-
胆管癌(cholangiocarcinoma, CCA)是一种起源于胆管上皮的恶性肿瘤,发病率约为(1~2)/10万人[1]。根据解剖位置,CCA可分为肝内胆管癌(intrahepatic CCA, iCCA)、肝门部胆管癌(perihilar CCA, pCCA)和远端胆管癌(distal cholangiocarcinoma, dCCA),后两者属于肝外胆管癌(extrahepatic CCA, EHCC)。迄今为止,根治性切除仍然是早期EHCC最有效的治疗方法。由于大多数EHCC就诊时已处于晚期[2-3],接受根治性切除的比例仅为20%~35%[4-5]。此外,49%~64%的EHCC病人术后发生复发或转移[6]。因此,EHCC的预后较差,pCCA和dCCA的5年总生存率(OS)约为11%~41%和27%~37%[7]。
对于不能行根治性切除的晚期EHCC病人,姑息治疗方式包括姑息性胆肠内引流术、经皮肝或经内镜胆道支架置入、射频消融术、放化疗、分子靶向治疗等[8-9]。经姑息性手术(PS)或胆道支架植入等途径行胆道引流可使病人恢复胆汁循环,从而减轻病人症状,提高生活质量及延长生存时间[10]。对于晚期EHCC病人,通过何种途径行胆道引流疗效更好的研究,目前国内外报道很少。本研究比较自膨式金属支架联合导管载入式125I粒子(self-expandable metallic stent combined with catheter loaded iodine-125 seeds, SEMS-CL-125I)及PS治疗晚期EHCC的疗效。
-
选择2014年1月至2018年6月在我科住院的晚期EHCC病人。纳入标准:(1)病理诊断为pCCA或DCCA,经多学科讨论后认为不适合做根治性切除;(2)病人不能耐受或不愿行根治性切除;(3)首次接受SEMS-CL-125I(SEMS-CL-125I组)或胆囊切除联合胆管空肠吻合术(PS组)。排除标准:(1)良性胆道疾病;(2)远处转移;(3)既往因EHCC接受过手术、内镜下胆道支架植入或化疗等治疗;(4)随访等资料不完整。最终纳入101例病人。其中,SEMS-CL-125I组67例,男47例,女20例,年龄>70岁41例,pCCA 28例;PS组34例,男27例,女7例,年龄>70岁15例,pCCA 28例。2组性别、年龄、肿瘤部位、血清总胆红素(TBIL)、直接胆红素(DBIL)、丙氨酸氨基转移酶(ALT)、天冬氨酸转氨酶(AST)、白蛋白(ALB)、糖类抗原19-9(CA19-9)和癌胚抗原(CEA)等差异均无统计学意义(P>0.05),具有可比性。本研究遵守赫尔辛基宣言[11],并通过蚌埠医学院第一附属医院伦理委员会的批准。
变量 总体 SEMS-CL-125I组
(n=67)PS组
(n=34)Z P 性别(男/女) 74/27 47/20 27/7 0.99* 0.05 年龄(≥70/ < 70)/岁 56/45 41/26 15/19 2.66* 0.05 肿瘤部位(pCCA/dCCA) 36/65 28/39 8/26 3.28* 0.05 CA19-9/ (ng/mL) 278.9(62.5~797.8) 293.0(72.5~989.8) 128.6(34.9~471.0) 1.61 0.05 CEA/(ng/mL) 4.2(2.6~6.4) 4.0(2.4~6.1) 4.3(2.8~6.7) 0.82 0.05 ALT/(U/L) 125(81~248) 131(92~239) 104(58~269) 1.00 0.05 AST/(U/L) 133(80.5~169) 137(97~191) 166(61~167) 1.72 0.05 TBIL/(μmol/L) 221(127~319) 244(150~343) 178(99~293) 1.67 0.05 DBIL/(μmol/L) 182.0(106.9~284.8) 194.8(121.7~298.8) 142.8(80.4~234.4) 1.74 0.05 ALB/(g/L) 34.2(31.4~37.7) 33.6(31.1~37.2) 35.1(32.9~39.9) 1.60 0.05 *示χ2值 表 1 基线资料比较
-
SEMS-CL-125I组67例均成功置入胆道支架和125I粒子。SEMS-CL-125I的具体操作过程已在前文进行描述[12-14]。PS组行姑息性手术。2组病人均进行随访,随访截止至2019年6月或死亡。随访内容:术后1、3、6个月行血清肿瘤标志物、生化常规、腹部B超、CT检查。
-
采用秩和检验、χ2检验、方差分析和q检验。
-
与术前相比,2组病人术后1、3、6个月血清TBIL、ALT、AST水平均降低,而ALB均升高,差异有统计学意义(P < 0.05)。SEMS-CL-125I组术后1个月ALB水平显著高于PS组(P < 0.05)(见表 2)。
分组 n 术前 术后1月 术后3月 术后6月 P F MS组内 ALT/(U/L) SEMS-CL-125I组 67 169.16±106.71 46.93±29.67** 37.81±29.72** 41.75±37.47** < 0.01 74.50 3 638.650 PS组 34 163.12±140.72 56.06±35.62** 40.38±15.56** 48.20±28.44** < 0.01 20.50 5 530.460 t — 0.24 1.36 0.47 0.88 — — — P — 0.05 0.05 0.05 0.05 — — — AST/(U/L) SEMS-CL-125I组 67 152.76±91.04 54.72±30.87** 41.22±34.11** 44.70±45.01** < 0.01 61.10 3 107.660 PS组 34 120.41±65.09 51.48±29.97** 35.50±13.18** 47.80±24.75** < 0.01 33.80 1 480.300 t — 1.84 0.50 0.94 0.37 — — — P — 0.05 0.05 0.05 0.05 — — — TBIL/(U/L) SEMS-CL-125I组 67 250.01±132.09 39.82±32.79** 17.63±22.01** 17.53±10.93** < 0.01 179.0 0 4 781.710 PS组 34 212.42±144.91 54.25±48.47** 15.16±10.95** 23.29±16.76** < 0.01 48.80 5 937.260 t — 1.31 1.77 0.62 2.08 — — — P — 0.05 0.05 0.05 0.05 — — — ALB/(g/L) SEMS-CL-125I组 67 34.45±4.87 39.07±3.83** 40.72±3.39** 39.86±3.32** < 0.01 34.50 15.225 PS组 34 35.80±4.38 36.60±5.58 39.65±2.09** 39.28±2.66* < 0.01 41.90 15.441 t — 1.36 2.61 1.68 0.88 — — — P — 0.05 0.05 0.05 0.05 — — — 与同组术前比较*P<0.05,**P<0.01 表 2 2组术后1、3、6个月血清中ALT、AST、TBIL及ALB的变化情况
-
SEMS-CL-125I组67例中术后有12例(17.9%)出现并发症,其中胰腺炎或血清淀粉酶升高6例,严重电解质紊乱3例,胆道感染3例。PS组术后12例(35.3%)出现并发症,其中吻合口瘘5例,切口感染4例,切口裂开1例,伤口出血1例,胃瘫1例。2组均未发生粒子放射相关的并发症。2组术后总体并发症差异无统计学意义(χ2=3.76,P>0.05)。与PS组病人的住院时间17~23 d相比,SEMS-CL-125I组12~18 d明显缩短(χ2=4.02,P < 0.01);与PS组病人的住院费用42 912~59 874元相比,SEMS-CL-125I组32 147~41 067元明显减少(χ2=6.06,P < 0.01)。
-
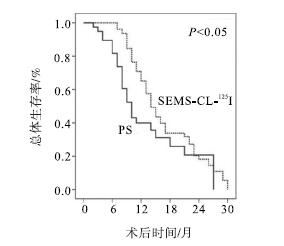
2组病人均获得了完整的随访,最长随访时间为30个月。SEMS-CL-125I组中分别有33例(49.3%)、12例(17.9%)及2例(3.0%)于术后6个月、12个月及18个月重新植入新鲜的125I粒子。至随访终点,SEMS-CL-125I组病死44例(65.7%),PS组病死24例(70.6%)。与PS组相比,SEMS-CL-125I组病人OS显著改善(中位生存时间14个月和10个月,P < 0.05)(见图 1)。SEMS-CL-125I组病人1年、2年OS率分别为65.0%和40.0%,而PS组则分别为20.7%和18.1%。肿瘤部位pCCA病人SEMS-CL-125I组可显著改善OS(P < 0.05),而在dCCA中2组OS差异无统计学意义(P>0.05)(见图 2)。术前TBIL、CA19-9、ALB和治疗方法是与术后OS相关的主要因素(见表 3)。
变量 OR(95%CI) P 性别(男/女) 1.040(0.654-1.652) >0.05 年龄(≥70/ < 0岁) 1.227(0.787-1.911) >0.05 肿瘤部位(pCCA/dCCA) 1.174(0.749-1.841) >0.05 CA19-9 (H vs L) 1.960(1.239-3.101) < 0.01 CEA (H vs L) 1.208(0.761-1.916) >0.05 ALT (H vs L) 1.027(0.655-1.610) >0.05 AST (H vs L) 1.145(0.689-1.901) >0.05 TBIL (H vs L) 1.762(1.124-2.762) < 0.05 ALB (L vs H) 1.757(1.111-2.779) < 0.05 治疗方法 1.640(1.030-2.611) < 0.05 (PS vs SEMS-CL-125I) 表 3 影响病人术后OS的单因素分析
-
因可缓解黄疸及提高生活质量,胆道引流是晚期EHCC主要的治疗方法之一[15]。其中,PS是胆道内引流的主要途径,可减轻胆道梗阻、改善肝功能、提高病人的生活质量,并延长生存时间。然而,PS有许多局限性,如术中出血风险大、术后并发症发生率高、伤口疼痛明显、住院时间长、住院费用高、预后改善不明显等[16]。
近年来,胆道支架植入已逐渐成为晚期EHCC的重要治疗方法[17-18]。与PS相比,胆道支架植入术具有创伤小、疗效相当等特点。然而,单独胆道支架植入并不能抑制肿瘤的生长,因而容易引起胆道支架的阻塞[19]。相比之下,125I近距离放射可通过释放大剂量的γ射线抑制肿瘤的进展[20]。研究[19-21]表明,与植入普通的支架相比,植入含125I的支架可延长支架通畅时间,降低支架再狭窄风险及延长病人生存时间。一项最新的荟萃分析进一步证实,与单独胆道支架相比,联合腔内125I近距离照射显着改善了病人的生存时间[22]。
SEMS-CL-125I是胆道支架置入与125I粒子植入术相结合的新兴技术,自2012年开始在我科开展应用。SEMS-CL-125I在恢复肝功能、降低胆道再梗阻风险和提高存活率方面优于单纯胆道支架植入[22]。SEMS-CL-125I操作过程中使用了独特的“P”型管,可以自由调节125I粒子的数量和分布,实现了精准内放疗。此外,在术后6个月失效时,125I粒子方便更换,避免了重复穿刺。本研究中,SEMS-CL-125I组近一半(49.3%)的病人接受了新鲜125I粒子的再植入。但病人出院后需带管在身,在日常生活中稍有不便。此外,由于“P”型管材质问题,可能引起引流管脱出支架外或125I粒子在管腔内活动。我们已选择更适合的“P”型管材质,同时在放置粒子后,在治疗腔剩余腔内放置软质导丝加以固定。此外,需加强病人及其家属日常护理教育,注意125I粒子污染防护,减少脱出及逆行感染风险。
由于植入的放射性粒子对局部肿瘤有直接作用,肿瘤内的放射活性远高于周围正常组织。因此,与外照射相比,内放射治疗引起的胃肠道溃疡、出血和肠炎等并发症明显减少。本研究中,SEMS-CL-125I组病人无一例发生放射相关的并发症,提示该治疗较为安全。我们还发现,SEMS-CL-125I在改善肝功能和缓解症状方面与PS相比疗效相当。由于创伤较小,SEMS-CL-125I组病人的住院时间明显缩短,经济负担也显著降低。更为重要的是,与PS组相比,SEMS-CL-125I组病人的生存时间明显延长。与既往报道[23]一致,术前CA19-9也是影响EHCC病人预后的独立危险因素。
多种方法已被用于获取非手术的EHCC病人的病理标本。其中,内镜逆行胰胆管造影基础上行胆道活检或刷检细胞学是一线诊断方法,但敏感性较低[24-25]。相比之下,通过胆道镜获取组织标本进行活检对胆道病变的诊断具有中度的敏感性[26]。然而,由于普通胆道镜管径较粗,不能一次直接进入相对较细的胆道引流管,而经皮穿刺连续扩张窦道可能需要较长的时间,从而了延误治疗。自2014年起,本中心将超细胆道镜应用于不能切除的EHCC病人。超细胆道镜可通过胆道引流管观察胆道病变的大小、形态、边界,并获取病理标本。本研究中,SEMS-CL-125I组病人均经超细胆道镜取材成功。
本研究仍有局限性。首先,本研究属于单中心回顾性队列研究,因此证据水平相对较低,可能存在不可避免的偏倚。此外,由于我们排除了无病理确诊的EHCC病人,研究的样本量相对较小。因此,仍需要多中心、大规模的前瞻性研究或随机对照研究。其次,研究结果可能会受潜在混杂因素的影响,如肿瘤分期、肿瘤直径等。
综上所述,对于失去根治性手术机会的晚期EHCC病人,SEMS-CL-125I可有效降低黄疸,改善肝功能。此外,与PS相比,SEMS-CL-125I显著缩短了住院时间,降低了住院费用,延长了生存时间,值得临床上进一步推广和应用。
胆道支架联合125I粒子置入与姑息性手术治疗晚期胆管癌的疗效比较
Comparison of therapeutic effect of biliary stent combined with 125I particle implantation and palliative surgery in the treatment of advanced cholangiocarcinoma
-
摘要:
目的比较自膨式金属支架联合导管载入式125I粒子(SEMS-CL-125I)与姑息性手术(PS)治疗晚期肝外胆管癌(EHCC)的疗效。 方法选择SEMS-CL-125I(SEMS-CL-125I组,n=67)或PS(PS组,n=34)治疗的晚期EHCC病人,比较2组术后并发症、住院时间、住院费用和总体生存率(OS)的差异。 结果与术前相比,2组病人术后1、3、6个月血清TBIL、ALT、AST水平均降低,而ALB均升高(P < 0.05)。SEMS-CL-125I组术后1个月ALB水平显著高于PS组(P < 0.05)。2组术后总体并发症差异无统计学意义(P>0.05)。与PS组相比,SEMS-CL-125I组病人的住院时间明显缩短,住院费用明显减少(P < 0.01),OS显著改善(P < 0.05)。术前TBIL、CA19-9、ALB和治疗方法是与术后OS相关的主要因素。 结论对于晚期EHCC病人,与PS相比,SEMS-CL-125I显著缩短了住院时间,降低了住院费用,延长了生存时间,值得临床推广和应用。 -
关键词:
- 肝外胆管癌 /
- 自膨式金属支架 /
- 导管载入式125I粒子 /
- 姑息性手术
Abstract:ObjectiveTo compare the efficacy of self-expanding metal stent combined with catheter loaded 125I particle (SEMS-CL-125I) and palliative surgery (PS) in the treatment of advanced extrahepatic cholangiocarcinoma (EHCC). MethodsPatients with advanced EHCC treated with SEMS-CL-125I (SEMS-CL-125I group, n=67) or PS (PS group, n 34) were selected.The postoperative complications, hospitalization time, hospitalization expenses and overall survival rate (OS) between the two groups were compared. ResultsCompared with that before operation, the levels of TBIL, ALT and AST in the two groups decreased at 1, 3 and 6 months after operation, while ALB increased (P < 0.05).The level of ALB in SEMS-CL-125I group was significantly higher than that in PS group at 1 month after operation (P < 0.05).There was no significant difference in postoperative complications between the two groups (P>0.05).Compared with PS group, The hospitalization time, hospitalization expenses and OS in SEMS-CL-125I group were significantly shorter than those in PS group (P < 0.01), and OS was significantly improved (P < 0.05).Preoperative TBIL, CA19-9, ALB and treatment method were the main factors related to postoperative OS. ConclusionsCompared with PS, SEMS-CL-125I significantly shortens the hospitalization time, reduces the hospitalization expenses and prolongs the survival time of patients with advanced EHCC, which is worthy of clinical promotion and application. -
表 1 基线资料比较
变量 总体 SEMS-CL-125I组
(n=67)PS组
(n=34)Z P 性别(男/女) 74/27 47/20 27/7 0.99* 0.05 年龄(≥70/ < 70)/岁 56/45 41/26 15/19 2.66* 0.05 肿瘤部位(pCCA/dCCA) 36/65 28/39 8/26 3.28* 0.05 CA19-9/ (ng/mL) 278.9(62.5~797.8) 293.0(72.5~989.8) 128.6(34.9~471.0) 1.61 0.05 CEA/(ng/mL) 4.2(2.6~6.4) 4.0(2.4~6.1) 4.3(2.8~6.7) 0.82 0.05 ALT/(U/L) 125(81~248) 131(92~239) 104(58~269) 1.00 0.05 AST/(U/L) 133(80.5~169) 137(97~191) 166(61~167) 1.72 0.05 TBIL/(μmol/L) 221(127~319) 244(150~343) 178(99~293) 1.67 0.05 DBIL/(μmol/L) 182.0(106.9~284.8) 194.8(121.7~298.8) 142.8(80.4~234.4) 1.74 0.05 ALB/(g/L) 34.2(31.4~37.7) 33.6(31.1~37.2) 35.1(32.9~39.9) 1.60 0.05 *示χ2值 表 2 2组术后1、3、6个月血清中ALT、AST、TBIL及ALB的变化情况
分组 n 术前 术后1月 术后3月 术后6月 P F MS组内 ALT/(U/L) SEMS-CL-125I组 67 169.16±106.71 46.93±29.67** 37.81±29.72** 41.75±37.47** < 0.01 74.50 3 638.650 PS组 34 163.12±140.72 56.06±35.62** 40.38±15.56** 48.20±28.44** < 0.01 20.50 5 530.460 t — 0.24 1.36 0.47 0.88 — — — P — 0.05 0.05 0.05 0.05 — — — AST/(U/L) SEMS-CL-125I组 67 152.76±91.04 54.72±30.87** 41.22±34.11** 44.70±45.01** < 0.01 61.10 3 107.660 PS组 34 120.41±65.09 51.48±29.97** 35.50±13.18** 47.80±24.75** < 0.01 33.80 1 480.300 t — 1.84 0.50 0.94 0.37 — — — P — 0.05 0.05 0.05 0.05 — — — TBIL/(U/L) SEMS-CL-125I组 67 250.01±132.09 39.82±32.79** 17.63±22.01** 17.53±10.93** < 0.01 179.0 0 4 781.710 PS组 34 212.42±144.91 54.25±48.47** 15.16±10.95** 23.29±16.76** < 0.01 48.80 5 937.260 t — 1.31 1.77 0.62 2.08 — — — P — 0.05 0.05 0.05 0.05 — — — ALB/(g/L) SEMS-CL-125I组 67 34.45±4.87 39.07±3.83** 40.72±3.39** 39.86±3.32** < 0.01 34.50 15.225 PS组 34 35.80±4.38 36.60±5.58 39.65±2.09** 39.28±2.66* < 0.01 41.90 15.441 t — 1.36 2.61 1.68 0.88 — — — P — 0.05 0.05 0.05 0.05 — — — 与同组术前比较*P<0.05,**P<0.01 表 3 影响病人术后OS的单因素分析
变量 OR(95%CI) P 性别(男/女) 1.040(0.654-1.652) >0.05 年龄(≥70/ < 0岁) 1.227(0.787-1.911) >0.05 肿瘤部位(pCCA/dCCA) 1.174(0.749-1.841) >0.05 CA19-9 (H vs L) 1.960(1.239-3.101) < 0.01 CEA (H vs L) 1.208(0.761-1.916) >0.05 ALT (H vs L) 1.027(0.655-1.610) >0.05 AST (H vs L) 1.145(0.689-1.901) >0.05 TBIL (H vs L) 1.762(1.124-2.762) < 0.05 ALB (L vs H) 1.757(1.111-2.779) < 0.05 治疗方法 1.640(1.030-2.611) < 0.05 (PS vs SEMS-CL-125I) -
[1] PATEL T.Increasing incidence and mortality of primary intrahepatic cholangiocarcinoma in the United States[J].Hepatology, 2001, 33(6):1353. doi: 10.1053/jhep.2001.25087 [2] XIANG S, LAU WY, CHEN XP.Hilar cholangiocarcinoma:controversies on the extent of surgical resection aiming at cure[J].Int J Colorectal Dis, 2015, 30(2):159. doi: 10.1007/s00384-014-2063-z [3] LENDVAI G, SZEKERCZES T, ILLYES I, et al.Cholangiocarcinoma:Classification, Histopathology and Molecular Carcinogenesis[J].Pathol Oncol Res, 2020, 26(1):3. doi: 10.1007/s12253-018-0491-8 [4] RIZVI S, KHAN SA, HALLEMEIER CL, et al.Cholangiocarcinoma-evolving concepts and therapeutic strategies[J].Nat Rev Clin Oncol, 2018, 15(2):95. doi: 10.1038/nrclinonc.2017.157 [5] PU LZ, SINGH R, LOONG CK, et al.Malignant Biliary Obstruction:Evidence for Best Practice[J].Gastroenterol Res Pract, 2016, 2016:3296801. [6] BLECHACZ B.Cholangiocarcinoma:current knowledge and new developments[J].Gut Liver, 2017, 11(1):13. doi: 10.5009/gnl15568 [7] KHAN SA, DAVIDSON BR, GOLDIN RD, et al.Guidelines for the diagnosis and treatment of cholangiocarcinoma:an update[J].Gut, 2012, 61(12):1657. doi: 10.1136/gutjnl-2011-301748 [8] LABIB PL, DAVIDSON BR, SHARMA RA, et al.Locoregional therapies in cholangiocarcinoma[J].Hepat Oncol, 2017, 4(4):99. doi: 10.2217/hep-2017-0014 [9] DELEON TT, AHN DH, BOGENBERGER JM, et al.Novel targeted therapy strategies for biliary tract cancers and hepatocellular carcinoma[J].Future Oncol, 2018, 14(6):553. doi: 10.2217/fon-2017-0451 [10] TANG Z, YANG Y, MENG W, et al.Best option for preoperative biliary drainage in Klatskin tumor:a systematic review and meta-analysis[J].Medicine (Baltimore), 2017, 96(43):e8372. doi: 10.1097/MD.0000000000008372 [11] WORLD MEDICAL A.World Medical Association Declaration of Helsinki:ethical principles for medical research involving human subjects[J].JAMA, 2013, 310(20):2191. doi: 10.1001/jama.2013.281053 [12] 胡小四, 庞青, 刘会春, 等.125I粒子腔内照射联合瘤体内粒子植入治疗局部进展期胰头癌[J].中国微创外科杂志, 2019, 19(9):794. [13] HU X, PANG Q, LIU H, et al.Inflammation-based prognostic scores in patients with extrahepatic bile duct lesions treated by percutaneous transhepatic biliary stenting combined with (125)I seeds intracavitary irradiation[J].Clin Transl Oncol, 2019, 21(5):665. doi: 10.1007/s12094-018-1969-2 [14] PANG Q, ZHOU L, HU XS, et al.Biliary stenting alone versus biliary stenting combined with (125)I particles intracavitary irradiation for the treatment of advanced cholangiocarcinoma[J].Sci Rep, 2019, 9(1):11348. doi: 10.1038/s41598-019-47791-4 [15] SQUADRONI M, TONDULLI L, GATTA G, et al.Cholangiocarcinoma[J].Crit Rev Oncol Hematol, 2017, 116:11. doi: 10.1016/j.critrevonc.2016.11.012 [16] KADABA RS, BOWERS KA, KHORSANDI S, et al.Complications of biliary-enteric anastomoses[J].Ann R Coll Surg Engl, 2017, 99(3):210. doi: 10.1308/rcsann.2016.0293 [17] SON RC, GWON DI, KO HK, et al.Percutaneous unilateral biliary metallic stent placement in patients with malignant obstruction of the biliary hila and contralateral portal vein steno-occlusion[J].Korean J Radiol, 2015, 16(3):586. doi: 10.3348/kjr.2015.16.3.586 [18] RAZUMILAVA N, GORES GJ.Classification, diagnosis, and management of cholangiocarcinoma[J].Clin Gastroenterol Hepatol, 2013, 11(1):13. [19] ZHU HD, GUO JH, HUANG M, et al.Irradiation stents vs.conventional metal stents for unresectable malignant biliary obstruction:a multicentre trial[J].J Hepatol, 2018, 68(5):970. doi: 10.1016/j.jhep.2017.12.028 [20] QIU H, JI J, SHAO Z, et al.The efficacy and safety of iodine-125 brachytherapy combined with chemotherapy in treatment of advanced lung cancer:a meta-analysis[J].J Coll Physicians Surg Pak, 2017, 27(4):237. [21] HASIMU A, GU JP, JI WZ, et al.Comparative study of percutaneous transhepatic biliary stent placement with or without Iodine-125 seeds for treating patients with malignant biliary obstruction[J].J Vasc Interv Radiol, 2017, 28(4):583. doi: 10.1016/j.jvir.2016.11.038 [22] XU X, LI J, WU J, et al.A systematic review and meta-analysis of intraluminal brachytherapy versus stent alone in the treatment of malignant wbstructive jaundice[J].Cardiovasc Intervent Radiol, 2018, 41(2):206. doi: 10.1007/s00270-017-1827-6 [23] LI M, LI K, QI X, et al.Percutaneous transhepatic biliary stent implantation for obstructive jaundice of perihilar cholangiocarcinoma:a prospective study on predictors of stent patency and survival in 92 patients[J].J Vasc Interv Radiol, 2016, 27(7):1047. doi: 10.1016/j.jvir.2016.02.035 [24] FORSMARK CE, DINIZ AL, ZHU AX.Consensus conference on hilar cholangiocarcinoma[J].HPB (Oxford), 2015, 17(8):666. doi: 10.1111/hpb.12451 [25] RAZUMILAVA N, GLEESON FC, GORES GJ.Awareness of tract seeding with endoscopic ultrasound tissue acquisition in perihilar cholangiocarcinoma[J].Am J Gastroenterol, 2015, 110(1):200. [26] NAVANEETHAN U, HASAN MK, LOURDUSAMY V, et al.Single-operator cholangioscopy and targeted biopsies in the diagnosis of indeterminate biliary strictures:a systematic review[J].Gastrointest Endosc, 2015, 82(4):608. doi: 10.1016/j.gie.2015.04.030 -





 下载:
下载: