-
自体动静脉内瘘(autogenous arteriovenous fistula,AVF)为维持性血液透析病人的首选通路[1]。随着生活方式和饮食结构的改变,高血压、糖尿病、动脉粥样硬化等慢性疾病逐渐增多,以及人口老龄化,病人自身血管条件差,无法满足建立AVF的需要,人工血管动静脉内瘘(arteriovenous graft,AVG)即成为首选通路[2]。各种原因引起的术后狭窄和闭塞,是AVG远期通畅率不高的主要原因[3],解除内瘘狭窄,是保持血管通路畅通的关键。关于动静脉内瘘通畅率的影响因素,国内外已有相关报道[4-5]。本研究主要探讨以AVG为透析通路的病人内瘘出现狭窄,行超声引导下经皮腔内血管成形术(percutaneous transluminal angioplasty,PTA)治疗的效果,进一步探讨影响术后通畅率的危险因素,现作报道。
-
收集2018年4月至2019年10月在我院行超声引导下PTA治疗AVG狭窄的病人共36例,其中男20例,女16例,年龄33~72岁,平均(52.1±10.8)岁; 原发病为慢性肾小球肾炎18例、糖尿病9例、高血压3例、多囊肾1例、不明病因5例。36例病人中肱动脉-贵要静脉AVG 29例,肱动脉-头静脉AVG 7例,病人已使用此血管通路进行规律血液透析,平均使用时间为(15.5±8.5)个月,每周透析2~3次。纳入标准: (1)彩超或血管造影明确内瘘管腔狭窄超过50%;(2)透析时静脉压高,透析血流量 < 200 mL/min,无法完成透析; (3)内瘘处触诊震颤减弱或消失,听诊血管杂音消失。排除标准: (1)中心静脉狭窄或堵塞; (2)存在严重凝血功能障碍; (3)内瘘出现感染。
-
病人术前已完善彩色多普勒超声明确狭窄部位,病人取仰卧位,患肢外展,常规消毒、铺巾、1%利多卡因局部浸润麻醉,在超声引导下于内瘘狭窄处近心端穿刺进入人工血管,进入导丝,拔出穿刺针,进入5F穿刺鞘,退出鞘芯及导丝,鞘内注射肝素15 mg,在超声引导下,沿穿刺鞘进入0.035超滑导丝,沿导丝进入6 mm×40 mm波科Mustang球囊,球囊通过狭窄部位逐渐增大压力至10个大气压,球囊逐渐膨起,1 min后逐渐减压,2 min后重复上述操作,退出球囊,反复扩张几次,直至超声显示狭窄段消失,血流良好,病人内瘘处可触及明显震颤,拔除穿刺鞘,5-0血管缝合线荷包缝合,予敷料包扎,手术结束。
-
收集病人PTA手术前后人工血管内瘘狭窄处内径、透析血流量、肱动脉血流量,评价手术效果以及有无出现并发症。PTA术后3、6、9、12个月随访病人,在彩超下检查内瘘有无狭窄或血栓形成,了解病人内瘘通畅情况。随访结果采用初级通畅率和次级通畅率表示,初级通畅率定义: PTA术后未经干预和治疗,AVG仍然保持通畅的病人人数占所有病人的比例;次级通畅率定义: 各种原因导致的内瘘无法完成血液透析,经治疗后AVG保持通畅的病人人数占所有病人的比例。
-
采用配对t检验、χ2检验和Fisher确切概率法,并绘制Kaplan-Meier生存曲线。
-
36例病人手术均成功,技术成功率为100%,未出现出血、感染、血管破裂、假性动脉瘤形成等并发症;结果显示,术后病人狭窄处内径较术前增大,透析血流量增加,肱动脉血流量增加,差异均有统计学意义(P < 0.01),治疗有效(见表 1)。
时间 n 狭窄处内径/mm 透析血流量/
(mL/min)肱动脉血流量/
(mL/min)术前 36 1.58±0.35 144.13±19.12 352.30±42.01 术后 36 3.71±0.62 275.75±14.29 616.83±45.92 t — 16.61 35.76 28.26 P — < 0.01 < 0.01 < 0.01 表 1 PTA手术前后测量指标比较(x±s)
-
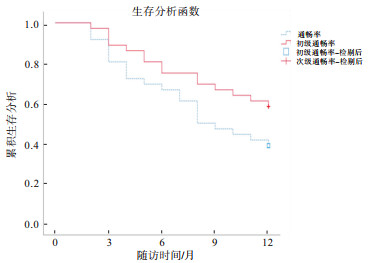
术后随访病人3、6、9、12个月,初级和次级通畅率分别为80.6%、66.7%、47.2%、38.9%和88.9%、75.0%、69.4%、58.3%,各随访时间初级通畅率和次级通畅率差异均无统计学意义(P>0.05)(见表 2、图 1)。
通畅率 术后3个月 术后6个月 术后9个月 术后12个月 初级通畅率 29(80.6) 24(66.7) 17(47.2) 14(38.9) 次级通畅率 32(88.9) 27(75.0) 25(69.4) 21(58.3) χ2 0.97 0.61 3.66 2.72 P >0.05 >0.05 >0.05 >0.05 表 2 病人PTA术后通畅率情况[n; 百分率(%)]
-
单因素分析发现,病人术后1年通畅组的年龄低于非通畅组,差异有统计学意义(P < 0.01),慢性肾小球肾炎、高血压、糖尿病等原发病以及性别、透析时间、静脉吻合口等方面2组差异无统计学意义(P>0.05)(见表 3)。
影响因素 通畅组
(n=14)非通畅组
(n=22)t P 年龄(x±s)/岁 42.71±6.90 58.09±8.34 5.75 < 0.01 性别 男 8(57.14) 12(54.55) — >0.05* 女 6(42.86) 10(45.45) 慢性肾小球肾炎 是 6(42.86) 12(54.55) — >0.05* 否 8(57.14) 10(45.45) 高血压 是 2(14.29) 1(4.55) — >0.05* 否 12(85.71) 21(95.45) 糖尿病 是 3(21.43) 6(27.27) — >0.05* 否 11(78.57) 16(72.73) 静脉吻合口 贵要静脉 10(71.43) 19(86.36) — >0.05* 头静脉 4(28.57) 3(13.64) 透析时间(x±s)/月 14.00±8.87 16.91±7.74 1.04 >0.05 *示Fisher′s确切概率法P值 表 3 病人术后1年初级通畅率的影响因素分析[n;百分率(%)]
-
终末期肾脏病的治疗方式为肾脏替代治疗,包括腹膜透析、血液透析和肾移植。美国一项对维持性血液透析病人统计数据进行分析的研究[6]发现,AVF为主要血管通路类型,其次是带隧道和涤纶套的透析导管(tunnel-cuffed catheter,TCC),AVG所占比例最低。AVF具有通畅率高、感染率低、死亡率低等优点[7],为首选血管通路。我国专家组对血管通路比例预期[1]: AVF>80%,TCC < 10%,当无法建立AVF时,建议提高AVG使用率。美国的透析结果和实践模式研究[8]显示,不同国家使用AVG的差异很大,使用率在2%~18%之间。影响内瘘功能的因素是内瘘狭窄和形成血栓,而狭窄是导致血栓形成的主要原因[9],早在1984年,GLANZ等[10]首次报道了PTA治疗血液透析通路的病变。国内也有研究[11]表明PTA是治疗AVG狭窄和闭塞的有效方法。随着腔内技术的成熟和治疗方式的创新,介入治疗不只局限于本研究采用的普通压力球囊扩张,对于普通压力的球囊无法完全扩张开的狭窄,有报道[12]高压球囊扩张可以取得较好的疗效,除此之外,外周切割球囊[13]对于治疗AVF和AVG狭窄也是安全、有效的。而当PTA术中发生血管破裂时,支架置入是一种紧急补救措施[14]。
本研究中PTA术后3、6、12个月初级通畅率分别是80.6%、66.7%和38.9%,次级通畅率分别是88.9%、75.0%和58.3%,OH等[15]研究显示PTA治疗AVG狭窄,术后3、6、12个月初级通畅率分别是84.8%、63.6%和24.2%,次级通畅率分别是95.2%、95.2%和85.7%,本研究初级通畅率结果与之相似,次级通畅率并不完全一致,可能与纳入病人本身之间存在差异等因素有关,但通畅率随着时间延长,均呈逐渐下降的趋势。有研究[16]发现影响AVG初级通畅率的因素有年龄、血管吻合位置,次级通畅率与舒张压、血清白蛋白水平、血红蛋白>10 g/dL呈正相关,这与本研究中的年龄为影响通畅率危险因素结论相符。我院收治的维持性血液透析病人大多伴有肾性贫血,根据上述研究结论,通过提高血红蛋白水平,可能有助于增加次级通畅率。曾敏等[17]提出PTA术能够降低内瘘再狭窄的风险,提高次级通畅率。本研究选择在彩超引导下行PTA,是因为彩超设备便携,对于病人来说,经济负担相对较小,与血管造影相比,没有造影剂及辐射危害,同时能够测量血管内径和血流动力学参数,评估病人内瘘情况,而且对于内瘘并发症具有早期发现、早期诊断的作用[18],及时有效地处理并发症,延长内瘘使用寿命。本研究尚存在许多不足之处,在回顾性分析超声引导下PTA治疗内瘘狭窄的有效性的同时,未设立对照组,比较不同治疗方式的效果。下一步工作可以延长随访时间,纳入更多病例,进行早期监测,及时干预。
综上所述,本研究选取的36例人工血管动静脉内瘘狭窄病人,PTA治疗有效,且创伤小,可节约病人珍贵的血管资源,可进一步推广应用,从而改善维持性血液透析病人血管通路的畅通情况。
超声引导下经皮腔内血管成形术治疗人工血管动静脉内瘘狭窄:单中心36例病人通畅率分析
Ultrasound-guided percutaneous endovascular angioplasty in the treatment of arteriovenous graft stenosis: analysis of patency rate in 36 patients at a single center
-
摘要:
目的探讨超声引导下经皮腔内血管成形术(percutaneous transluminal angioplasty, PTA)治疗人工血管动静脉内瘘(arteriovenous graft, AVG)狭窄的有效性和可行性, 分析PTA术后通畅率的影响因素。 方法回顾性分析36例维持性血液透析病人AVG发生狭窄采用PTA治疗前后内瘘狭窄处内径、透析血流量, 肱动脉血流量变化, 评估治疗效果。术后每3个月定期复查通畅情况。 结果36例病人手术均成功, 术前36例病人狭窄处内径(1.58±0.35) mm, 透析血流量(144.13±19.12) mL/min, 肱动脉血流量(352.30±42.01) mL/min; PTA术后狭窄处内径(3.71±0.62) mm, 透析血流量(275.75±14.29) mL/min, 肱动脉血流量(616.83±45.92) mL/min, 差异均有统计学意义(P < 0.01), 治疗有效。术后随访3、6、9、12个月, 初级和次级通畅率分别为80.6%、66.7%、47.2%、38.9%和88.9%、75.0%、69.4%、58.3%。各随访时间初级通畅率和次级通畅率差异均无统计学意义(P>0.05)。病人术后1年通畅组的年龄低于非通畅组, 差异有统计学意义(P < 0.01)。 结论超声引导下PTA是治疗人AVG狭窄的有效方法, 且年龄越大, 术后通畅率越低。 Abstract:ObjectiveTo investigate the effectiveness and feasibility of ultrasound-guided percutaneous transluminal angioplasty(PTA)in the treatment of arteriovenous graft(AVG)stenosis, and analyze the influencing factors of patency rate after PTA. MethodsThe inner diameter of stenosis, hemodialysis blood flow, brachial artery blood flow changes in 36 maintenance hemodialysis patients with AVG stenosis before and after PTA were retrospectively analyzed, and the therapeutic effects were evaluated.The patency rate was detected every 3 months after surgery. ResultsAll 36 patients were successfully operated.Before the treatment of PTA, the diameter of stenosis, AVG blood flow and brachial artery blood flow were(1.58±0.35)mm, (144.13±19.12)mL/min and(352.30±42.01)mL/min, respectively.After the treatment of PTA, the diameter of stenosis, AVG blood flow and brachial artery blood flow were(3.71±0.62)mm, (275.75±14.29)mL/min and(616.83±45.92)mL/min, respectively.The differences of the diameter of stenosis, AVG blood flow and brachial artery blood flow between before and after treatment were statistically significant(P < 0.01), and which indicated that the treatment was effective.The patients were followed up for 3, 6, 9 and 12 months, and the primary patency and secondary patency rates were 80.6%, 66.7%, 47.2% and 38.9%, and 88.9%, 75.0%, 69.4% and 58.3% at each time-point, respectively.There was no statistical significance at each follow-up time between primary patency rate and secondary patency rate(P>0.05).The age of patients with postoperative 1 year patency was lower than that of patients with postoperative 1 year non-patency(P < 0.01). ConclusionsThe PTA guided by ultrasound is an effective method in the treatment of AVG stenosis, and the older the patient is, the lower the postoperative patency rate is. -
表 1 PTA手术前后测量指标比较(x±s)
时间 n 狭窄处内径/mm 透析血流量/
(mL/min)肱动脉血流量/
(mL/min)术前 36 1.58±0.35 144.13±19.12 352.30±42.01 术后 36 3.71±0.62 275.75±14.29 616.83±45.92 t — 16.61 35.76 28.26 P — < 0.01 < 0.01 < 0.01 表 2 病人PTA术后通畅率情况[n; 百分率(%)]
通畅率 术后3个月 术后6个月 术后9个月 术后12个月 初级通畅率 29(80.6) 24(66.7) 17(47.2) 14(38.9) 次级通畅率 32(88.9) 27(75.0) 25(69.4) 21(58.3) χ2 0.97 0.61 3.66 2.72 P >0.05 >0.05 >0.05 >0.05 表 3 病人术后1年初级通畅率的影响因素分析[n;百分率(%)]
影响因素 通畅组
(n=14)非通畅组
(n=22)t P 年龄(x±s)/岁 42.71±6.90 58.09±8.34 5.75 < 0.01 性别 男 8(57.14) 12(54.55) — >0.05* 女 6(42.86) 10(45.45) 慢性肾小球肾炎 是 6(42.86) 12(54.55) — >0.05* 否 8(57.14) 10(45.45) 高血压 是 2(14.29) 1(4.55) — >0.05* 否 12(85.71) 21(95.45) 糖尿病 是 3(21.43) 6(27.27) — >0.05* 否 11(78.57) 16(72.73) 静脉吻合口 贵要静脉 10(71.43) 19(86.36) — >0.05* 头静脉 4(28.57) 3(13.64) 透析时间(x±s)/月 14.00±8.87 16.91±7.74 1.04 >0.05 *示Fisher′s确切概率法P值 -
[1] 金其庄, 王玉柱, 叶朝阳, 等. 中国血液透析用血管通路专家共识(第2版)[J]. 中国血液净化, 2019, 18(6): 365. doi: 10.3969/j.issn.1671-4091.2019.06.001 [2] MOUSA AY, PATTERSON W, ABU-HALIMAH S, et al. Patency in arteriovenous grafts in hemodialysis patients[J]. Vasc Endovascular Surg, 2013, 47(6): 438. doi: 10.1177/1538574413493678 [3] BACHLEDA P, UTIKAL P, KOCHER M, et al. Arteriovenous graft for hemodialysis, graft venous anastomosis closure-current state of knowledge. Minireview[J]. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub, 2015, 159(1): 27. doi: 10.5507/bp.2014.027 [4] 李宁, 彭侃夫. 维持性血液透析病人发生动静脉内瘘栓塞的危险因素分析[J]. 蚌埠医学院学报, 2017, 42(11): 1510. doi: 10.13898/j.cnki.issn.1000-2200.2017.11.023 [5] VALERIANOVA A, KUDLICKA J, CHYTILOVA E, et al. Factors influencing dialysis arteriovenous graft survival[J]. J Vasc Access, 2017, 18(2): 139. doi: 10.5301/jva.5000632 [6] PISONI RL, ZEPEL L, PORT FK, et al. Trends in US vascular access use, patient preferences, and related practices: an update from the US DOPPS practice monitor with international comparisons[J]. Am J Kidney Dis, 2015, 65(6): 905. doi: 10.1053/j.ajkd.2014.12.014 [7] ARHUIDESE IJ, ORANDI BJ, NEJIM B, et al. Utilization, patency, and complications associated with vascular access for hemodialysis in the United States[J]. J Vasc Surg, 2018, 68(4): 1166. doi: 10.1016/j.jvs.2018.01.049 [8] AGARWAL AK, HADDAD NJ, VACHHARAJANI TJ, et al. Innovations in vascular access for hemodialysis[J]. Kidney Int, 2019, 95(5): 1053. doi: 10.1016/j.kint.2018.11.046 [9] MASUD A, COSTANZO EJ, ZUCKERMAN R, et al. The complications of vascular access in hemodialysis[J]. Semin Thromb Hemost, 2018, 44(1): 57. doi: 10.1055/s-0037-1606180 [10] GLANZ S, GORDON D, BUTT KM, et al. Dialysis access fistulas: treatment of stenoses by transluminal angioplasty[J]. Radiology, 1984, 152(3): 637. doi: 10.1148/radiology.152.3.6235537 [11] 彭嘉欣, 万恒, 刘正军. 经皮腔内血管成形术治疗人工血管移植物内瘘狭窄或闭塞的疗效[J]. 中国普通外科杂志, 2019, 28(12): 1497. doi: 10.7659/j.issn.1005-6947.2019.12.009 [12] 赵意平, 郭相江, 施娅雪, 等. 高压球囊治疗人工血管动静脉内瘘狭窄的初步体会[J]. 中国血液净化, 2015, 14(1): 25. [13] 缪鹏, 谭正力, 田然, 等. 外周切割球囊在血液透析通路血管狭窄的临床应用研究[J/CD]. 中国血管外科杂志: 电子版, 2019, 11(2): 104. [14] PLANKEN RN, VAN KESTEREN F, REEKERS JA. Treatment of hemodialysis vascular access arteriovenous graft failure by percutaneous intervention[J]. J Vasc Access, 2014, 15(7_suppl): 114. doi: 10.5301/jva.5000234 [15] OH E, KIM YJ, GOO DE, et al. Percutaneous transluminal angioplasty for dysfunctional femoral hemodialysis graft[J]. Diagn Interv Radiol, 2015, 21(2): 154. doi: 10.5152/dir.2014.14231 [16] PARK J, KIM J, HWANG S, et al. Arteriovenous graft patency outcomes and prognostic factors[J]. Vascular, 2019, 27(2): 128. doi: 10.1177/1708538118807844 [17] 曾敏, 杨定平, 郑婷. 超声引导下经皮球囊扩张术治疗内瘘狭窄的疗效观察[J]. 临床肾脏病杂志, 2019, 19(1): 23. [18] 丁红, 顾奇澜, 朱宇莉, 等. 高频超声监测稳定血透患者动静脉内瘘并发症的临床价值再评价[J]. 中国临床医学影像杂志, 2015, 26(2): 118. -





 下载:
下载: