-
原发性肝癌是一种高度恶性肿瘤,在引起肿瘤相关性死因中居第二位,大多数肝癌病人就诊时已处于进展阶段,无法接受根治性治疗,预后差,复发率和病死率高[1-2]。肝细胞癌(hepatocellular carcinoma, HCC)约占原发性肝癌中的90%[2]。欧洲肝脏病学会和美国肝脏病学会推荐的治疗HCC的主要治疗手段包括手术切除、经动脉化疗栓塞术、肝移植及局部消融等[3]。尽管近年来这些手段有了一定的发展,但由于肝癌转移和复发率较高,预后仍较差。所以,迫切需要寻找新的治疗方法。
近年来,伴随着免疫学、分子生物学、生物工程的发展,肿瘤免疫治疗已成为最有潜力的治疗手段之一。肿瘤中浸润的巨噬细胞称肿瘤相关巨噬细胞(tumor associated macrophages,TAM),为一类重要的免疫细胞。TAM被认为主要是M2型巨噬细胞[4],主要作用是免疫抑制,促进血管生成,分泌多种促进肿瘤生长的因子等。核因子κB(nuclear factor κB,NF-κB)是一种细胞核转录因子,广泛存在于真核细胞中。NF-κB参与了机体炎症反应、免疫应答及细胞凋亡等,可以通过调节细胞因子等,促进肿瘤血管生成、浸润和转移。NF-κB p50是其重要家庭成员之一,对于NF-κB蛋白在肿瘤中扮演的角色起到重要作用。但NF-κB与M2巨噬细胞的关系尚不明确。本文研究了肝癌中M2巨噬细胞及NF-κB p50的表达情况,探讨它们在不同分期肝癌中表达的相关性。
-
本研究整群收集2012-2015年在安徽医科大学第一附属医院初次诊断并接受手术的80例HCC病人和10例肝内胆管结石病人(对照组)的石蜡标本;2015年10月至2016年1月在安徽医科大学第一附属医院肝胆外科手术切除的HCC病人肝癌新鲜组织20例及癌旁新鲜组织6例(对照组)。取分界线以外相应一侧2 cm的组织为癌旁组织。全组病例需符合的条件有:(1)手术前未接受过化疗或放疗;(2)有TNM分期等资料;(3)标本组织学经安徽医科大学第一附属医院病理科专家再次确诊为HCC。
HCC病人肝组织石蜡切片中临床分期Ⅰ+Ⅱ期病人34例(42.5%),Ⅲ+Ⅳ期46例(57.5%);HCC病人新鲜肝组织标本中临床分期Ⅰ+Ⅱ期病人12例(60%),Ⅲ+Ⅳ期病人8例(40%)。分期标准按照美国肿瘤联合会(American Joint Committee on Cancer, AJCC)2010年对原发性肝癌分期(第六版)制定的TNM分期。
-
CD163、CD206抗体(美国Santa Cruz公司);AlexaFluor594、AlexaFluor488荧光二抗(美国ThermoFisher Scitific公司);无水乙醇、甲醇、二甲苯(上海中试化总公司);Triton X-100和DAPI(上海碧云天生物技术公司);山羊血清(美国Gibco公司);分离胶缓冲液、浓缩胶缓冲液、Western一抗、二抗稀释液(江苏碧云天公司);BCA蛋白定量试剂盒(美国Thermo)。
石蜡切片机、激光共聚焦显微镜[德国莱卡(Leica)仪器有限公司];电泳仪(美国BIO-RAD公司);4 ℃冰箱(海尔);高速台式冷冻离心机(美国Sigma公司)。
-
HCC组织经甲醛固定,梯度乙醇脱水后,将组织和蜡块置于包埋框内,冷却后即成蜡块。进行防脱处理后,Leica切片机切成厚4~6 μm薄片,将蜡片裱贴于载玻片上,烤干待用。梯度乙醇脱蜡后,用Triton浸润切片。用枸橼酸盐修复抗原后,滴加3%过氧化氢阻断内源性过氧化酶。玻片上滴加山羊血清室温下孵育20 min,PBS洗3次,每次3 min。CD163和CD206均以1: 50稀释,混合后滴加在切片组织上,放置于湿盒中,4 ℃孵育过夜后,将湿盒从冰箱取出,复温,PBS洗3次,每次3 min。在切片组织上滴加羊抗兔或羊抗鼠(稀释度1: 100)荧光二抗,37 ℃避光孵1 h;PBS洗3次,每次3 min。在切片组织上滴加DAPI染色,室温避光孵育10 min,PBS洗3次,每次3 min。滴加抗荧光淬灭剂后,将盖玻片覆盖在组织上,固定后,显微镜下观察并拍照。
-
将RIPA和PMSF按100: 1配制裂解液。从-80 ℃冰箱取出冻存肝脏组织,每例剪取约50 mg,用PBS冲洗,放入匀浆器中,按10 μL/mg组织加入裂解液。冰上研磨10 min,4 ℃、14 000 r/min离心20 min,吸取上清,即为总蛋白。
-
配制10%的SDS-聚丙烯酰胺分离胶, 进行上样、电泳、转膜等操作,用5%脱脂奶粉室温下封闭2 h。然后加入一抗4 ℃孵育过夜。TBST洗3次,每次5 min。加入二抗室温孵育2 h,再用TBST洗3次,每次5 min后,ECL试剂盒显影,Image-J软件分析图片。以β-actin为内参,结果以目的条带与β-actin条带灰度值的比值作为目的蛋白的相对表达量。
-
采用方差分析、q检验和直线相关分析。
-
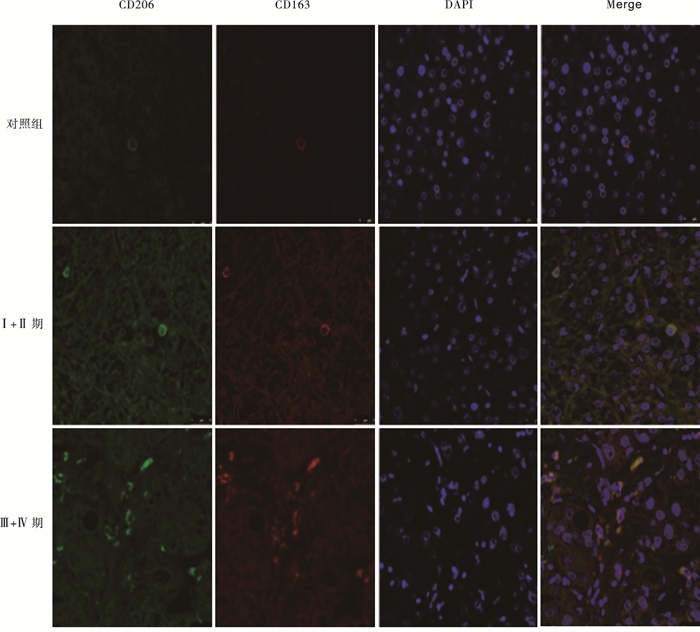
绿色荧光标记CD206蛋白,红色荧光标记CD163蛋白,DAPI染细胞核,Merge可显示绿色和红色荧光共定位情况,两种荧光重合的细胞表示该细胞存在CD206和CD163的共定位。免疫荧光双染结果显示,对照组中CD163和CD206的共表达较少,且荧光强度较弱,而TNMⅠ+Ⅱ期病人肝组织切片中CD163和CD206的共表达增多,荧光强度增加。随着肝癌进展,TNM Ⅲ+Ⅳ期中CD163和CD206的共表达明显比TNMⅠ+Ⅱ期病人增多,且荧光强度明显增强(见图 1)。
-
Western blotting结果显示,CD163和CD206在肝癌病人肝组织中表达上升, Ⅲ+Ⅳ期病人上升较Ⅰ+Ⅱ期病人更为明显(P<0.05~P<0.01)(见图 2、表 1)。
分组 CD163 CD206 p50 对照组 0.32±0.05 0.34±0.05 0.31±0.04 Ⅰ+Ⅱ期 0.48±0.07** 0.62±0.04* 0.85±0.04** Ⅲ+Ⅳ期 0.81±0.08**△△ 0.88±0.07**△△ 1.79±0.04**△△ F 40.044 66.482 991.034 P <0.01 <0.01 <0.01 MS组内 0.005 0.003 0.003 注:相对灰度值=目的蛋白灰度值/β-actin灰度值; q检验:与control组比较*P<0.05, **P<0.01;与Ⅰ+Ⅱ期组比较△△P<0.01 表 1 不同分期肝癌病人新鲜肝脏组织中CD163、CD206、p50表达相对灰度值的比较(x±s; ni=3)
-
Western blotting结果显示,NF-κB p50的表达在肝癌病人肝组织中上升, Ⅲ+Ⅳ期病人上升较Ⅰ+Ⅱ期病人更为明显(P<0.01)(见图 3、表 1)。
-
相关性分析发现,肝癌病人肝脏组织中NF-κB p50与CD163、CD206的表达在各组中呈现明显正相关(r=0.963, P<0.01;r=0.944, P<0.01)(见图 4)。
-
HCC为消化系统常见肿瘤之一,病因尚不完全明确,且缺乏有效的治疗手段[5-7]。HCC是多步骤、多阶段发生的疾病,高侵袭性和转移能力是其特点,也是影响病人疗效和预后的主要因素。HCC的发生、发展受到多种因素的影响。肿瘤微环境已被证实在肿瘤的发展转移和复发过程中起到重要作用,肿瘤微环境包括肿瘤细胞、细胞外基质和基质细胞(包括成纤维细胞、免疫和炎性细胞、脂肪细胞等)[8],而巨噬细胞是肿瘤微环境中浸润的一类重要免疫细胞。
在本研究中,CD163和CD206被用来标记M2巨噬细胞,检测M2巨噬细胞在不同分期肝癌样本中的表达。CD163是一种在人体中被CD163基因编码的蛋白质,是对血红蛋白-结合珠蛋白复合物高亲和力的清道夫受体, 其是一类吞噬细胞表面的跨膜蛋白。CD163仅仅在单核-巨噬细胞系中表达,在多种含成熟巨噬细胞的组织器官中高表达,如肺泡巨噬细胞、脾脏的血浆巨噬细胞、肝脏巨噬细胞(枯否细胞)等[9]。CD206,即甘露糖受体1,是一种主要存在于巨噬细胞和未成熟DC细胞的C型凝集素。有报道[10]称,其可促进巨噬细胞活化、抗原提呈和免疫应答。CD163和CD206是公认的鉴定M2型巨噬细胞的标志物[11-12],研究它们的共定位更能准确地定位M2型巨噬细胞。在本研究中,CD163和CD206在肝癌组织石蜡切片中的共定位明显高于对照组表达,同时在Ⅲ+Ⅳ期的共定位明显高于Ⅰ+Ⅱ期,提示M2巨噬细胞的表达可能与HCC的恶性度及转移有关。肝癌新鲜组织的Western blotting结果也显示,M2巨噬细胞在肝癌组织中的表达高于癌旁组织,同时晚期肝癌中CD163和CD206的表达明显高于早期肝癌。
NF-κB家庭主要成员包括RelA(p65)、c-Rel和RelB、NF-κB1(p50蛋白及其前体p105)、NF-κB2(p52蛋白及其前体p100)。有研究[13]表明,NF-κB可通过持续活化促进肿瘤细胞增殖,减少凋亡,提高肿瘤细胞的浸润和转移能力[13]。在哺乳动物中,p50是最重要的蛋白之一,参与了多种肿瘤细胞的表达,发挥重要作用。经典的NF-κB信号通路参与了机体的自身免疫[14],而脊椎动物的炎症反应是自身免疫的重要表现形式。肝癌是典型的炎症-肿瘤相关疾病。本研究结果显示,随着肝癌进展,p50表达明显增高。相似的,PORTA等[15]报道p50参与了结直肠癌的进展。同时M2巨噬细胞的数量也增多,而进一步研究NF-κB p50的表达与M2巨噬细胞的相关性发现,NF-κB p50蛋白的表达与M2巨噬细胞的表达呈显著正相关。既往研究[16-17]表明,TAM可释放多种细胞因子、趋化因子、生长因子、基质金属蛋白酶等,促进肿瘤进展。TAM促进肿瘤生长浸润可能与其高分泌白细胞介素-10、肿瘤坏死因子-α等细胞因子,激活NF-κB通路,上调NF-κB p50蛋白的表达相关。而NF-κB从细胞质进入细胞核内,调控炎性细胞因子的表达,上调信号转导和转录激活因子-3等的表达,进一步促进肿瘤进展[18]。同时,NK-κB p50能诱导M2巨噬细胞相关基因,是M2巨噬细胞驱动的炎症反应中的重要组分[19],提示M2巨噬细胞和NK-κB p50可能存在协同作用,共同促进肝癌进展。
综上所述,随着肝癌的进展,M2巨噬细胞和NK-κB p50的表达均上升,且具有明显相关性,提示M2巨噬细胞增多,可能通过激活NF-κB信号通路,上调p50表达,促进了HCC的恶性进程;而p50表达的上升可能进一步增多M2巨噬细胞,从而进一步促进肝癌的进展。本研究结果有助于进一步了解M2型巨噬细胞促进肿瘤浸润生长的机制,为肝癌靶向治疗提供新线索。
肝细胞癌中M2巨噬细胞标志物与NF-κB p50相关性研究
Expression of M2 macrophages in hepatocellular carcinoma, and its correlation with NF-κB p50
-
摘要:
目的探讨不同分期肝细胞癌病人组织中M2巨噬细胞及核因子κB(NF-κB)p50的表达情况,并分析其表达的相关性。 方法整群收集2012-2015年80例肝细胞肝癌病人和10例肝内胆管结石病人(对照组)的组织标本,采用免疫荧光双染法检测CD163和CD206的表达;收集2015年10月至2016年1月20例接受肝细胞癌手术病人的新鲜组织标本,采用Western blotting法检测不同分期的肝癌组织中CD163、CD206的表达及NF-κB p50的表达。 结果肝癌病人石蜡组织切片中CD163和CD206的共定位高于对照组,Ⅲ+Ⅳ期肝癌切片中的共定位高于Ⅰ+Ⅱ期;Western blotting结果表明,肝癌组织中CD163、CD206及NF-κB p50的表达高于癌旁组织(P < 0.05~P < 0.01),Ⅲ+Ⅳ期肝癌组织中的表达高于Ⅰ+Ⅱ期(P < 0.01)。NF-κB p50与CD163、CD206的表达均呈正相关(r=0.963、0.944,P < 0.01)。 结论随着肝癌进展,M2巨噬细胞数量和NF-κB p50的表达均升高,提示两者可能具有协同作用,共同促进肝癌进展。 Abstract:ObjectiveTo evaluate the expression of M2 macrophages and nuclear factor κB(NF-κB) p50 in patients with different stages of hepatocellular carcinoma(HCC), and analyze the relationship between them. MethodsThe expression levels of CD163 and CD206 in 80 cases of paraffin sections of patients with HCC and 10 cases of patients with hepatolithiasis(control group) were detected.The expression levels of CD163, CD206 and NF-κB p50 in 20 cases of fresh liver tissues of patients with different stages of HCC were detected using Western blotting from October 2015 to January 2016. ResultsThe co-localization of CD163 and CD206 in paraffin section of HCC was higher than that in control group, and the co-localization of which in TNM Ⅲ+Ⅳ stage HCC was higher than that in TNM Ⅰ+Ⅱstage HCC.The results of Western blotting showed that the expression levels of CD163, CD206 and NF-κB p50 in hepatocellular carcinoma tissue were higher than those in para-carcinoma tissue(P < 0.05 to P < 0.01), and the expression levels of CD163, CD206 and NF-κB p50 in Ⅲ+Ⅳ stage hepatocellular carcinoma tissue were higher than those in Ⅰ+Ⅱ stage hepatocellular carcinoma tissue(P < 0.05 to P < 0.01).The expression of NF-κB p50 was positively correlated with the expressions of CD163 and CD206(r=0.963, r=0.944, P < 0.01). ConclusionsWith the development of liver tissue, the number of M2 macrophages and NF-κB p50 level increase, which suggest that the two may have synergistic effects to promote the progression of liver cancer. -
表 1 不同分期肝癌病人新鲜肝脏组织中CD163、CD206、p50表达相对灰度值的比较(x±s; ni=3)
分组 CD163 CD206 p50 对照组 0.32±0.05 0.34±0.05 0.31±0.04 Ⅰ+Ⅱ期 0.48±0.07** 0.62±0.04* 0.85±0.04** Ⅲ+Ⅳ期 0.81±0.08**△△ 0.88±0.07**△△ 1.79±0.04**△△ F 40.044 66.482 991.034 P <0.01 <0.01 <0.01 MS组内 0.005 0.003 0.003 注:相对灰度值=目的蛋白灰度值/β-actin灰度值; q检验:与control组比较*P<0.05, **P<0.01;与Ⅰ+Ⅱ期组比较△△P<0.01 -
[1] LLOVET JM, ZUCMAN-ROSSI J, PIKARSKY E, et al.Hepatocellular carcinoma[J].Nat Rev Dis Primers, 2016, 2:16018. doi: 10.1038/nrdp.2016.18 [2] TORRE LA, BRAY F, SIEGEL RL, et al.Global cancer statistics, 2012[J].CA Cancer J Clin, 2015, 65:87. doi: 10.3322/caac.21262 [3] BORZIO M, FORNARI F, DE SIO I, et al.Adherence to American Association for the Study of Liver Diseases guidelines for the management of hepatocellular carcinoma:results of an Italian field practice multicenter study[J].Future Oncol, 2013, 9(2):283. [4] MANTOVANI A, ALLAVENA P, SICA A, et al.Cancer-related inflammation[J].Nature, 2008, 454(7203):436. doi: 10.1038/nature07205 [5] GUARDIOLA AA, GOMEZ RR, ROMERO GM, et al.Characteristics and course of chronic hepatitis B e antigen-negative infection[J].Gastroenterol Hepatol, 2017, 40(2):59. doi: 10.1016/j.gastrohep.2016.11.002 [6] INOUE T, TANAKA Y.Hepatitis B virus and its sexually transmitted infection - an update[J].Microb Cell, 2016, 3(9):420. [7] SIA D, VILLANUEVA A, FRIEDMAN SL, et al.Liver cancer cell of origin, molecular class, and effects on patient prognosis[J].Gastroenterology, 2017, 152(4):745. doi: 10.1053/j.gastro.2016.11.048 [8] SPANO D, ZOLLO M.Tumor microenvironment:a main actor in the metastasis process[J].Clin Exp Metastasis, 2012, 29(4):381. doi: 10.1007/s10585-012-9457-5 [9] PAPP H, LASZLO B, JAKAB F, et al.Review of group A rotavirus strains reported in swine and cattle[J].Vet Microbiol, 2013, 165(3/4):190. [10] LAWRENCE T, NATOLI G.Transcriptional regulation of macrophage polarization:enabling diversity with identity[J].Nat Rev Immunol, 2011, 11(11):750. doi: 10.1038/nri3088 [11] REBELO SP, PINTO C, MARTINS TR, et al.3D-3-culture:a tool to unveil macrophage plasticity in the tumour microenvironment[J].Biomaterials, 2018, 163:185. doi: 10.1016/j.biomaterials.2018.02.030 [12] WANG S, ZHANG J, SUI L, et al.Antibiotics induce polarization of pleural macrophages to M2-like phenotype in patients with tuberculous pleuritis[J].Sci Rep, 2017, 7(1):14982. doi: 10.1038/s41598-017-14808-9 [13] GONG H, ZULIANI P, KOMURAVELLI A, et al.Analysis and verification of the HMGB1 signaling pathway[J].BMC Bioinformatics, 2010, 11(Suppl 7):S10. [14] SMALE ST.Hierarchies of NF-kappaB target-gene regulation[J].Nat Immunol, 2011, 12(8):689. doi: 10.1038/ni.2070 [15] PORTA C, IPPOLITO A, CONSONNI FM, et al.Protumor Steering of Cancer Inflammation by p50 NF-κB Enhances Colorectal Cancer Progression[J].Cancer Immunol Res, 2018, 6(5):578. doi: 10.1158/2326-6066.CIR-17-0036 [16] BISWAS SK, ALLAVENA P, MANTOVANI A.Tumor-associated macrophages:functional diversity, clinical significance, and open questions[J].Semin Immunopathol, 2013, 35(5):585. doi: 10.1007/s00281-013-0367-7 [17] COOK J, HAGEMANN T.Tumour-associated macrophages and cancer[J].Curr Opin Pharmacol, 2013, 13(4):595. doi: 10.1016/j.coph.2013.05.017 [18] LEE H, HERRMANN A, DENG JH, et al.Persistently activated Stat3 maintains constitutive NF-κB activity in tumors[J].Cancer Cell, 2009, 15(4):283. doi: 10.1016/j.ccr.2009.02.015 [19] PORTA C, RIMOLDI M, RAES G, et al.Tolerance and M2 (alternative) macrophage polarization are related processes orchestrated by p50 nuclear factor kappaB[J].Proc Natl Acad Sci U S A, 2009, 106(35):14978. doi: 10.1073/pnas.0809784106 -





 下载:
下载: