百分率(%)]
-
由2名经验丰富的内镜医生分别对449枚结直肠息肉病变图片进行独立诊断,一致性检验结果提示Kappa值=0.807(注水组=0.812,注气组=0.773),一致性较好(见图 3、4,表 2)。最终的NICE分型结果由2名经验丰富的内镜医生共同讨论得出,病理检查证实,449枚结直肠息肉病变中非肿瘤性息肉98枚,肿瘤性息肉(包括4枚高级别上皮内瘤变)351枚,2组NICE分型诊断的敏感性、特异性、准确率见表 3。
分组 n 非肿瘤性 肿瘤性 Kappa 注水组 1 40 36 4 0.812 2 136 8 128 注气组 1 42 39 3 0.773 2 231 15 216 表 2 2组结直肠息肉NICE分型诊断的一致性检验(n)
分组 敏感性 特异性 准确率 阳性
预测值阴性
预测值注水组 97.0 81.8 94.3 94.1 90.0 注气组 98.6 72.0 93.4 93.5 92.9 表 3 2组结直肠息肉NICE分型诊断的敏感性、特异性和准确率(%)
-
本研究也邀请了一名初学者,进行短期NICE分型培训后,以非放大NBI下NICE分型对同样的449枚结直肠息肉病变的图片进行诊断。结果显示,注水组、注气组在初学者以非放大NBI下NICE分型预判结直肠小息肉病理类型的准确率分别为56.8%、49.5%,2组差异无统计学意义(χ2=0.13,P>0.05)。以Kappa检验分别比较经验丰富的内镜医生及初学者对于注水组、注气组以非放大NBI下NICE分型预判病理类型的准确性,Kappa值分别为0.199、0.076,一致性均较低(见表 4),以病理结果为标准,其诊断敏感性、特异性和准确率亦均不佳(见表 5)。
分组 n 非肿瘤性 肿瘤性 注水组 1 96 35 64 2 80 12 68 注气组 1 152 34 118 2 121 20 101 表 4 初学者对2组NICE分型诊断(n)
分组 敏感性 特异性 准确率 阳性预测值 阴性预测值 注水组 51.5 72.7 56.8 85.0 33.3 注气组 46.1 63.0 49.5 83.0 22.4 表 5 初学者对2组结直肠息肉NICE分型诊断与病理诊断对照(%)
-
关于结直肠息肉病变的病理预判,针对结直肠息肉表面腺管结构或微血管形态有数种分型方式,NICE分型和JNET分型是综合考虑结直肠息肉病变上述两方面特点并预判其病理类型的分型方式。NICE分型对于非肿瘤性病变(NICE1型)及需外科手术的黏膜下深层以深病变(NICE3型)具有较高的准确性[14-15],且由于可适用于非放大的NBI技术,方法简便、迅速,操作要求不高,适合设备简单的基层医院及内镜总量较大的大型医院。但NICE2型并不能很好区分腺瘤性息肉、低级别上皮内瘤变、高级别上皮内瘤变、局限于黏膜下浅层以浅的早期癌。与之相比,JNET分型[16]更进一步细化了NICE2型的分类方式,预判更为精准,但需要在放大内镜下完成,放大内镜价格昂贵且操作耗时、对初学者不友好,不能很好普及。对于适合收治日间病房的结直肠小息肉病变,NICE分型的简单快速更为合适。
本研究中,两名经验丰富的内镜医生依据非放大NBI下NICE分型对于结直肠小息肉独立诊断能达成较好一致性,且准确性、敏感度、特异度较高,这与多个中心的研究[17-20]结果相一致。本研究中,注水组、注气组肿瘤性息肉检出率差异无统计学意义,提示经验丰富的内镜医生以非放大NBI下NICE分型预判结直肠小息肉病理类型的诊断能力强,诊断价值高,无论注水、注气条件,考虑到注水肠镜操作较注气肠镜复杂耗时,日常的常规检查中,对于经验丰富的内镜医生,传统的注气肠镜已基本够用。同时,本研究结果显示,注水组能检出更多扁平息肉(0~Ⅱ),这与陈建华等[10]对于注水+非放大NBI下NICE分型的研究结果大致相同。其原因可能与以下几方面有关:(1)水有自身重力,注水肠镜整体进镜过程舒缓,避免过度成角,视觉盲区较少,在肠道准备合格的病人中,注水肠镜能观察到更多肠腔黏膜,发现处于死角的息肉;(2)水有浮力,肠腔黏膜及息肉会漂浮在水中,使藏在褶皱中的小息肉更明显、更易被观察到(见图 3),尤其是隆起不明显的扁平息肉;(3)注水肠镜操作较为费时,延长了退镜时间,且注水有助于清洁肠道,帮助发现易漏诊的息肉,尤其是颜色与周围黏膜相差不大的扁平息肉。在国内外研究[21-23]显示,注水法肠镜较之注气法,能明显提高腺瘤检出率,也与以上几方面有关。但本研究中2组腺瘤检出率差异无统计学意义,可能原因为本研究纳入标准限定为结直肠小息肉(直径≤10 mm),注水肠镜检出的更多结直肠扁平小息肉是非肿瘤性息肉。
有研究[17, 24-25]显示,经过系统的短期培训,NICE分型对于初学者与经验丰富者一样友好,准确性较高。但在本研究中,初学者经简单培训后,其非放大NBI下NICE分型预判结直肠小息肉病理类型的敏感性、特异性、准确率均较低,且反复有涂改,诊断时间是经验丰富的内镜医生的2倍以上(涂改及诊断时间延长代表低信心度),这种现象尤其出现在注气组。初学者在注气组、注水组间的准确性差异无统计学意义,与经验丰富内镜医生的诊断一致性均低。可见虽然水下提供了更多有助于进行NICE分型的异常性信息,但对于低信心度的初学者,无论是注水还是注气内镜条件下,诊断均较为困难。注水一定程度上有利于初学者提高信心度,但无益于诊断。对于初学者而言,推荐以发现-切除-送检策略处理发现的息肉,或者进行更多NBI下NICE分型的诊断培训。
本研究449枚息肉中,共发现无蒂锯齿状腺瘤11枚。锯齿状腺瘤可通过锯齿状途径癌变,部分具有不稳定及快速进展的高恶变潜能[26-27],建议在内镜下切除。有研究[28-30]显示,NICE分型无法区分肿瘤性的锯齿状腺瘤和非肿瘤性的增生性息肉,因为锯齿状腺瘤和增生性息肉同属于锯齿状病变,在内镜下表现为相似的外观。锯齿状腺瘤的内镜下独立诊断因素集中在边界模糊、积云样堆积、黏液帽、直径>5 mm、近端结肠(右半结肠),放大内镜下特点为隐窝黑点、扩张的微血管[28, 31-34]。目前对于锯齿状腺瘤,仅靠非放大NBI+NICE分型无法与增生性息肉做到准确区分。有研究[10]表明,注水肠镜能更加容易区分锯齿状腺瘤的以上特点,但在本研究中,注水组、注气组锯齿状腺瘤检出率差异无统计学意义。锯齿状腺瘤内镜下诊断确实存在一定的困难,需要与病理诊断相互印证。
本研究存在不足:(1)纳入的结直肠小息肉多为良性腺瘤性息肉,仅有4枚病理提示高级别上皮内瘤变,没有更加进展期的癌变息肉,在样本选择上存在一定偏倚,需要更大样本量验证研究结果;(2)纳入病人的微小息肉大多按照本中心日间病房的息肉切除策略予氩气刀灼除,送检的微小息肉样本量不足,在样本选择上同样存在一定的实验偏倚,对于微小息肉的处理策略仍需进一步研究明确;(3)参与研究的内镜医生都属于本中心,不同中心的内镜医生得出结果可能会有不同;(4)本研究中息肉图片尚存在其他辅助判断分型的征象,如白斑征,可能帮助内镜医生预判病理结果。
自日间病房开设以来,本中心一直在探索更加合适高效的息肉处理策略。目前的临床实践中,对于结直肠小息肉,尽量满足其入院治疗需求,以EMR完整切除后送病理检查明确诊断,指导术后随访或进一步治疗策略。对于微小息肉合并6~10 mm小息肉,倾向于一并入院治疗,小息肉完整切除后送病理检查,微小息肉大多予氩气刀灼除;仅发现微小息肉时,倾向于活检摘除。根据本研究成果,经验丰富的内镜医生在内镜下能较好区分肿瘤性和非肿瘤性结直肠小息肉, 结合临床实践,对于经验丰富的内镜医生所发现的结直肠小息肉病变,其处理策略可稍作改变:对于直径较大、内镜下预判为肿瘤性息肉或内镜下预判息肉倾向于锯齿状腺瘤诊断的病人建议优先入院,减少其院外等待时间,早期切除以预防病变快速进展;内镜下预判为非肿瘤性息肉的病人,尤其是非肿瘤性微小息肉的病人,可门诊随访,以减少手术的并发症及治疗、病理检查费用;对于预判为肿瘤性息肉的,尽量完整切除后送病理检查(对于小息肉,EMR足够满足整块切除需求);对于非肿瘤性息肉,较大者送检,较小者不追求病理检查报告,灵活选择治疗方案,可选择创面更小、术后并发症更少、花费也更少的治疗方式。
综上,经验丰富的内镜医师以非放大NBI下NICE分型能够较好区分肿瘤性、非肿瘤性结直肠小息肉,无论注水还是注气条件,诊断价值均较高,对于日间病房的息肉治疗策略有很好的指导意义。
注水与注气肠镜+非放大NBI下NICE分型对于结直肠小息肉内镜下治疗的临床价值
Clinical value of NICE typing under water injection and gas injection colonoscopy plus non-magnifying narrow-band imaging for endoscopic treatment of small colorectal polyps
-
摘要:
目的比较注水与注气结肠镜下非放大窄带成像(NBI)下NICE分型对于结直肠小息肉的诊断价值。 方法回顾性选取449枚结直肠小息肉,其中176枚为注水法肠镜下发现(注水组),273枚为注气法肠镜下发现(注气组)。观察2组病人留取的非放大NBI图像,应用NICE分型预判病理类型,并与病理检查结果进行分析对比。 结果2组腺瘤检出率差异无统计学意义(P>0.05),注水组扁平息肉检出率高于注气组(P < 0.05)。经验丰富的内镜医生对NICE分型诊断结果一致性较高(Kappa值=0.807),对注水组和注气组非放大NBI下NICE分型诊断的敏感性、特异性、准确率分别为97.0%(128/132)、81.8%(36/44)、94.3%(166/176)和98.6%(216/219)、72.0%(39/54)、93.4%(255/273)。初学者对注水组和注气组非放大NBI下NICE分型诊断的敏感性、特异性、准确率分别为51.5%(68/132)、72.7%(32/44)、56.8%(100/176)和46.1%(101/219)、63.0%(34/54)、49.5%(135/273)。初学者和经验丰富的内镜医生对2组NICE分型诊断结果一致性均较低(Kappa值=0.199、0.076)。 结论无论注水还是注气条件,经验丰富的内镜医生以非放大NBI下NICE分型能够很好地区分肿瘤性、非肿瘤性结直肠小息肉,诊断价值高,对于日间病房的息肉治疗策略有很好的指导意义。 Abstract:ObjectiveTo compare the diagnostic value of NICE typing under water injection and gas injection colonoscopy plus non-magnifying narrow-band imaging (NBI) in small colorectal polyps. MethodsA total of 449 small colorectal polyps were enrolled in this study.Among them, 176 cases were found by water injection colonoscopy(water injection group), and 273 cases were found by gas injection colonoscopy(gas injection group).Each lesion was observed by non-magnifying NBI, and the histology was predicted and compared according to NICE typing system. ResultsThere was no statistically significant difference in the examination results in the colorectal adenoma detection rate between the two groups(P>0.05), and the detection rate of flat polyps in the water injection group was higher than that in the gas injection group(P < 0.05).Experienced endoscopists had a high consistency in the diagnosis results of NICE typing(κ=0.807).The sensitivity, specificity and accuracy of NICE typing under non-magnifying NBI in the water injection group and the gas injection group were 97.0%(128/132), 81.8%(36/44), 94.3%(166/176) and 98.6%(216/219), 72.0%(39/54) and 93.4%(255/273), respectively.For novice endoscopists, the sensitivity, specificity and accuracy of NICE typing diagnosis under non-magnifying NBI in water injection group and gas injection group were 51.5%(68/132), 72.7% (32/44), 56.8% (100/176), 46.1%(101/219), 63.0%(34/54), 49.5%(135/273), respectively.The agreement of highly experienced endoscopists and novice endoscopists was low in two groups respectively(κ=0.199, 0.076). ConclusionsBetween whatever water or gas injection colonoscopy, NICE typing under non-magnifying NBI is effective in distinguishing neoplastic from non-neoplastic small colorectal polyps for highly experienced endoscopists, which has high diagnostic value and is instructive for treatment strategy of small colorectal polyps in ambulatory ward. -
表 1 2组结直肠息肉检出情况[n;百分率(%)]
分组 n 形态 病理类型 腺瘤检出 无蒂锯齿状腺瘤检出 0~Ⅰ 0~Ⅱ 非肿瘤性 肿瘤性 注水组 176 141(80.1) 35(19.9) 44(25.0) 132(75.0) 132(75.0) 4(2.3) 注气组 273 241(88.3) 32(11.7) 54(19.8) 219(80.2) 219(80.2) 7(2.6) χ2 — 5.62 1.71 1.71 0.04 P — < 0.05 >0.05 >0.05 >0.05 表 2 2组结直肠息肉NICE分型诊断的一致性检验(n)
分组 n 非肿瘤性 肿瘤性 Kappa 注水组 1 40 36 4 0.812 2 136 8 128 注气组 1 42 39 3 0.773 2 231 15 216 表 3 2组结直肠息肉NICE分型诊断的敏感性、特异性和准确率(%)
分组 敏感性 特异性 准确率 阳性
预测值阴性
预测值注水组 97.0 81.8 94.3 94.1 90.0 注气组 98.6 72.0 93.4 93.5 92.9 表 4 初学者对2组NICE分型诊断(n)
分组 n 非肿瘤性 肿瘤性 注水组 1 96 35 64 2 80 12 68 注气组 1 152 34 118 2 121 20 101 表 5 初学者对2组结直肠息肉NICE分型诊断与病理诊断对照(%)
分组 敏感性 特异性 准确率 阳性预测值 阴性预测值 注水组 51.5 72.7 56.8 85.0 33.3 注气组 46.1 63.0 49.5 83.0 22.4 -
[1] GLOVER B, PATEL N, ASHRAFIAN H, et al. Diagnostic accuracy of i-scan image enhancement for real-time endoscopic diagnosis of small colorectal polyps: a meta-analysis[J]. Therap Adv Gastroenterol, 2018, 11: 1756284818814948. [2] NGUYEN LH, GOEL A, CHUNG DC. Pathways of colorectal carcinogenesis[J]. Gastroenterology, 2020, 158(2): 291. doi: 10.1053/j.gastro.2019.08.059 [3] KANDEL P, WALLACE MB. Should we resect and discard low risk diminutive colon polyps?[J]. Clin Endosc, 2019, 52(3): 239. doi: 10.5946/ce.2018.136 [4] FERLITSCH M, MOSS A, HASSAN C, et al. Colorectal polypectomy and endoscopic mucosal resection (EMR): European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline[J]. Endoscopy, 2017, 49(3): 270. doi: 10.1055/s-0043-102569 [5] HAMADA Y, TANAKA K, KATSURAHARA M, et al. Utility of the narrow-band imaging international colorectal endoscopic classification for optical diagnosis of colorectal polyp histology in clinical practice: a retrospective study[J]. BMC Gastroenterol, 2021, 21(1): 336. doi: 10.1186/s12876-021-01898-z [6] PATRUN J, OKREŠA L, IVEKOVIĆH, et al. Diagnostic accuracy of NICE classification system for optical recognition of predictive morphology of colorectal polyps[J]. Gastroenterol Res Pract, 2018, 2018: 7531368. [7] 沙杰, 朱炳良, 徐丽芳, 等. 窄带成像放大肠镜下NICE分型对结直肠小息肉的诊断价值[J]. 中国内镜杂志, 2015, 21(11): 1134. [8] 朱宇均. 非放大内镜窄带成像技术基础上的NICE分型法在结直肠息肉性质判定中的临床诊断价值[J]. 中外医学研究, 2020, 18(25): 86. doi: 10.14033/j.cnki.cfmr.2020.25.034 [9] 卓宪华, 陈俊榕, 卢祎, 等. 非放大内镜下结直肠病变的国际NICE分型判断结直肠息肉的应用及学习曲线[J]. 中华胃肠内镜电子杂志, 2021, 8(3): 105. doi: 10.3877/cma.j.issn.2095-7157.2021.03.004 [10] 陈建华, 施宏, 陈素玉, 等. 注气与注水肠镜下结直肠息肉的内镜形态与病理关系的比较[J]. 中国肿瘤临床, 2020, 47(5): 236. doi: 10.3969/j.issn.1000-8179.2020.05.013 [11] 孟令君, 杨幼林. 注水式结肠镜检查的优势[J]. 胃肠病学和肝病学杂志, 2017, 26(10): 1183. [12] ROSTOM A, JOLICOEUR E. Validation of a new scale for the assessment of bowel preparation quality[J]. Gastrointest Endosc, 2004, 59(4): 482. doi: 10.1016/S0016-5107(03)02875-X [13] TANAKA S, SANO Y. Aim to unify the narrow band imaging (NBI) magnifying classification for colorectal tumors: current status in Japan from a summary of the consensus symposium in the 79th Annual Meeting of the Japan Gastroenterological Endoscopy Society[J]. Dig Endosc, 2011, 23(Suppl 1): 131. [14] HAYASHI N, TANAKA S, HEWETT DG, et al. Endoscopic prediction of deep submucosal invasive carcinoma: validation of the narrow-band imaging international colorectal endoscopic (NICE) classification[J]. Gastrointest Endosc, 2013, 78(4): 625. doi: 10.1016/j.gie.2013.04.185 [15] PUIG I, LÓPEZ-CERÓN M, ARNAU A, et al. Accuracy of the narrow-band imaging international colorectal endoscopic classification system in identification of deep invasion in colorectal polyps[J]. Gastroenterology, 2019, 156(1): 75. doi: 10.1053/j.gastro.2018.10.004 [16] SANO Y, TANAKA S, KUDO SE, et al. Narrow-band imaging (NBI) magnifying endoscopic classification of colorectal tumors proposed by the Japan NBI Expert Team[J]. Dig Endosc, 2016, 28(5): 526. doi: 10.1111/den.12644 [17] WANG Y, LI WK, WANG YD, et al. Diagnostic performance of narrow-band imaging international colorectal endoscopic and Japanese narrow-band imaging expert team classification systems for colorectal cancer and precancerous lesions[J]. World J Gastrointest Oncol, 2021, 13(1): 58. doi: 10.4251/wjgo.v13.i1.58 [18] VIŞOVAN II, TAN ÅU M, PASCU O, et al. The role of narrow band imaging in colorectal polyp detection[J]. Bosn J Basic Med Sci, 2017, 17(2): 152. [19] 吴友伟, 张健, 赵素平, 等. 结肠镜窄带成像技术对直径<2 cm结肠息肉样病变的诊断价值及误诊分析[J]. 诊断学理论与实践, 2020, 19(6): 588. [20] 沈煜枫, 戴军, 李晓波, 等. 非放大内镜下窄带成像技术结合NICE分型在诊断结直肠病变中的应用[J]. 上海交通大学学报(医学版), 2019, 39(2): 176. [21] FUCCIO L, FRAZZONI L, HASSAN C, et al. Water exchange colonoscopy increases adenoma detection rate: a systematic review with network meta-analysis of randomized controlled studies[J]. Gastrointest Endosc, 2018, 88(4): 589. doi: 10.1016/j.gie.2018.06.028 [22] HSIEH YH, LEUNG FW. An overview of deep learning algorithms and water exchange in colonoscopy in improving adenoma detection[J]. Expert Rev Gastroenterol Hepatol, 2019, 13(12): 1153. doi: 10.1080/17474124.2019.1694903 [23] LIU Y, HUANG QK, DONG XL, et al. Water exchange versus air insufflation for colonoscopy: a meta-analysis[J]. Saudi J Gastroenterol, 2018, 24(6): 311. doi: 10.4103/sjg.SJG_118_18 [24] ZHANG QW, ZHANG JJ, YANG AM, et al. Feasibility of using narrow band imaging international colorectal endoscopic classification for diagnosing colorectal neoplasia in China: a multicenter pilot observational study[J]. J Dig Dis, 2020, 21(2): 88. doi: 10.1111/1751-2980.12841 [25] 董金斌, 石益海. 非放大内镜应用窄带成像国际结直肠内镜分型判定结直肠息肉样病变性质的临床价值[J]. 浙江医学, 2021, 43(10): 1101. doi: 10.12056/j.issn.1006-2785.2021.43.10.2021-22 [26] EAST JE, ATKIN WS, BATEMAN AC, et al. British Society of Gastroenterology position statement on serrated polyps in the colon and rectum[J]. Gut, 2017, 66(7): 1181. doi: 10.1136/gutjnl-2017-314005 [27] KINOSHITA S, NISHIZAWA T, URAOKA T. Progression to invasive cancer from sessile serrated adenoma/polyp[J]. Dig Endosc, 2018, 30(2): 266. doi: 10.1111/den.12988 [28] URAOKA T, HIGASHI R, HORII J, et al. Prospective evaluation of endoscopic criteria characteristic of sessile serrated adenomas/polyps[J]. J Gastroenterol, 2015, 50(5): 555. doi: 10.1007/s00535-014-0999-y [29] DJINBACHIAN R, DUBÉ AJ, VON RENTELN D. Optical diagnosis of colorectal polyps: recent developments[J]. Curr Treat Options Gastroenterol, 2019, 17(1): 99. doi: 10.1007/s11938-019-00220-x [30] MURAKAMI T, SAKAMOTO N, NAGAHARA A. Endoscopic diagnosis of sesslie serrated adenoma/polyp with and without dysplasia/carcinoma[J]. World J Gastroenterol, 2018, 24(29): 3250. doi: 10.3748/wjg.v24.i29.3250 [31] HAZEWINKEL Y, LÓPEZ-CERÓN M, EAST JE, et al. Endoscopic features of sessile serrated adenomas: validation by international experts using high-resolution white-light endoscopy and narrow-band imaging[J]. Gastrointest Endosc, 2013, 77(6): 916. doi: 10.1016/j.gie.2012.12.018 [32] HASEGAWA S, MITSUYAMA K, KAWANO H, et al. Endoscopic discrimination of sessile serrated adenomas from other serrated lesions[J]. Oncol Lett, 2011, 2(5): 785. [33] MURAKAMI T, SAKAMOTO N, NAGAHARA A. Endoscopic diagnosis of sessile serrated adenoma/polyp with and without dysplasia/carcinoma[J]. World J Gastroenterol, 2018, 24(29): 3250. doi: 10.3748/wjg.v24.i29.3250 [34] NISHIZAWA T, YOSHIDA S, TOYOSHIMA A, et al. Endoscopic diagnosis for colorectal sessile serrated lesions[J]. World J Gastroenterol, 2021, 27(13): 13219. -





 下载:
下载: