-
血脂代谢异常在动脉粥样硬化中的重要作用已被证实。研究[1]表明,血脂水平异常升高促进脂质沉积,并在动脉粥样硬化的形成及粥样斑块发展进程具有重要作用。载脂蛋白E(apolipoprotein E,ApoE)缺陷小鼠是目前通过基因敲除手段研究血脂代谢紊乱形成动脉硬化动物模型,最新研究[2]发现糖尿病可进一步诱发ApoE-/-小鼠血脂代谢紊乱,加重动脉硬化发生。TR3受体作为孤核受体超家族的成员,介导了许多种激素、维生素和药物细胞生物学效应[3]。有研究[4]显示TR3受体激活后增加糖尿病病人对胰岛素敏感性,加速血糖代谢,降低血糖水平,减轻糖尿病糖代谢性疾病;也有研究发现TR3受体可降低细胞增殖核转录因子-κB(NF-κB)/细胞周期蛋白D1(cylinD1)蛋白表达,抑制细胞增殖;在动脉粥样硬化斑块中发现斑块组织NF-κB/cylinD1蛋白表达水平增高,同时动脉硬化斑块周围平滑肌层细胞增生明显,降低NF-κB p65/CylinD1表达,斑块周围增生细胞明显减少,减缓动脉粥样斑块的形成[5]。因此我们认为TR3受体对糖尿病ApoE-/-小鼠血脂代谢、细胞增殖调控NF-κB/CylinD1通路与动脉硬化斑块形成与发展有密切相关,本研究将观察TR3受体激动剂(6-mercaptopurine,6-MP)对糖尿病ApoE-/-小鼠糖脂代谢及NF-κB p65/CylinD1通路的影响,探讨TR3受体激动剂改善糖尿病动脉粥样硬化可能机制。
-
ApoE-/-小鼠(SPF级)购于北京大学医学部,链脲佐菌素(streptozotocin,STZ)、6-MP(Sigma公司),β肌动蛋白(β-actin)单克隆抗体、NF-κB p65/CylinD1一抗(CST公司),HRP标记二抗(北京中杉公司),丙烯酰胺、Tris-base(Ameresco),PVDF膜(millipore公司),免疫印迹化学发光试剂(Santa Cruz公司)。
-
8周龄SPF级雄性ApoE-/-小鼠,体质量(23±2)g,饲养条件,室温(22±2)℃,湿度(55±5)%,人工光照明暗各12 h/d,24 h自由取食饮水,饲料由上海斯莱克公司提供。随机选取10只ApoE-/-小鼠为对照组(ApoE-/-组),糖尿病小鼠30只,随机抽样法分为3组(每组10只)。实验动物共分为4组:ApoE-/-组(n=10),STZ+ApoE-/-组(n=10),STZ+ApoE-/-+6-MP 2.5 mg·kg-1·d-1组(n=10),STZ+ApoE-/-+6-MP 5 mg·kg-1·d-1(n=10)。将6-MP溶于0.9%氯化钠溶液中,腹腔注射法给药,1次/天,连续8周,其余各组动物均用等量0.9%氯化钠溶液腹腔注射,1次/天。
-
STZ配制:柠檬酸缓冲液(0.01 mol/L,pH 4.4)10 mL加100 mg STZ充分溶解(STZ浓度:10 mg/mL),所有动物禁食12 h,于腹腔注射STZ:柠檬酸溶液0.01 mL/g,给药后立即给予饲料自由取食,3 d后测定小鼠空腹血糖,动物血糖水平于12~25 mmol/L用于本试验。
-
取小鼠胸主动脉弓下0.5 cm,PBS漂洗2~3次,10%甲醛固定24 h,常规脱水、透明、石蜡包埋、切片、HE染色,中性树脂封片,胸主动脉内膜斑块面积采用自动成像分析软件系统(Olympus, Japan)分析,求斑块面积平均值。
-
小鼠胸主动脉总蛋白提取:取小鼠胸主动脉弓约1 cm,去除血管外膜结缔组织,0.9%氯化钠溶液漂洗1~2次,加0.5 mL RIPA裂解缓冲液[含0.1 mol/L NaCl,0.01 mol/L Tris.Cl pH7.6,0.001 mol/L EDTA,100 mmol/L DTT,1 μg/mL抑肽酶(1:100),100 μg/mL苯甲基磺酰氟(PMSF)]匀浆,加入0.5 mL 2×SDS上样缓冲液,剧烈震荡15 s, -80 ℃冰箱冻融2次,100 ℃水浴10 min,12 000 g,4 ℃离心,取上清,凝胶电泳、转膜、封闭、一抗孵育、二抗孵育、显影等步骤。
-
采用方差分析和q检验。
-
6-MP对糖尿病ApoE-/-小鼠血糖的影响与ApoE-/-组相比,STZ-ApoE-/-组小鼠空腹血糖水平显著升高(P<0.01);TR3受体激动剂6-MP可呈剂量依赖性降低糖尿病ApoE-/-小鼠空腹血糖水平(P<0.01),与STZ-ApoE-/-组相比,STZ-ApoE-/-+6-MP5组小鼠空腹血糖水平显著降低(P<0.01),STZ-ApoE-/-+6-MP 10组空腹血糖水平进一步降低(P<0.01)(见表 1)。
分组 n 血糖/(mmol/L) ApoE-/-组 10 6.91±0.69 STZ-ApoE-/-组 10 18.68±2.30** STZ-ApoE-/-+6-MP5组 10 15.42±2.68**# STZ-ApoE-/-+6-MP10组 10 13.64±1.95**## F — 40.07 P — <0.01 MS组内 — 3.090 q检验:与ApoE-/-组比较**P<0.01;与STZ-ApoE组比较#P<0.05, ##P<0.01 表 1 TR3受体激动剂6-MP对糖尿病ApoE-/-小鼠血糖的影响(x±s)
-
与ApoE-/-组相比,STZ-ApoE-/-组小鼠空腹总胆固醇(TC)和低密度脂蛋白胆固醇(LDL-C)显著升高(P<0.01),2组间高密度脂蛋白胆固醇(HDL-C)和三酰甘油(TG)差异无统计学意义(P>0.05),TR3受体激动剂6-MP呈剂量依赖性降低糖尿病ApoE-/-组小鼠空腹TC、LDL-C、TG水平;与STZ-ApoE-/-组相比,STZ-ApoE-/-+6-MP 5组小鼠空腹血脂TC、LDL-C、TG水平均显著降低(P<0.01),STZ-ApoE-/-+6-MP10组空腹血脂TC、LDL-C、TG进一步降低(P<0.01);STZ-ApoE-/-组、STZ-ApoE-/-+6-MP5及STZ-ApoE-/-+6-MP10三组HDL-C差异无统计学意义(P>0.05)(见表 2)。
分组 TG TC HDL-C LDL-C ApoE-/-组 5.28±0.52 41.40±3.31 14.04±2.21 32.92±3.83 STZ-ApoE-/-组 5.60±0.43 62.04±7.12** 16.20± 4.83 50.96±6.83** STZ-ApoE-/-+6-MP5组 5.05±1.34 47.70±6.46# 15.95±0.98 42.50±4.92**## STZ-ApoE-/-+6-MP10组 4.57±1.45 40.82±5.19## 16.18±1.76 38.45±5.11**## F 1.73 29.91 1.34 20.67 P >0.05 <0.01 >0.05 <0.01 MS组内 1.088 32.580 8.068 27.909 q检验:与ApoE-/-组比较*P<0.05,**P<0.01;与STZ-ApoE-/-组比较#P<0.05,##P<0.01 表 2 TR3受体激动剂6-MP对糖尿病ApoE-/-小鼠血糖和血脂的影响(ni=10;x±s;mmol/L)
-
与C57组相比,ApoE-/-组小鼠肝脏FGF21 mRNA及蛋白表达水平均差异无统计学意义(P>0.05);与ApoE-/-组相比,STZ-ApoE-/-组小鼠肝脏FGF21 mRNA、蛋白及血清FGF21水平均明显增高(P<0.01);FGF21 miRNA可显著降低糖尿病ApoE-/-小鼠肝脏FGF21 mRNA、蛋白及血清FGF21水平(P<0.05)(见图 1、表 3)。
分组 n NF-κB p65 Cylin D1 ApoE-/-组 10 0.053±0.008 0.234±0.026 STZ-ApoE-/-组 10 0.358±0.046** 0.617±0.107** STZ-ApoE-/-+6-MP5组 10 0.213±0.025**## 0.226±0.031## STZ-ApoE-/-+6-MP10组 10 0.022±0.003**## 0.109±0.013**## F — 343.51 147.62 P — <0.01 <0.01 MS组内 — 0.001 0.003 注:蛋白印迹电泳胶片条带光密度(OD值)分析,NF-κB p65或CylinD1/β-actin光密度比值表示目的蛋白表达水平。q检验:与ApoE-/-组比较*P<0.05,**P<0.01;与STZ-ApoE-/-组比较#P<0.05,##P<0.01 表 3 TR3受体激动剂6-MP对糖尿病ApoE-/-小鼠胸主动脉NF-κB p65/CylinD1蛋白表达的影响(x±s)
-
6-MP对糖尿病ApoE-/-小鼠胸主动脉内膜脂质沉积的影响ApoE-/-组胸主动脉血管内膜有少量小团块状红染脂质,STZ-ApoE-/-组胸主动脉血管内膜有片状团块状红染脂质;STZ-ApoE-/-+6-MP5组或STZ-ApoE-/-+6-MP10组胸主动脉血管内膜少量小片状红染脂质(见图 2)。
-
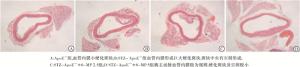
6-MP对糖尿病ApoE-/-小鼠胸主动脉内膜粥样斑块形成的影响ApoE-/-组主动脉血管内膜可见小硬化斑块,斑块底部血管增厚;STZ-ApoE-/-组胸主动脉内膜可见巨大斑块突起,斑块中央有空洞形成,斑块底部血管增厚且不规则,斑块空腔周围可见大量细胞增生;STZ-ApoE-/-+6-MP5组主动脉血管内膜硬化斑块斑块较小,斑块中央空洞较小,斑块底部及周围有少量增生组织;STZ-ApoE-/-+6-MP5组胸主动脉内膜只见小硬化斑块隆起,斑块底部及周围有少量增生组织(见图 3)。
斑块面积分析:与ApoE-/-组相比,STZ-ApoE-/-组胸主动脉血管内膜斑块面积明显增加(P<0.05);TR3受体激动剂6-MP可呈剂量依赖的方式抑制STZ诱导的糖尿病ApoE-/-小鼠胸主动脉硬化斑块增加,与STZ-ApoE-/-组相比STZ-ApoE-/-+6-MP 5组胸主动脉内膜斑块面积明显降低(P<0.05);STZ-ApoE-/-+6-MP10组与STZ-ApoE-/-+6-MP 5组之间斑块面积差异有统计学意义(P<0.05)(见表 4)。
分组 n 斑块面积/μm2 ApoE-/-组 10 123.56±15.38 STZ-ApoE-/-组 10 245.16±30.29** STZ-ApoE-/-+6-MP5组 10 172.43±20.55**## STZ-ApoE-/-+6-MP10组 10 154.09±18.73**## F — 55.40 P — <0.01 MS组内 — 481.786 q检验:与ApoE-/-组比较*P<0.05,**P<0.01;STZ-ApoE-/-组比较#P<0.05,##P<0.01 表 4 TR3受体激动剂6-MP对糖尿病ApoE-/-小鼠胸主动脉内膜内膜粥样斑块面积的影响(x±s)
-
本研究发现,链尿左菌素诱导ApoE-/-小鼠血糖水平升高,血脂代谢进一步异常,胸主动脉内膜脂质沉积与斑块面积也显著增加,说明糖尿病可导致血脂紊乱,加速ApoE-/-小鼠动脉粥样硬化进展。即往研究已证实糖尿病是冠心病重要危险因素,其对动脉粥样硬化的发生、发展及预后均起着至关重要的作用[6-7];本研究又观察到用TR3受体激动剂6-MP治疗的STZ诱导糖尿病ApoE-/-小鼠可降低血糖水平,改善血脂代谢,减少胸主动脉内膜脂质沉积及斑块面积,说明TR3受体激动剂6-MP可改善糖尿病ApoE-/-小鼠糖脂代谢,减缓糖尿病诱导动脉粥样硬化发展。在TR3受体激动剂6-MP抑制平滑肌细胞增殖的研究中已证实6-MP可以激活TR3进而抑制静脉平滑肌细胞的过度增殖[8]。最近有相关研究又发现TR3核受体可与肝核受体LXR结合,促进肝细胞对糖的摄取,降低血糖水平[9-10]。也有研究[11]认为TR3核受体激活血脂的改善是由于血糖水平降低引起的,更有研究[12]认为TR3核受体可直接促进肝及脂肪组织脂质转化,减少糖尿病引起脂肪代谢紊乱。因此TR3核受体激激动剂被认为治疗高血糖性代谢疾病药物。我们研究还发现糖尿病ApoE-/-小鼠胸主动脉细胞周期调控蛋白NF-κBp65及Cylin D1表达水平显著增加,胸主动脉斑块周围大量增生组织,说明糖尿病不但可诱发脂质斑块形成,同时可促进斑块周围组织细胞增生,并与糖尿病ApoE-/-小鼠NF-κB p65,Cylin D1高表达有关。相关动脉硬化的研究也证实动脉斑块周围组织增生与NF-κB p65,Cylin D1水平密切相关,认为巨噬细胞侵入动脉中膜形成泡沫细胞过程增加平滑肌层细胞NF-κB p65,Cylin D1表达水平,促进平滑肌细胞增殖,加速斑块形成[13]。有研究认为NF-κB p65,Cylin D1增加导致平滑细胞增生具有稳定斑块作用[14];也有研究认为NF-κB p65,Cylin D1也可促进斑块底部平滑肌层增生可增斑块破裂的风险[15]。本研究用TR3受体激动剂6-MP治疗后发现6-MP可呈剂量依赖性抑制糖尿病诱导胸主动脉NF-κB p65,Cylin D1水平增加,同时斑块周围细胞增生层变薄,斑块底增生组织减少,斑块面积降低,说明TR3受体激动后可降低糖尿病诱导胸主动脉NF-κB p65、Cylin D1,减少斑块周围细胞增生,且这种作用可能是TR3受体通过调控NF-κB p65、Cylin D1表达抑制动脉斑块的形成,减少动脉硬化的发生。即往大量研究认为动脉硬化的发生发展与平滑肌层泡沫细胞形成过程平滑肌细胞增生起至关重要作用[16]。近年来,有学者通过相关研究认为在早期动脉粥样硬化抑制平滑细胞增生可阻止或延缓冠心病的发展[17]。有研究体外细胞试验也发现抑制平滑肌细胞NF-κB p65,Cylin D1可降低平滑肌细胞增殖迁移[18]。
综上所述,本研究结果发现TR3受体激动剂6-MP抗动脉粥样硬化作用可能是通过降低糖尿病ApoE-/-小鼠血糖水平,改善血脂代谢,降低胸主动脉NF-κB p65、Cylin D1表达。
TR3受体激动剂6-mercaptopurine对糖尿病ApoE-/-小鼠NF-κB p65/CylinD1通路影响及其与抗动脉硬化关系
Effect of TR3 receptor agonist 6-mercaptopurine on NF-κB p65/CylinD1 pathway in diabetic ApoE-/- mice, and its relationship with anti-arteriosclerosis
-
摘要:
目的观察TR3受体激动剂(6-mercaptopurine,6-MP)对链脲佐菌素(streptozotocin,STZ)诱导的载脂蛋白E(apolipoprotein E,ApoE-/-)敲除糖尿病小鼠糖脂代谢及NF-κB p65/CylinD1通路的影响及其与抗动脉硬化的关系,探讨TR3受体在改善糖尿病动脉粥样硬化可能机制。 方法40只雄性ApoE-/-小鼠随机分为4组:ApoE-/-组、STZ-ApoE-/-组、STZ-ApoE-/-+6-MP5(5 mg·kg-1·d-1)组、STZ-ApoE-/-+6-MP10(10 mg·kg-1·d-1)组,每组10只;6-MP腹腔注射法给药,每天1次,连续8周;STZ 60 mg/kg腹腔注射ApoE-/-小鼠建立糖尿病动脉粥样硬化模型,血糖试纸法测血糖水平;酶法或匀相法测血脂水平;蛋白免疫印迹试验(Western-blotting)测胸主动脉组织NF-κB p65/CylinD1蛋白水平;油红O染色观察胸主动脉内膜动脉脂质沉积;HE染色测胸主动脉内膜粥样斑块面积。 结果与ApoE-/-组相比,STZ-ApoE-/-组血糖及血清TG、TCHO、LDL-C均显著增高(P < 0.05);TR3受体激动剂6-MP呈剂量依赖性降低糖尿病ApoE-/-小鼠血糖、血脂水平,与STZ-ApoE-/-组相比,STZ-ApoE-/-+6-MP5组血糖、TG、LDL-C、TC均显著降低(P < 0.05),与ApoE-/-组相比,STZ-ApoE-/-组胸主动脉NF-κBp65、CylinD1蛋白表达水平均显著增加(P < 0.05);6-MP呈剂量依赖性降低糖尿病ApoE-/-小鼠胸主动脉NF-κBp65/CylinD1蛋白表达,STZ-ApoE-/-+6-MP5组NF-κB p65/CylinD1蛋白表达水平均明显低于STZ-ApoE-/-组(P < 0.05);STZ-ApoE-/-+6-MP10组与STZ-ApoE-/-+6-MP5组之间胸主动脉NF-κB p65/CylinD1蛋白表达水平差异均有统计学意义(P < 0.05)。油红O染色显示:TR3受体激动剂6-MP可呈剂量依赖性抑制STZ-ApoE-/-小鼠胸主动脉内膜脂质沉积;HE染色显示:TR3受体激动剂6-MP可呈剂量依赖性降低STZ-ApoE-/-小鼠胸主动脉内膜斑块面积,减少斑块内部有空洞(脂质)形成,抑制斑块周围组织及斑块基底部平滑肌层增生,与STZ-ApoE-/-组相比,STZ-ApoE-/-+6-MP5组胸主动脉内膜斑块面积明显减小(P < 0.05),STZ-ApoE-/-6-MP10组斑块面积进一步减小(P < 0.01)。 结论TR3激动剂6-MP可明显降低糖尿病ApoE-/-小鼠动脉粥样硬化的形成,其机制可能与其改善糖脂代谢,抑制NF-κB p65/CylinD1信号通路有关。 Abstract:ObjectiveTo observe the effects of TR3 receptor agonist 6-mercaptopurine(6-MP) on the glucose and lipid metabolism and NF-κB p65/CylinD1 pathway in streptozotocin (STZ)-induced diabetic apolipoprotein E knockout(ApoE-/-) mice, and explore the possible mechanism of TR3 receptors in improving diabetic atherosclerosis. MethodsForty ApoE-/- mice were randomly divided into the ApoE-/- group, STZ-ApoE-/- group, STZ-ApoE-/- +6-MP5(5 mg·kg-1·d-1) and STZ-ApoE-/- +6-MP10(10 mg·kg-1·d-1) group(10 rats each group).The 6-MP intraperitoneal injection was implemented, once a day, for 8 weeks.The diabetic animal model was established using intraperitoneal injection of STZ.The levels of blood glucose and blood lipid were determined using the blood glucose test strip and the enzymic method or selective homogeneous method, respectively.The Western-blotting was used to detect the protein levels of NF-κB p65/CylinD1 in thoracic aorta tissue, the lipid deposition in the thoracic aortic intima was observed by oil red O staining, and the patch area in the thoracic aorta was measured using HE staining. ResultsCompared with the ApoE-/- mice group, the levels of blood glucose, triglyceride(TG), total cholesterol(TCHO) and low-density lipoprotein-cholesterol(LDL-C) significantly increased in STZ-ApoE-/- group(P < 0.05), and the level of TR3 receptor agonist 6-MP decreased the levels of plasma glucose and lipid with dose-dependently.Comapred the STZ-ApoE-/- group, the levels of the blood glucose and TG, LDL-A and TC significantly decreased in the STZ-ApoE-/- +6-MP5 group(P < 0.05).Compared with the ApoE-/- group, the levels of NF-κB p65 and CylinD1 remarkably increased in the STZ-ApoE-/- group(P < 0.05).The 6-MP reduced the expression level of NF-κB p65/CylinD1 protein in thoracic aorta of diabetic ApoE-/- mice in a dose-dependent manner, and the protein level of NF-κB p65/CylinD1 in STZ-ApoE-/- +6-MP5 group were significantly lower than that in STZ-ApoE-/- group(P < 0.05).The differences of the levels of NF-κB p65/CylinD1 in thoracic aorta between STZ-ApoE-/- +6-MP10 group and STZ-ApoE-/- +6-MP5 group were statistically significnat(P < 0.05).The results of oil red O staining showed that 6-MP inhibited the intima lipid deposition in the thoracic aorta of STZ-ApoE-/- mice in a dose-dependent manner.The results of HE staining showed that 6-MP reduced the area of intima plaque and formation of vacuoles(lipids) inside plaque, and inhibited the proliferation of smooth muscle layer around plaque and plaque base in the thoracic aorta of STZ-ApoE-/- mice in a dose-dependent manner.Compared with the STZ-ApoE-/- group, the plaque area in the STZ-ApoE-/- +6-MP5 group significantly reduced(P < 0.05), and further reduced in the STZ-ApoE-/- 6-MP10 group(P < 0.01). ConclusionsThe TR3 receptor agonist 6-MP can significantly decrease the formation of atherosclerosis in diabetic ApoE-/- mice, which may be related to the improvement of glycolipid metabolism and inhibition of NF-κB p65/CylinD1 signaling pathway. -
Key words:
- diabetes mellitus /
- ApoE-/- mice /
- 6-mercaptopurine /
- atherosclerosis /
- blood lipid /
- TR3 /
- NF-κB p65/CylinD1
-
表 1 TR3受体激动剂6-MP对糖尿病ApoE-/-小鼠血糖的影响(x±s)
分组 n 血糖/(mmol/L) ApoE-/-组 10 6.91±0.69 STZ-ApoE-/-组 10 18.68±2.30** STZ-ApoE-/-+6-MP5组 10 15.42±2.68**# STZ-ApoE-/-+6-MP10组 10 13.64±1.95**## F — 40.07 P — <0.01 MS组内 — 3.090 q检验:与ApoE-/-组比较**P<0.01;与STZ-ApoE组比较#P<0.05, ##P<0.01 表 2 TR3受体激动剂6-MP对糖尿病ApoE-/-小鼠血糖和血脂的影响(ni=10;x±s;mmol/L)
分组 TG TC HDL-C LDL-C ApoE-/-组 5.28±0.52 41.40±3.31 14.04±2.21 32.92±3.83 STZ-ApoE-/-组 5.60±0.43 62.04±7.12** 16.20± 4.83 50.96±6.83** STZ-ApoE-/-+6-MP5组 5.05±1.34 47.70±6.46# 15.95±0.98 42.50±4.92**## STZ-ApoE-/-+6-MP10组 4.57±1.45 40.82±5.19## 16.18±1.76 38.45±5.11**## F 1.73 29.91 1.34 20.67 P >0.05 <0.01 >0.05 <0.01 MS组内 1.088 32.580 8.068 27.909 q检验:与ApoE-/-组比较*P<0.05,**P<0.01;与STZ-ApoE-/-组比较#P<0.05,##P<0.01 表 3 TR3受体激动剂6-MP对糖尿病ApoE-/-小鼠胸主动脉NF-κB p65/CylinD1蛋白表达的影响(x±s)
分组 n NF-κB p65 Cylin D1 ApoE-/-组 10 0.053±0.008 0.234±0.026 STZ-ApoE-/-组 10 0.358±0.046** 0.617±0.107** STZ-ApoE-/-+6-MP5组 10 0.213±0.025**## 0.226±0.031## STZ-ApoE-/-+6-MP10组 10 0.022±0.003**## 0.109±0.013**## F — 343.51 147.62 P — <0.01 <0.01 MS组内 — 0.001 0.003 注:蛋白印迹电泳胶片条带光密度(OD值)分析,NF-κB p65或CylinD1/β-actin光密度比值表示目的蛋白表达水平。q检验:与ApoE-/-组比较*P<0.05,**P<0.01;与STZ-ApoE-/-组比较#P<0.05,##P<0.01 表 4 TR3受体激动剂6-MP对糖尿病ApoE-/-小鼠胸主动脉内膜内膜粥样斑块面积的影响(x±s)
分组 n 斑块面积/μm2 ApoE-/-组 10 123.56±15.38 STZ-ApoE-/-组 10 245.16±30.29** STZ-ApoE-/-+6-MP5组 10 172.43±20.55**## STZ-ApoE-/-+6-MP10组 10 154.09±18.73**## F — 55.40 P — <0.01 MS组内 — 481.786 q检验:与ApoE-/-组比较*P<0.05,**P<0.01;STZ-ApoE-/-组比较#P<0.05,##P<0.01 -
[1] 赵冬.中国人群的血脂流行病学研究[J].临床荟萃, 2006, 21(8):533. doi: 10.3969/j.issn.1004-583X.2006.08.001 [2] JAISWAL M, SCHINSKE A, POP-BUSUI R.Lipids and lipid management in diabetes[J].Best Pract Res Clin Endocrinol Metab, 2014, 28(3):325. doi: 10.1016/j.beem.2013.12.001 [3] LI Y, BOURBON PM, GRANT MA, et al.Requirement of novel amino acid fragments of orphan nuclear receptor TR3/Nur77 for its functions in angiogenesis[J].Oncotarget, 2015, 6(27):24261. [4] HARANT H.Negative cross-talk between the human orphan nuclear receptor Nur77/NAK-1/TR3 and nuclear factor-κB[J].Nucleic Acids Res, 2004, 32(17):5280. doi: 10.1093/nar/gkh856 [5] WU L, CHEN L.Characteristics of Nur77 and its ligands as potential anticancer compounds(Review)[J].Mol Med Rep, 2018, 18(6):4793. [6] FEODOROFF M, HARJUTSALO V, FORSBLOM C, et al.Dose-dependent effect of smoking on risk of coronary heart disease, heart failure and stroke in individuals with type 1 diabetes[J].Diabetologia, 2018, 61(12):2580. doi: 10.1007/s00125-018-4725-9 [7] NEWMAN JD, ROCKMAN CB, KOSIBOROD M, et al.Diabetes mellitus is a coronary heart disease risk equivalent for peripheral vascular disease[J].Am Heart J, 2017, 184:114. doi: 10.1016/j.ahj.2016.09.002 [8] WAARD VD, ARKENBOUT EK, VOS M, et al.TR3 nuclear orphan receptor prevents cyclic stretch-induced proliferation of venous smooth muscle cells[J].Am J Pathol, 2006, 168(6):2027. doi: 10.2353/ajpath.2006.050932 [9] ZHAO Y, BRUEMMER D.NR4A orphan nuclear receptors:transcriptional regulators of gene expression in metabolism and vascular biology[J].Arterioscler Thromb Vasc Biol, 2010, 30(8):1535. doi: 10.1161/ATVBAHA.109.191163 [10] MIN AK, BAE KH, JUNG YA, et al.Orphan nuclear receptor Nur77 mediates fasting-induced hepatic fibroblast growth factor 21 expression[J].Endocrinology, 2014, 155(8):2924. doi: 10.1210/en.2013-1758 [11] CHAO LC, WROBLEWSKI K, ILKAYEVA OR, et al.Skeletal muscle Nur77 expression enhances oxidative metabolism and substrate utilization[J].J Lipid Res, 2012, 53(12):2610. doi: 10.1194/jlr.M029355 [12] DE VERA IM, GIRI PK, MUNOZ-TELLO P, et al.Identification of a Binding Site for Unsaturated Fatty Acids in the Orphan Nuclear Receptor Nurr1[J].ACS Chem Biol, 2016, 11(7):1795. doi: 10.1021/acschembio.6b00037 [13] FENG M, KONG SZ, WANG ZX, et al.The protective effect of coptisine on experimental atherosclerosis ApoE-/- mice is mediated by MAPK/NF-κB-dependent pathway[J].Biomed Pharmacother, 2017, 93:721. doi: 10.1016/j.biopha.2017.07.002 [14] AL-SHOBAILI HA, FARHAN J, ZAFAR U, et al.Functional role of human interleukin-32 and nuclear transcription factor-kB in patients with psoriasis and psoriatic arthritis[J].Int J Health Sci, 2018, 12(3):29. [15] EID AH, GAD AM, FIKRY EM, et al.Venlafaxine and carvedilol ameliorate testicular impairment and disrupted spermatogenesis in rheumatoid arthritis by targeting AMPK/ERK and PI3K/AKT/mTOR pathways[J].Toxicol Appl Pharmacol, 2019, 364:83. doi: 10.1016/j.taap.2018.12.014 [16] GUILLERMIER C, DOHERTY SP, WHITNEY AG, et al.Imaging mass spectrometry reveals heterogeneity of proliferation and metabolism in atherosclerosis[J].JCI Insight, 2019, 4(11):12858. [17] 袁帅, 廖思聪, 王大新.血管平滑肌细胞与动脉粥样硬化关系的研究进展[J].实用心脑肺血管病杂志, 2018(2):6. doi: 10.3969/j.issn.1008-5971.2018.02.002 [18] 刘宇宏, 王莎莎, 沈凌汛, 等.IL-32γ对大鼠血管平滑肌细胞增殖与细胞周期的影响[J].中国病理生理杂志, 2012, 28(2):239. doi: 10.3969/j.issn.1000-4718.2012.02.009 -





 下载:
下载: