-
脑血管疾病为致人死亡的重要原因,发病风险及病死风险均较高,其中有很多属于缺血性脑血管疾病。急性缺血性脑卒中即急性脑梗死,是由于颅内的血管内出现血液供应障碍、血管闭塞(不同因素导致),导致局部的脑组织出现缺血性病变,其严重程度与脑梗死发病部位、血管狭窄部位、不良生活习惯等有关,且对预后亦有一定影响[1-2]。血常规在临床上应用比较广泛,其检验结果能够反映体内的病理变化,亦可用于评价治疗效果、康复状况。脑梗死的演变过程中均可能有炎性反应的参与,如动脉粥样硬化斑块形成以及治疗后脑组织恢复及预后等。检验指标水平及炎性因子的检测与颈动脉粥样硬化斑块的稳定性等有关,对脑梗死病情发展能够产生一定的影响[3-4]。血常规检验中平均血小板体积(mean platelet volume,MPV)、平均血小板体积与淋巴细胞比值(mean platelet volume to lymphocyte ratio,MPVLR)等检测对于诊断、评估治疗效果等能够发挥积极作用。MPV升高已被确认为急性脑梗死的独立危险因素,并且MPV水平被认为是急性脑梗死病人预后的影响因子[5-6]。既往研究表明,脑梗死后淋巴细胞数量会立即下降,然而其在接下来的14 d内会逐步升高[7]。脑梗死病灶面积的增加及神经功能的恶化与淋巴细胞计数的降低有关[8-9]。此外,MPVLR作为一种新型炎症指标,与急性冠状动脉综合征病人冠状动脉病变的严重程度和长远的生存率密切相关[10]。由于心脑血管发病机制上有相似性,本研究旨在探讨入院时MPVLR检测对预测急性脑梗死严重程度的价值,现作报道。
-
回顾性分析2019年1月至2020年3月我院收治的首发且发病72 h内的急性脑梗死病人共109例,均符合中华医学会神经病学分会《中国急性缺血性卒中诊治指南》标准[11]。据2016年《高危非致残性缺血性脑血管事件诊疗指南》标准[12]NIHSS评分将其分为轻度组(NIHSS评分≤5分)64例、中重度组(NIHSS评分>5分) 45例。纳入标准:(1)经影像学检查等确诊;(2)首发≤72 h,无精神系统疾病;(3)年龄≥18岁;(4)病人及家属同意。排除标准:(1)出血性脑梗死;(2)颅内出血、感染、恶性肿瘤;(3)血液、免疫系统疾病;(4)服用抗炎及免疫抑制类药物;(5)严重肝肾功能不全。
-
收集病人一般临床资料、入院时实验室检查数据等,并根据MPV和淋巴细胞计数计算MPVLR值;记录病人入院NIHSS评分。
-
采用χ2检验、独立样本t检验、秩和检验和二元logistic回归分析,利用ROC曲线评价MPVLR对急性脑梗死严重程度的预测价值。
-
2组病人在年龄、性别等方面差异均无统计学意义(P>0.05)(见表 1)。
指标 轻度组
(n=64)中重度组
(n=45)χ2 P 年龄[P50(P25, P75)]/岁 66.00(56.00, 75.00) 66.00(51.00, 76.00) 0.46* >0.05 男/女 42/22 23/22 2.31 >0.05 吸烟 32(50.00) 23(51.11) 0.01 >0.05 饮酒 29(45.31) 27(60.00) 2.28 >0.05 高血压 44(68.75) 36(80) 1.71 >0.05 糖尿病 22(34.38) 17(37.78) 0.13 >0.05 高脂血症 23(35.93) 19(42.22) 0.44 >0.05 冠心病 9(14.06) 13(28.88) 3.61 >0.05 心房颤动 5(7.81) 5(11.11) 0.35 >0.05 *示uc值 表 1 一般临床资料的比较[n;百分率(%)]
-
中重度组病人MPVLR及单核细胞计数均高于轻度组,差异有统计学意义(P < 0.05),其他实验室指标2组差异均无统计学意义(P>0.05)(见表 2)。
指标 轻度组(n=64) 中重度组(n=45) uc P MPVLR 4.90(3.96, 6.70) 7.25(5.44, 9.26) 4.35 < 0.05 单核细胞计数/(×109/L) 0.33(0.21, 0.54) 0.43(0.34, 0.49) 2.41 ﹤0.05 中性粒细胞计数/(×109/L) 4.28(3.05, 5.61) 4.73(3.40, 6.51) 0.85 >0.05 白细胞计数/(×109/L) 7.28±2.54 7.14±3.15 0.27* >0.05 低密度脂蛋白/(mmol/L) 2.33±7.09 2.21±0.71 0.85* >0.05 总胆固醇/(mmol/L) 3.83±1.03 3.75±1.09 0.37* >0.05 尿酸/(μmol/L) 289.25±102.36 310.42±95.24 1.09* >0.05 血小板计数/(×109/L) 205.00(157.50, 253.75) 185.00(153.00, 240.50) 1.16 >0.05 D-二聚体/(mg/L) 0.46(0.22, 0.76) 0.47(0.31, 1.41) 1.42 >0.05 高密度脂蛋白/(mmol/L) 0.94(0.76, 1.21) 1.06(0.84, 1.26) 1.60 >0.05 三酰甘油/(mmol/L) 1.39(1.03, 1.82) 1.41(1.01, 1.87) 0.19 >0.05 空腹血糖/(mmol/L) 5.55(4.60, 7.35) 5.83(4.93, 7.43) 0.90 >0.05 胱抑素C/(mg/L) 0.90(0.78, 1.02) 0.99(0.84, 1.10) 1.59 >0.05 *示t值 表 2 实验室指标比较[P50(P25, P75)]
-
将MPVLR及单核细胞计数纳入logistic回归分析,结果显示MPVLR升高是急性脑梗死严重程度的独立危险因素(P < 0.01)(见表 3)。
变量 B SE Waldχ2 P OR(95%CI) MPVLR 0.244 0.076 10.40 < 0.01 1.277(1.101~1.481) 单核细胞 1.862 1.084 2.95 >0.05 6.438(0.769~53.895) 表 3 二分类logistic回归分析
-
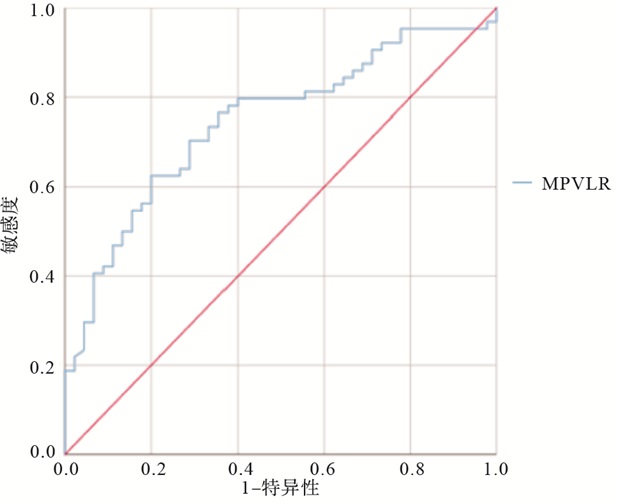
ROC曲线分析显示,MPVLR预测急性脑梗死严重程度的曲线下面积为0.745(95%CI: 0.653~0.824),最佳截断值为5.36,敏感度与特异度分别为80.0%和62.5% (见图 1)。
-
在脑梗死的病程中,炎性反应伴随脑组织缺血过程始终,因此通过血液检验指标,了解体内炎性反应发生情况,对预测急性脑梗死病人病情严重程度十分重要。如果可以在其发病初期使用简单、便捷的方法,快速评估急性脑梗死的严重程度,有利于对病人进行临床护理和治疗,改善预后。血常规检测结果与心脑血管及相关疾病诊疗之间有一定关联[13-14]。
急性脑梗死形成的主要原因是动脉粥样硬化,其形成的过程同血小板的聚集活化密不可分。虽然于生理状态下,血管壁受损后,血小板被暴露在外的细胞外基质蛋白激活的同时,仍可黏附血液中额外的血小板,从而在损伤部位形成牢固的血小板凝块,以止血;但是在病理条件下,血小板在激活和聚集的过程中易失去控制,并可能导致血管闭塞[15]。MPVLR近年来在临床上逐渐被重视,是将MPV和淋巴细胞计数整合为一体的新型炎症标志物,既可以反映凝血功能,又能反映炎症程度。MPV与淋巴细胞计数作为单一指标,可能不仅易受机体感染的影响,机体血容量的变化也可能会影响检测结果,所以把MPV与淋巴细胞计数比值整合成MPVLR的模式,即相当于分子与分母同时改变,也许能减少感染等相关因素的影响,进而更加真实地反映急性脑梗死的病情严重变化,可能从一定程度上反映冠脉侧支循环及血管造影复流程度。KURTUL等[16]证明MPVLR是ST段抬高型心肌梗死病人无复流和30 d死亡率的强有力的独立预测因子。同时,HUDZIK等[17]研究结果显示,MPVLR升高可以预测ST段抬高型心肌梗死病人随访期间的临床不良结果。MPV能够用于反映血小板功能及活力方面,其水平高度与机体是否患有血栓性疾病密切有关。MPV高的病人比MPV正常的病人更容易发生急性脑梗死[5]。目前国内外主要集中于MPV、中性粒细胞与淋巴细胞比值、血小板与淋巴细胞比值或两者联合检测判断急性脑梗死严重程度的相关性临床研究,而MPVLR与急性脑梗死严重程度相关性的报道尚不明确。故本次研究旨在分析MPVLR水平对预测急性脑梗死入院时严重程度的价值。
本研究显示,与轻度组比较,中重度组MPVLR水平较高,且是急性脑梗死病人病情的独立预测因素。MPVLR预测急性脑梗死严重程度的曲线下面积为0.745(95%CI: 0.653~0.824),最佳截断值为5.36,敏感度为80.0%,特异度为62.5%。研究结果可能与以下原因有关:血小板活化主要通过G蛋白偶联受体或免疫受体酪氨酸激活基序受体的信号转导通路,激活细胞内的信号级联反应,涉及细胞质内Ca2+浓度的升高、细胞骨架的重排、致密颗粒的动员、血小板激动剂的释放和整合素黏附受体的构象变化,引起血小板的黏附、聚集和血栓形成[18]。活化后的血小板可合成并释放趋化因子4、凝血因子、生长因子等生物活性物质,在上述细胞因子的趋化与诱导下,迁入内膜下的单核细胞不断摄取已发生氧化修饰的脂蛋白,进而形成单核细胞源性泡沫细胞,参与动脉粥样硬化斑块的产生。P-选择素和可溶性CD40L等促炎分子的释放、血小板促凝活性的表达以及GPIIb/IIIa(αIIβ3-Integrin)受体的激活,亦会加剧动脉斑块破裂以及病理性血栓的形成[19]。最早参与动脉粥样硬化斑块形成的细胞之一是淋巴细胞,其可通过释放内皮细胞黏附分子的表达或激活巨噬细胞或其他白细胞种群介体,最终激发脑卒中后炎性反应。据相关文献[20]报道,淋巴细胞减少与机体的生理应激和急性炎症反应有关,可能归因于炎症相关淋巴细胞凋亡的增加,从而使个体易患缺血性卒中。
本研究还发现单核细胞增加可能与更加严重的神经功能受损有关,这与相关文献报道[21]基本一致,即中重度脑梗死组病人单核细胞计数高于轻度脑梗死组病人。其产生机制可能是脑梗死可引起血脑屏障受损,通过激活小胶质细胞,使其释放促炎释放因子并影响细胞黏附分子表达,促使单核细胞被募集至损伤部位[22],进而使白细胞介素-1、白细胞介素-6、肿瘤坏死因子-α等细胞因子被单核细胞释放, 以促进炎症发生作用。
综上所述,MPVLR相对于单一指标而言,其数据水平更加稳定,而且能够体现体内血栓及机体免疫应答过程,对急性脑梗死的严重程度具有一定的预测价值,进而对临床识别高危病人有一定的价值。在实际临床工作中,建议与其他指标联合使用,进而加强临床价值,为提供更为合理的治疗及康复计划提供根据。但本项研究是基于回顾性收集病人相关临床数据,不可避免地产生了系统性偏差。且本次研究属于单中心小样本研究,并未进行随访,有一定的局限性,加之NIHSS评分易受主观因素等影响。因此,上述结论还需要进行大样本量、多个中心联合研究,来进一步证实。
平均血小板体积/淋巴细胞比值对急性脑梗死严重程度的预测价值
The predictive value of mean platelet volume-to-lymphocyte ratio in the severity of acute cerebral infarction
-
摘要:
目的探讨平均血小板体积/淋巴细胞比值(mean platelet volume-to-lymphocyte ratio,MPVLR)对急性脑梗死严重程度的预测价值。 方法回顾性分析109例急性脑梗死病人的临床资料,根据NIHSS评分将其分为轻度组(n=64)和中重度组(n=45)。根据2组血常规相关指标,计算MPVLR。采用二分类logistic回归分析急性脑梗死病情的影响因素,利用ROC曲线评价入院时MPVLR水平对急性脑梗死病变严重程度的预测价值。 结果轻度组MPVLR、单核细胞计数均低于中重度组,差异均有统计学意义(P < 0.05)。二元logistic回归分析显示,入院时MPVLR水平的增加,为急性脑梗死病情严重程度的独立危险因素。ROC曲线显示,MPVLR预测急性脑梗死严重程度的ROC曲线下面积为0.745(95%CI:0.653~0.824),最佳截断值为5.36,敏感度为80.0%,特异度为62.5%。 结论MPVLR对急性脑梗死严重程度具有一定预测价值。 -
关键词:
- 急性脑梗死 /
- 平均血小板体积/淋巴细胞比值
Abstract:ObjectiveTo explore the predictive value of the mean platelet volume-to-lymphocyte ratio(MPVLR) in the severity of acute cerebral infarction. MethodsA total of 109 patients with acute cerebral infarction were retrospectively analyzed, and divided into the mild group(n=64) and moderate-severe group(n=45) according to the NIHSS score.The MPVLR was calculated according to the blood routine indexes of two groups.The influencing factors of acute cerebral infarction were analyzed using dichotomous logistic regression, and the predictive value of MPVLR level on admission in the severity of acute cerebral infarction was evaluated using ROC curve. ResultsThe MPVLR and monocyte counts in mild group were lower than those in moderate group(P < 0.05).The results of binary logistic regression analysis showed that the increasing of MPVLR level on admission was an independent predictor of acute cerebral infarction.The results of ROC curve showed that the area under the curve of MPVLR predicting the severity of acute cerebral infarction was 0.745(95%CI: 0.653~0.824), and the best cutoff value, sensitivity and specificity were 5.36, 80.0% and 62.5%, respectively. ConclusionsThe MPVLR has certain predictive value in judging the severity of acute cerebral infarction. -
表 1 一般临床资料的比较[n;百分率(%)]
指标 轻度组
(n=64)中重度组
(n=45)χ2 P 年龄[P50(P25, P75)]/岁 66.00(56.00, 75.00) 66.00(51.00, 76.00) 0.46* >0.05 男/女 42/22 23/22 2.31 >0.05 吸烟 32(50.00) 23(51.11) 0.01 >0.05 饮酒 29(45.31) 27(60.00) 2.28 >0.05 高血压 44(68.75) 36(80) 1.71 >0.05 糖尿病 22(34.38) 17(37.78) 0.13 >0.05 高脂血症 23(35.93) 19(42.22) 0.44 >0.05 冠心病 9(14.06) 13(28.88) 3.61 >0.05 心房颤动 5(7.81) 5(11.11) 0.35 >0.05 *示uc值 表 2 实验室指标比较[P50(P25, P75)]
指标 轻度组(n=64) 中重度组(n=45) uc P MPVLR 4.90(3.96, 6.70) 7.25(5.44, 9.26) 4.35 < 0.05 单核细胞计数/(×109/L) 0.33(0.21, 0.54) 0.43(0.34, 0.49) 2.41 ﹤0.05 中性粒细胞计数/(×109/L) 4.28(3.05, 5.61) 4.73(3.40, 6.51) 0.85 >0.05 白细胞计数/(×109/L) 7.28±2.54 7.14±3.15 0.27* >0.05 低密度脂蛋白/(mmol/L) 2.33±7.09 2.21±0.71 0.85* >0.05 总胆固醇/(mmol/L) 3.83±1.03 3.75±1.09 0.37* >0.05 尿酸/(μmol/L) 289.25±102.36 310.42±95.24 1.09* >0.05 血小板计数/(×109/L) 205.00(157.50, 253.75) 185.00(153.00, 240.50) 1.16 >0.05 D-二聚体/(mg/L) 0.46(0.22, 0.76) 0.47(0.31, 1.41) 1.42 >0.05 高密度脂蛋白/(mmol/L) 0.94(0.76, 1.21) 1.06(0.84, 1.26) 1.60 >0.05 三酰甘油/(mmol/L) 1.39(1.03, 1.82) 1.41(1.01, 1.87) 0.19 >0.05 空腹血糖/(mmol/L) 5.55(4.60, 7.35) 5.83(4.93, 7.43) 0.90 >0.05 胱抑素C/(mg/L) 0.90(0.78, 1.02) 0.99(0.84, 1.10) 1.59 >0.05 *示t值 表 3 二分类logistic回归分析
变量 B SE Waldχ2 P OR(95%CI) MPVLR 0.244 0.076 10.40 < 0.01 1.277(1.101~1.481) 单核细胞 1.862 1.084 2.95 >0.05 6.438(0.769~53.895) -
[1] CHEN Y, LIU Y, LUO C, et al. Analysis of multiple factors involved in acute progressive cerebral infarction and extra- and intracranial arterial lesions[J]. Exp Ther Med, 2014, 7(6): 1495. doi: 10.3892/etm.2014.1624 [2] MILLÁN M, REMOLLO S, QUESADA H, et al. Vessel patency at 24 hours and its relationship with clinical outcomes and infarct volume in REVASCAT trial(randomized trial of revascularization with solitaire FR device versus best medical therapy in the treatment of acute stroke due to anterior circulation large vessel occlusion presenting within eight hours of symptom onset)[J]. Stroke, 2017, 48(4): 983. doi: 10.1161/STROKEAHA.116.015455 [3] YANG Y, XUE T, ZHU J, et al. Serum lipoprotein-associated phospholipase A2 predicts the formation of carotid artery plaque and its vulnerability in anterior circulation cerebral infarction[J]. Clin Neurol Neurosurg, 2017, 160: 40. doi: 10.1016/j.clineuro.2017.06.007 [4] XUE QZ, MENG AG, WANG T, et al. Correlation between of small dense low-density lipoprotein cholesterol with acute cerebral infarction and carotid atherosclerotic plaque stability[J]. J Clin Lab Anal, 2019, 33(6): e22891. [5] GREISENEGGER S, ENDLER G, HSIEH K, et al. Is elevated mean platelet volume associated with a worse outcome in patients with acute ischemic cerebrovascular events[J]. Stroke, 2004, 35(7): 1688. doi: 10.1161/01.STR.0000130512.81212.a2 [6] LIN CY, CHANG CY, SUN CH, et al. Platelet count and early outcome in patients with spontaneous cerebellar hemorrhage: a retrospective study[J]. PLoS One, 2015, 10(3): e0119109. doi: 10.1371/journal.pone.0119109 [7] PARK MG, KIM MK, CHAE SH, et al. Lymphocyte-to-monocyte ratio on day 7 is associated with outcomes in acute ischemic stroke[J]. Neurol Sci, 2018, 39(2): 243. doi: 10.1007/s10072-017-3163-7 [8] REN X, AKIYOSHI K, DZIENNIS S, et al. Regulatory B cells limit CNS inflammation and neurologic deficits in murine experimental stroke[J]. J Neurosci, 2011, 31(23): 8556. doi: 10.1523/JNEUROSCI.1623-11.2011 [9] LIESZ A, SURI-PAYER E, VELTKAMP C, et al. Regulatory T cells are key cerebroprotective immunomodulators in acute experimental stroke[J]. Nat Med, 2009, 15(2): 192. doi: 10.1038/nm.1927 [10] KILIC A, KURTUL A. RETRACTED: Mean platelet volume-to-lymphocyte ratio as a novel marker for severity and complexity of coronary atherosclerosis in patients with acute coronary syndrome[J]. Angiology, 2017: 3319717724274. [11] 中华医学会神经病学分会, 中华医学会神经病学分会脑血管病学组. 中国急性缺血性脑卒中诊治指南2018[J]. 中华神经科杂志, 2018, 51(9): 666. doi: 10.3760/cma.j.issn.1006-7876.2018.09.004 [12] 王伊龙, 赵性泉, 刘新峰, 等. 高危非致残性缺血性脑血管事件诊疗指南[J]. 中国卒中杂志, 2016, 11(6): 481. doi: 10.3969/j.issn.1673-5765.2016.06.011 [13] BARON T, BESKOW A, JAMES S, et al. Biobank linked to SWEDEHEART quality registry-routine blood sample collection opens new opportunities for cardiovascular research[J]. Ups J Med Sci, 2019, 124(1): 12. doi: 10.1080/03009734.2018.1498957 [14] MO L, CHEN Y, LI Z, et al. Red blood cell distribution width as a marker of cerebral infarction in hemodialysis patients[J]. Ren Fail, 2017, 39(1): 712. doi: 10.1080/0886022X.2017.1398664 [15] LIEN LM, LIN KH, HUANG LT, et al. Licochalcone a prevents platelet activation and thrombus formation through the inhibition of PLCγ2-PKC, Akt, and MAPK pathways[J]. Int J Mol Sci, 2017, 18(7): 1500. doi: 10.3390/ijms18071500 [16] KURTUL A, ACIKGOZ SK. Usefulness of mean platelet volume-to-lymphocyte ratio for predicting angiographic no-reflow and short-term prognosis after primary percutaneous coronary intervention in patients with st-segment elevation myocardial infarction[J]. Am J Cardiol, 2017, 120(4): 534. doi: 10.1016/j.amjcard.2017.05.020 [17] HUDZIK B, SZKODZIŃSKI J, LEKSTON A, et al. Mean platelet volume-to-lymphocyte ratio: a novel marker of poor short- and long-term prognosis in patients with diabetes mellitus and acute myocardial infarction[J]. J Diabetes Complications, 2016, 30(6): 1097. doi: 10.1016/j.jdiacomp.2016.04.010 [18] STEGNER D, KLAUS V, NIESWANDT B. Platelets as modulators of cerebral ischemia/reperfusion injury[J]. Front Immunol, 2019, 10: 2505. doi: 10.3389/fimmu.2019.02505 [19] JENNINGS LK. Mechanisms of platelet activation: need for new strategies to protect against platelet-mediated atherothrombosis[J]. Thromb Haemost, 2009, 102(2): 248. [20] CHEN SY, LIN YS, CHENG YF, et al. Mean platelet volume-to-lymphocyte ratio predicts poor functional outcomes among ischemic stroke patients treated with intravenous thrombolysis[J]. Front Neurol, 2019, 10: 1274. doi: 10.3389/fneur.2019.01274 [21] 李玲玲, 李永娟, 陈志斌. 急性脑梗死严重程度的影响因素及与淋巴细胞/单核细胞比值的关系[J]. 中国医学导报, 2018, 15(26): 62. [22] CHAUHAN A, HUDOBENKO J, Al MAMUN A, et al. Myeloid-specific TAK1 deletion results in reduced brain monocyte infiltration and improved outcomes after stroke[J]. J Neuroinflammation, 2018, 15(1): 148. doi: 10.1186/s12974-018-1188-3 -




 下载:
下载: