-
口腔鳞癌(OSCC)是最常见的头颈部肿瘤亚型,全球每年有超过35万新确诊病例和18万死亡病例[1]。尽管在手术技术和放化疗方面取得了较大进展,但OSCC病人总生存率并未得到明显改善。OSCC具有局部侵袭性强和颈部淋巴结转移特点,深入研究其进展和转移分子机制是开发新型有效OSCC治疗方法的关键。环状RNA(circRNA)是由共价键形成的闭合型RNA分子,其包含miRNA特异性结合位点,可通过抑制miRNA的调控作用来调节mRNA表达,广泛参与细胞代谢、增殖、转移和凋亡等诸多生物学过程[2]。circFOXK2在胰管腺癌细胞和大多数原发肿瘤中显著上调,促进细胞生长和侵袭,参与细胞周期进展[3]。然而目前鲜见circFOXK2和OSCC的相关报道。miR-4677-3p是肺癌的抑制因子,上调其表达能够抑制肺癌细胞增殖和迁移[4]。此外,miR-4677-3p还介导敲减circ_0001421对肺癌细胞的恶性行为和糖酵解的抑制作用[5]。靶基因预测显示miR-4677-3p是circFOXK2的潜在靶点,但circFOXK2靶向miR-4677-3p在OSCC进展中的调控作用并不清楚。为此,本研究重点探讨circFOXK2和miR-4677-3p对OSCC恶性行为的影响和调控机制,旨在为OSCC的防治提供重要的实验数据和理论依据。
-
OSCC组织和匹配癌旁组织来源于2017年3月至2020年12月在我院行手术治疗的45例OSCC病人,其中男32例,女13例,年龄48~75岁。所有OSCC病人在手术前均未接受过辅助放疗或化疗。所有样本均由我院2位病理专家证实,然后立即在-80 ℃保存。病人均签署知情同意书,且该研究获得了我院伦理委员会的批准。
-
OSCC细胞HSC-3购自美国ATCC;Transwell板购自美国Corning公司;细胞计数试剂盒(CCK-8)购自北京博奥森公司;磷酸甘油醛脱氢酶(GAPDH)兔多抗(ab9485)、基质金属蛋白酶2(MMP-2)兔多抗(ab97779)、MMP-9兔多抗(ab38898)购自上海艾博抗公司;重组报告质粒、si-NC、si-circFOXK2、模拟物阴性对照miR-NC、miR-4677-3p模拟物、pcDNA-circFOXK2、抑制物阴性对照anti-miR-NC、miR-4677-3p抑制物(anti-miR-4677-3p)、pcDNA购自苏州鸿迅生物;逆转录试剂盒购自大连宝生生物公司;Power SYBRTM Green PCR Master Mix购自美国Thermo Fisher公司。
-
采用Trizol试剂从OSCC组织中分离总RNA,RNA质量和浓度通过紫外分光光度法确定。用逆转录试剂盒合成cDNA,用TB GreenTM Premix Ex TaqTM Ⅱ进行RT-qPCR。用2-ΔΔCt法方法计算circFOXK2(GAPDH为内参)和miR-4677-3p(U6为内参)相对表达量。
-
HSC-3细胞在补充有10%胎牛血清和1%青链霉素双抗的DMEM/F12培养基中培养,培养箱环境为37 ℃、含5%CO2。在6孔板中接种5×105个对数期HSC-3细胞,将Lipo2000与si-NC、si-circFOXK2、miR-NC、miR-4677-3p模拟物、pcDNA、pcDNA-circFOXK2、si-circFOXK2+anti-miR-NC、si-circFOXK2+anti-miR-4677-3p加入到60%融合度的HSC-3细胞中,培养48 h收获细胞,RT-qPCR检测circFOXK2或miR-4677-3p表达后用于进一步实验。根据转染序列或载体不同共分为si-NC组、si-circFOXK2组、miR-NC组、miR-4677-3p组、pcDNA组、pcDNA-circFOXK2组、si-circFOXK2+anti-miR-NC组、si-circFOXK2+ anti-miR-4677-3p组。
-
转染48 h后,用胰酶消化细胞,将1×105个转染细胞接种到6孔板,培养2周后,去除培养基,用4%多聚甲醛固定细胞集落10 min,随后将细胞用0.5%结晶紫染色。显微镜下计数>50个细胞的克隆数。
-
在96孔板中接种1×105个细胞,孵育2 d后每孔加入10 μL的CCK-8试剂,继续孵育2 h,用酶标仪在450 nm处测定吸光度(OD)值。
-
用记号笔在6孔板背面划一条直线。在6孔板中接种5×105个转染细胞,37 ℃培养箱孵育至细胞基本融合,用200 μL移液管尖端沿6孔板背面直线划伤单层细胞,PBS洗涤去除细胞碎片。继续孵育24 h,显微镜下拍照,Image J软件测定划痕宽度,并计算划痕愈合率。划痕愈合率(%)=(0 h划痕宽度-24 h划痕宽度)/0 h划痕宽度×100%。
-
用无血清培养基重悬转染48 h的HSC-3细胞,取200 μL接种在预包被基质胶的上室中。在下室中加入完全培养液作为引诱剂。37 ℃培养24 h,用棉签轻轻去除留在上表面的HSC-3细胞,将穿膜细胞固定,并进行结晶紫染色。在光学显微镜下随机选择5个视野拍照,以细胞数的均值表示侵袭数。
-
用预冷RIPA缓冲液提取总蛋白质,然后在SDS-PAGE上分离并转移到聚偏二氟乙烯膜上。在5%脱脂牛奶中封闭1 h,将膜与适当浓度的一抗(包括MMP-2、MMP-9和内参GAPDH抗体)在4 ℃孵育过夜。用酶标二抗在室温下与膜结合2 h。用化学发光检测试剂盒检测蛋白条带,并使用Quantity One软件分析目的蛋白相对灰度值。
-
分别将包含miR-4677-3p结合位点的circFOXK2野生(WT)序列或突变(MUT)序列分别克隆到psiCHECK-2报告载体,产生WT-circFOXK2、MUT-circFOXK2。在HSC-3细胞中共转染报告质粒与miR-NC或miR-4677-3p模拟物,转染48 h后检测细胞裂解物中相对荧光素酶活性。
-
采用t检验、方差分析和q检验。
-
OSCC组织circFOXK2表达水平显著高于癌旁组织(P < 0.01),miR-4677-3p表达水平显著低于癌旁组织(P < 0.01)(见表 1)。
分组 n circFOXK2 miR-4677-3p 癌旁组织 45 1.00±0.08 1.00±0.09 OSCC组织 45 3.67±0.33 0.38±0.05 t — 52.75 40.40 P — < 0.01 < 0.01 表 1 OSCC组织中circFOXK2和miR-4677-3p表达(x±s)
-
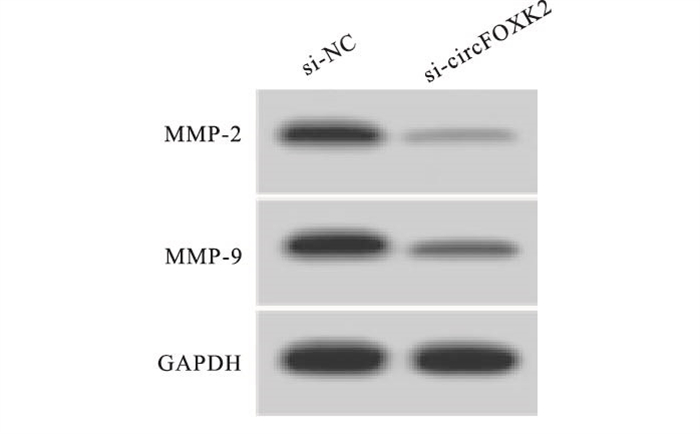
与转染si-NC相比,转染si-circFOXK2导致HSC3细胞circFOXK2表达水平降低,差异有统计学意义(P < 0.01)。与si-NC组相比,si-circFOXK2组HSC3细胞MMP-2和MMP-9蛋白水平、细胞OD值、克隆形成数、划痕愈合率、侵袭数均显著降低,差异有统计学意义(P < 0.01)(见表 2、图 1)。
分组 circFOXK2 OD值(450 nm) 细胞克隆形成数/个 侵袭细胞数/个 划痕愈合率/% MMP-2蛋白 MMP-9蛋白 si-NC 1.00±0.00 0.77±0.06 85.57±6.95 111.51±11.89 67.14±4.36 0.57±0.04 0.74±0.06 si-circFOXK2 0.27±0.02 0.35±0.03 36.23±4.43 52.25±5.19 26.96±2.61 0.16±0.02 0.29±0.03 t 109.50 18.78 17.96 13.70 23.72 27.50 20.13 P < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 表 2 抑制circFOXK2表达对HSC3细胞增殖、侵袭和迁移的影响(x±s;ni=9)
-
与转染miR-NC比较, 转染miR-4677-3p模拟物导致HSC3细胞miR-4677-3p表达水平升高,差异有统计学意义(P < 0.01)。miR-4677-3p组HSC3细胞MMP-2和MMP-9蛋白水平、细胞OD值、克隆形成数、划痕愈合率、侵袭数显著低于miR-NC组(P < 0.01)(见图 2、表 3)。
分组 miR-4677-3p OD值(450 nm) 细胞克隆形成数/个 侵袭细胞数/个 划痕愈合率/% MMP-2蛋白 MMP-9蛋白 miR-NC 1.00±0.00 0.78±0.07 87.44±5.39 115.72±11.24 68.84±5.21 0.58±0.04 0.76±0.06 miR-4677-3p 3.59±0.26 0.43±0.04 42.95±4.12 60.85±4.87 32.41±3.47 0.25±0.02 0.33±0.03 t 29.80 13.02 19.67 13.44 17.46 22.14 19.23 P < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 表 3 miR-4677-3p过表达对HSC3细胞增殖、侵袭和迁移的影响(x±s;ni=9)
-
StarBase预测到circFOXK2的序列中含有miR-4677-3p的结合位点(见图 3)。与WT-circFOXK2共转染时,转染miR-4677-3p模拟物与转染miR-NC相比导致细胞相对荧光素酶活性降低,差异有统计学意义(P < 0.01) (见表 4)。pcDNA-circFOXK2组HSC3细胞miR-4677-3p表达水平显著低于pcDNA组(P < 0.05),si-circFOXK2组HSC3细胞miR-4677-3p表达水平显著高于si-NC组(P < 0.05)(见表 5)。
分组 n WT-circFOXK2 MUT-circFOXK2 miR-NC 9 1.04±0.09 1.05±0.11 miR-4677-3p 9 0.51±0.04 1.01±0.10 t — 16.14 0.81 P — < 0.01 >0.05 表 4 双荧光素酶报告实验(x±s)
分组 n miR-4677-3p F P MS组内 pcDNA 9 1.00±0.00 pcDNA-circFOXK2 9 0.44±0.04* 518.96 < 0.01 0.018 si-NC 9 0.98±0.09 si-circFOXK2 9 2.78±0.25# q检验:与pcDNA组比较*P < 0.05;与si-NC组比较#P < 0.05 表 5 circFOXK2调控miR-4677-3p的表达(x±s; ni=9)
-
si-circFOXK2+anti-miR-4677-3p组HSC3细胞miR-4677-3p表达水平显著低于si-circFOXK2+anti-miR-NC组(P < 0.01),MMP-2和MMP-9蛋白水平、细胞OD值、克隆形成数、划痕愈合率、侵袭数显著高于si-circFOXK2+anti-miR-NC组(P < 0.01)(见图 4、表 6)。
分组 miR-4677-3p OD值(450 nm) 细胞克隆形成数/个 侵袭细胞数/个 划痕愈合率/% MMP-2蛋白 MMP-9蛋白 si-circFOXK2+anti-miR-NC 1.00±0.00 0.33±0.03 35.34±3.68 50.76±4.18 24.24±3.25 0.14±0.02 0.26±0.03 si-circFOXK2+anti-miR-4677-3p 0.36±0.03 0.68±0.05 74.84±5.97 99.03±8.37 58.39±4.81 0.45±0.04 0.63±0.05 t 64.00 18.01 16.90 15.48 17.65 20.80 19.04 P < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 表 6 干扰miR-4677-3p表达逆转抑制circFOXK2表达对HSC3细胞增殖、侵袭和迁移的作用(x±s;ni=9)
-
circRNA是新发现的功能性调节非编码RNA,特定circRNA差异表达与各种癌症的临床特征显著相关。目前已发现circ_0000140低表达与OSCC病人预后不良相关[6]。circ_101036通过诱导内质网应激来抑制OSCC细胞迁移和侵袭[7]。然而circ_002178高表达OSCC病人晚期病理分期和远端转移发生率较高,敲除circ_002178能够有效抑制OSCC细胞增殖和侵袭[8]。阐明circRNA在OSCC发生和发展机制中的作用将为OSCC的治疗提供新的方向。本研究检测到OSCC组织中circFOXK2表达增加,抑制circFOXK2表达导致HSC3细胞活力和克隆形成数降低,迁移和侵袭能力降低,表明抑制circFOXK2表达具有抗增殖、抗转移作用。肿瘤转移涉及多个步骤,其中MMPs是肿瘤细胞降解细胞外基质并向局部或远处延伸的必要条件[9]。在包括OSCC在内的多种肿瘤中MMP-2和MMP-9异常表达或激活与肿瘤侵袭或转移有关[10-11]。本研究检测转移相关标志蛋白MMP-2和MMP-9的表达水平,结果显示,抑制circFOXK2表达导致HSC3细胞中MMP-2和MMP-9蛋白下调,transwell实验结果与之一致。以上研究表明circFOXK2在OSCC进展中扮演致癌基因角色。
研究[12-13]显示circRNA可结合miRNA调控肿瘤进展。本研究证实miR-4677-3p是circFOXK2的靶点,过表达circFOXK2可抑制miR-4677-3p表达,而抑制circFOXK2则促进miR-4677-3p表达。miR-4677-3p已被证明在人类癌症中异常表达并调控癌症进展。如在胃癌中miR-4677-3p异常低表达,miR-142-5p的上调可抑制胃癌细胞增殖和迁移[14]; LINC00313通过下调miR-4677-3p表达水平,诱导CDK6表达上调,从而促进宫颈癌进展[15]; miR-4677-3p还作为TTN-AS1和LINC02418等lncRNA的下游miRNA来抑制肺癌进展[16-17]。本研究中,OSCC组织中miR-4677-3p表达明显降低,过表达miR-4677-3p可下调MMP-2和MMP-9蛋白表达,并抑制HSC3细胞活力、迁移、克隆形成和侵袭能力降低,这与抑制circFOXK2表达的抗癌作用类似。为证实抑制circFOXK2的作用是通过上调miR-4677-3p表达实现的,本研究共转染si-circFOXK2和anti-miR-4677-3p至HSC3细胞,结果显示,下调miR-4677-3p表达显著减弱抑制circFOXK2表达对HSC3细胞恶性行为的影响。这些结果表明circFOXK2靶向miR-4677-3p调控OSCC细胞的恶性行为。然而,本研究并未在动物实验中验证circFOXK2和miR-4677-3p的功能,这将是下一步研究的重点。
总之,本研究表明抑制circFOXK2可通过促进miR-4677-3p表达来抑制OSCC细胞增殖、迁移和侵袭,这些发现有助于理解OSCC的发病机制,并可能为OSCC的治疗提供新靶点。
circFOXK2靶向miR-4677-3p促进口腔鳞癌细胞增殖、侵袭和迁移
circFOXK2 targets miR-4677-3p to promote the proliferation, invasion and migration of oral squamous cell carcinoma cells
-
摘要:
目的探讨circFOXK2靶向miR-4677-3p对口腔鳞癌(OSCC)细胞恶性行为的影响。 方法RT-qPCR检测circFOXK2和miR-4677-3p在OSCC组织和癌旁组织的表达。分别将si-circFOXK2、si-NC、miR-NC、miR-4677-3p模拟物、pcDNA、pcDNA-circFOXK2、si-circFOXK2+anti-miR-NC、si-circFOXK2+anti-miR-4677-3p转染OSCC细胞HSC3。通过划痕愈合实验、CCK-8法、Transwell实验、集落形成实验检测circFOXK2和miR-4677-3p表达对HSC3细胞恶行行为的影响。circFOXK2和miR-4677-3p的靶向关系通过双荧光素酶法确定。 结果OSCC组织中circFOXK2表达水平显著高于癌旁组织(P < 0.01),miR-4677-3p表达水平显著低于癌旁组织(P < 0.01)。抑制circFOXK2表达后HSC3细胞吸光度(OD)值、划痕愈合率、克隆形成数、侵袭数显著降低(P < 0.01),miR-4677-3p表达水平显著升高(P < 0.01)。过表达miR-4677-3p后HSC3细胞OD值、划痕愈合率、克隆形成数、侵袭数显著降低(P < 0.01)。miR-4677-3p是circFOXK2的靶基因。下调miR-4677-3p显著减弱抑制circFOXK2表达对HSC3细胞OD值、划痕愈合率、克隆形成数、侵袭数的影响(P < 0.01)。 结论抑制circFOXK2可通过促进miR-4677-3p表达来抑制OSCC细胞增殖、迁移和侵袭。 Abstract:ObjectiveTo study the effect of circFOXK2 targeting miR-4677-3p on the malignant behavior of oral squamous cell carcinoma (OSCC) cells. MethodsThe expression of circFOXK2 and miR-4677-3p in OSCC tissues and adjacent tissues was calculated by RT-qPCR.Si-circFOXK2, si-NC, miR-NC, miR-4677-3p mimic, pcDNA, pcDNA-circFOXK2, si-circFOXK2+anti-miR-NC, and si-circFOXK2+anti-miR-4677-3p were respectively transfected into OSCC cells HSC3.The effects of circFOXK2 and miR-4677-3p expression on the malignant behavior of HSC3 cells were detected by scratch healing, CCK-8, Transwell and colony formation assays.The targeting relationship between circFOXK2 and miR-4677-3p was determined by dual luciferase method. ResultsThe expression level of circFOXK2 in OSCC tissue was significantly higher than that in adjacent tissues (P < 0.01), while the expression level of miR-4677-3p was significantly lower than that in adjacent tissues (P < 0.01).After inhibiting the expression of circFOXK2, the HSC3 cell optical density (OD) value, scratch healing rate, clone formation number and invasion number were significantly reduced (P < 0.01), and the expression level of miR-4677-3p was significantly increased (P < 0.01).After overexpression of miR-4677-3p, the HSC3 cell OD value, scratch healing rate, clone formation number and invasion number were significantly reduced (P < 0.01).MiR-4677-3p was a target gene of circFOXK2.MiR-4677-3p down-regulation significantly reduced the effect of inhibiting circFOXK2 on cell OD value, scratch healing rate, clone formation number, and number of invasion of HSC3 cells (P < 0.01). ConclusionsInhibition of circFOXK2 can suppress the proliferation, migration and invasion of OSCC cells by promoting miR-4677-3p expression. -
Key words:
- oral squamous cell carcinoma /
- circFOXK2 /
- miR-4677-3p /
- proliferation /
- migration /
- invasion
-
表 1 OSCC组织中circFOXK2和miR-4677-3p表达(x±s)
分组 n circFOXK2 miR-4677-3p 癌旁组织 45 1.00±0.08 1.00±0.09 OSCC组织 45 3.67±0.33 0.38±0.05 t — 52.75 40.40 P — < 0.01 < 0.01 表 2 抑制circFOXK2表达对HSC3细胞增殖、侵袭和迁移的影响(x±s;ni=9)
分组 circFOXK2 OD值(450 nm) 细胞克隆形成数/个 侵袭细胞数/个 划痕愈合率/% MMP-2蛋白 MMP-9蛋白 si-NC 1.00±0.00 0.77±0.06 85.57±6.95 111.51±11.89 67.14±4.36 0.57±0.04 0.74±0.06 si-circFOXK2 0.27±0.02 0.35±0.03 36.23±4.43 52.25±5.19 26.96±2.61 0.16±0.02 0.29±0.03 t 109.50 18.78 17.96 13.70 23.72 27.50 20.13 P < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 表 3 miR-4677-3p过表达对HSC3细胞增殖、侵袭和迁移的影响(x±s;ni=9)
分组 miR-4677-3p OD值(450 nm) 细胞克隆形成数/个 侵袭细胞数/个 划痕愈合率/% MMP-2蛋白 MMP-9蛋白 miR-NC 1.00±0.00 0.78±0.07 87.44±5.39 115.72±11.24 68.84±5.21 0.58±0.04 0.76±0.06 miR-4677-3p 3.59±0.26 0.43±0.04 42.95±4.12 60.85±4.87 32.41±3.47 0.25±0.02 0.33±0.03 t 29.80 13.02 19.67 13.44 17.46 22.14 19.23 P < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 表 4 双荧光素酶报告实验(x±s)
分组 n WT-circFOXK2 MUT-circFOXK2 miR-NC 9 1.04±0.09 1.05±0.11 miR-4677-3p 9 0.51±0.04 1.01±0.10 t — 16.14 0.81 P — < 0.01 >0.05 表 5 circFOXK2调控miR-4677-3p的表达(x±s; ni=9)
分组 n miR-4677-3p F P MS组内 pcDNA 9 1.00±0.00 pcDNA-circFOXK2 9 0.44±0.04* 518.96 < 0.01 0.018 si-NC 9 0.98±0.09 si-circFOXK2 9 2.78±0.25# q检验:与pcDNA组比较*P < 0.05;与si-NC组比较#P < 0.05 表 6 干扰miR-4677-3p表达逆转抑制circFOXK2表达对HSC3细胞增殖、侵袭和迁移的作用(x±s;ni=9)
分组 miR-4677-3p OD值(450 nm) 细胞克隆形成数/个 侵袭细胞数/个 划痕愈合率/% MMP-2蛋白 MMP-9蛋白 si-circFOXK2+anti-miR-NC 1.00±0.00 0.33±0.03 35.34±3.68 50.76±4.18 24.24±3.25 0.14±0.02 0.26±0.03 si-circFOXK2+anti-miR-4677-3p 0.36±0.03 0.68±0.05 74.84±5.97 99.03±8.37 58.39±4.81 0.45±0.04 0.63±0.05 t 64.00 18.01 16.90 15.48 17.65 20.80 19.04 P < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 < 0.01 -
[1] BRAY F, FERLAY J, SOERJOMATARAM I, et al. Global cancer statistics 2018: globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6): 394. doi: 10.3322/caac.21492 [2] 吴银洁, 徐晓彦, 徐飞, 等. 环状RNA的海绵活性作为肿瘤新型生物标志物可行性的研究进展[J]. 临床肿瘤学杂志, 2018, 23(1): 79. [3] WONG CH, LOU UK, LI Y, et al. CircFOXK2 promotes growth and metastasis of pancreatic ductal adenocarcinoma by complexing with rna-binding proteins and sponging miR-942[J]. Cancer Res, 2020, 80(11): 2138. doi: 10.1158/0008-5472.CAN-19-3268 [4] WANG T, ZHAI R, LÜ X, et al. LINC02418 promotes malignant behaviors in lung adenocarcinoma cells by sponging miR-4677-3p to upregulate KNL1 expression[J]. BMC Pulm Med, 2020, 20(1): 217. doi: 10.1186/s12890-020-01229-0 [5] ZHANG K, HU H, XU J, et al. Circ_0001421 facilitates glycolysis and lung cancer development by regulating miR-4677-3p/CDCA3[J]. Diagn Pathol, 2020, 15(1): 133. doi: 10.1186/s13000-020-01048-1 [6] PENG QS, CHENG YN, ZHANG WB, et al. circRNA_0000140 suppresses oral squamous cell carcinoma growth and metastasis by targeting miR-31 to inhibit Hippo signaling pathway[J]. Cell Death Dis, 2020, 11(2): 112. doi: 10.1038/s41419-020-2273-y [7] DENG W, FU J, WANG T, et al. Hsa_circRNA_101036 acts as tumor-suppressor in oral squamous cell carcinoma cells via inducing endoplasmic reticulum stress[J]. Eur Rev Med Pharmacol Sci, 2020, 24(11): 6111. [8] YANG Y, CI HS, MAO YL, et al. CircRNA_002178 promotes the proliferation and migration of oral squamous cell carcinoma cells by activating the Akt/mTOR pathway[J]. Eur Rev Med Pharmacol Sci, 2020, 24(11): 6122. [9] 贾玉杰, 崔慧斐. 以MMPs为靶点的抗肿瘤药物研究[J]. 生命的化学, 2017, 37(5): 733. [10] 王璞, 韦丽宾, 倪广晓. CCNB1基因通过调控PI3K/Akt信号通路对口腔鳞癌细胞增殖、侵袭和迁移的影响[J]. 实用肿瘤学杂志, 2020, 34(2): 144. [11] SHUANG Y, LI C, ZHOU X, et al. MicroRNA-195 inhibits growth and invasion of laryngeal carcinoma cells by directly targeting DCUN1D1[J]. Oncol Rep, 2017, 38(4): 2155. doi: 10.3892/or.2017.5875 [12] 周海霞, 王璐瑶, 陈帅, 等. 环状RNA hsa_circ_0008898对口腔鳞状细胞癌细胞的影响及其机制研究[J]. 中华口腔医学杂志, 2020, 55(8): 578. [13] 余昆, 李强, 王伟雅, 等. 环状RNA RNF13通过下调miR-196b-5p的表达促进结直肠癌细胞的增殖[J]. 肿瘤, 2019, 39(11): 887. [14] MI C, ZHANG D, LI Y, et al. miR-4677-3p participates proliferation and metastases of gastric cancer cell via CEMIP-PI3K/AKT signaling pathway[J]. Cell Cycle, 2021, 20(19): 1978. doi: 10.1080/15384101.2021.1971375 [15] ZHAI Y, LIU Y, WANG Z, et al. Long non-coding RNA LINC00313 accelerates cervical carcinoma progression by miR-4677-3p/CDK6 axis[J]. Onco Targets Ther, 2021, 14(1): 2213. [16] ZHONG Y, WANG J, LÜ W, et al. LncRNA TTN-AS1 drives invasion and migration of lung adenocarcinoma cells via modulation of miR-4677-3p/ZEB1 axis[J]. J Cell Biochem, 2019, 120(10): 17131. doi: 10.1002/jcb.28973 [17] HAN B. LncRNA LINC02418 regulates proliferation and apoptosis of non-small cell lung cancer cells by regulating miR-4677-3p/SEC61G[J]. Eur Rev Med Pharmacol Sci, 2019, 23(23): 10354. -





 下载:
下载: