-
急性心肌梗死(AMI)是临床常见的危急重症,其主要病理机制为多种因素作用下冠状动脉(冠脉)内由胆固醇及其他沉积物组成的斑块发生破裂、出血、继发性血栓形成,使冠脉管腔进一步狭窄或完全闭塞。可能因素包括心脏血管退变、激素代谢紊乱和免疫应答等。其中机体免疫机制与心肌梗死范围及预后、并发症密切相关。为了进一步研究心肌梗死与细胞免疫功能的关系,本研究采用流式细胞技术,观察PCI与药物治疗两种途径的AMI病人淋巴细胞亚群表达情况。现作报道。
-
选择2017年12月至2019年6月我院心血管内科AMI病人60例作为观察组,排除标准:(1)肿瘤、结缔组织疾病及应用免疫抑制剂病人;重要脏器严重器质性损害者;(2)近半年有过手术、外伤、脑出血、脑梗死及消化系统出血等病史;(3)入院后有心肺复苏史;(4)不能排除主动脉夹层、急性胰腺炎或者胃溃疡穿孔者;(5)随访期间病人再次发生心肌梗死、心源性猝死等。其中30例行介入+药物治疗(PCI组)(均符合2016年中国急诊PCI治疗指南),另外30例采用常规药物治疗(药物组), 30 d后门诊随访。2组年龄、性别、合并基础疾病等一般资料比较差异均无统计学意义(见表 1)。择同期健康体检中心30名无特殊治疗的健康者作为对照组。
分组 n 年龄/岁 男 女 吸烟 高血压 糖尿病 体质量指数/(kg/m2) 谷丙转氨酶/(IU/L) 肌酐/(μmol/L) 低密度脂蛋白/(mmol/L) 高密度脂蛋白/(mmol/L) 三酰甘油/(mmol/L) 胆固醇/(mmol/L) PCI组 30 63.63±12.58 25 5 17(57) 21(70) 13(43) 21.46±2.62 37.93±20.10 84.33±31.56 2.75±0.72 1.37±0.43 1.30±0.71 4.32±0.87 药物组 30 63.73±14.41 23 7 16(60) 20(67) 20(67) 22.12±2.16 32.60±11.10 72.00±16.71 2.40±0.81 1.36±0.44 1.48±0.62 3.90±1.05 t — 0.03 0.42 0.07 0.08 3.30 1.06 1.27* 1.89* 1.77 0.09 1.05 1.69 P — >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 *示t′值 表 1 PCI组与药物组一般资料比较[n;百分率(%)]
-
所有研究对象均采集静脉血2 mL并于6 h内作染色处理,使用美国Beckman Coulter公司Epics XL型细胞流式仪,检测CD3+ CD8+,CD3+CD4+以及CD19+B百分比,计算CD4+/CD8+比值,罗氏全自动生化分析仪测定心肌肌钙蛋白Ⅰ(cTnI)水平。
-
采用t(或t′)检验、χ2检验、Pearson相关性分析。
-
观察组CD3+CD8+,CD19+B百分比高于对照组,而CD3+CD4+百分比及CD4+/CD8+比值低于对照组,差异均有统计学意义(P < 0.05~P < 0.01)(见表 2)。
分组 n CD3+/% CD3+CD4+/% CD3+CD8+/% CD4+/CD8+ CD19+B/% 观察组 60 58.75±5.06 28.93±4.68 26.58±4.53 1.15±0.39 14.00±3.01 对照组 30 57.42±5.18 31.40±3.06 23.81±3.84 1.35±0.25 12.78±1.39 t — 1.17 3.00* 2.87 2.94* 2.63 P — >0.05 < 0.01 < 0.01 < 0.01 < 0.05 *示t′值 表 2 观察组与对照组淋巴细胞亚群比较(x±s)
-
入组时,PCI组与药物组淋巴细胞亚群表达差异均无统计学意义(P>0.05)(见表 3);治疗30 d后,PCI组CD3+CD8+,CD19+B百分比低于药物组,CD3+CD4+百分比及CD4+/CD8+比值高于药物组,差异均有统计学意义(P < 0.05~P < 0.01)(见表 4)。
分组 n CD3+/% CD3+CD4+/% CD3+CD8+/% CD4+/CD8+ CD19+B/% PCI组 30 57.59±4.67 28.77±4.96 26.04±4.89 1.17±0.39 13.55±3.46 药物组 30 59.91±5.24 29.09±4.47 27.11±4.16 1.13±0.40 14.38±2.46 t — 1.81 0.26 0.91 0.39 1.07 P — >0.05 >0.05 >0.05 >0.05 >0.05 表 3 入组时PCI组与药物组淋巴细胞亚群比较(x±s)
分组 n CD3+/% CD3+CD4+/% CD3+CD8+/% CD4+/CD8+ CD19+B% PCI组 30 59.12±6.12 36.15±4.49 21.90±4.73 1.72±0.39 11.51±2.25 药物组 30 57.72±4.23 32.87±5.32 24.99±6.25 1.38±0.39 12.96±2.00 t — 1.03 2.58 2.16 3.38 2.64 P — 0.31 < 0.05 < 0.05 < 0.01 < 0.05 表 4 治疗30 d后PCI组与药物组淋巴细胞亚群比较(x±s)
-
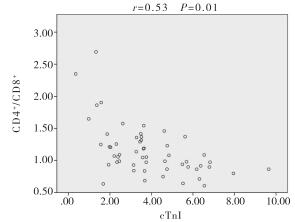
Pearson相关性分析结果显示,CD4+/CD8+比值与cTnI呈负相关(r=-0.53,P < 0.05)(见图 1)。
-
近年来冠脉粥样硬化性心脏病严重危害人类健康,其中AMI逐渐成为冠心病致死最主要原因[1]。有研究[2]显示免疫防御机制参与动脉粥样硬化斑块形成, 当血管壁受损后随之激活免疫炎性反应,淋巴细胞在免疫炎性反应中起关键作用。DE等[3]在实验中运用RT-PCR技术检测出动脉粥样易损斑块较稳定斑块T淋巴细胞相对增加,其促使局部的免疫应答信号增强,同时分泌细胞因子及生长因子,促进血管平滑肌细胞的增生、迁移及吞噬[4-5]。因此,T淋巴细胞参与斑块的稳定。
按细胞表面膜标志(CD)分子T淋巴细胞CD3+主要可分为:CD4+和CD8+两群,两者比例失调可导致免疫功能紊乱,这可能成为AMI的发生与发展的使动因素。CD4+细胞称为激活的辅助性T细胞(Th),通过一系列抗原抗体反应被激活,分泌细胞因子,调节免疫反应,产生大量的促炎性细胞因子,对细胞外基质的代谢和内皮细胞的功能起到很大影响[6]。SHAW[7]研究发现将低龄无动脉粥样硬化的小鼠被注入高龄并且缺乏载脂蛋白E老鼠CD4+T细胞,不久发现小鼠有冠脉粥样硬化病理改变。CD8+细胞又称细胞毒性T细胞(Tc),在抗原与抗体反应中起到杀伤靶细胞的作用。黄立娟等[8]研究表明冠心病病人中CD8+比例与冠脉病变严重程度呈正相关。然而CD4+与CD8+比例失衡仍然是动脉硬化病变特征性表现之一[9]。
B淋巴细胞在免疫防御机制中的核心作用为产生抗体[10-11]。KONSTANTINOVA等[12-13]研究发现心肌坏死组织释放分离抗原,需要足够的抗体与之结合,因此检测B淋巴细胞较前明显增多。本研究发现观察组较对照组B淋巴细胞比例升高。
本研究表明,AMI病人CD3+CD8+及CD19+B百分比升高,CD3+CD4+百分比下降,可见淋巴细胞亚群参与AMI免疫炎性反应。PCI组较药物组CD3+CD8+及CD19+B百分比下降,CD3+CD4+百分比升高,提示PCI使AMI病人心肌免疫炎性损伤得到不同程度修复。
目前用于诊断AMI辅助的常用实验室指标为cTnI,其增高程度与心肌损伤严重程度成正比。有研究[14]提示, AMI病人预后与CD4+/CD8+比值相关。本实验发现CD4+/CD8+比值与cTnI呈负相关,CD4+/CD8+比值越低,心肌损伤越严重,预后越差。
多项研究[15-17]表明免疫反应在AMI的发生、发展中具有重要作用,更有研究从免疫应答的角度观察了淋巴细胞与AMI病人心功能的关系,但这些研究尚处于初级阶段,较少深刻探讨何种信号通路参与其中的过程以及如何进行有效的免疫干预治疗,仍需大量的临床试验,这可能成为今后该领域研究的方向。
不同治疗途径的急性心肌梗死病人淋巴细胞亚群表达变化分析
Analysis of the lymphocyte subsets expression in acute myocardial infarction patients treated with different treatment approaches
-
摘要:
目的分析急诊经皮冠脉介入治疗(PCI)与药物治疗两种途径的急性心肌梗死(AMI)病人淋巴细胞亚群表达情况,探讨AMI发病的免疫防御机制。 方法选取60例AMI病人作为观察组,其中30例行PCI+药物治疗(PCI组),另外30例采用常规药物治疗(药物组),并择同期健康体检中心30名无特殊治疗的健康者作为对照组。采集所有研究对象入院当日外周血以及治疗30 d后观察组门诊随访的外周血,通过流式细胞术测定淋巴细胞亚群表达情况,同时检测观察组当日外周血心肌肌钙蛋白Ⅰ(cTnI)含量。 结果与对照组比较,观察组CD3+CD8+、CD19+B百分比增加,CD3+CD4 +百分比及CD4+/CD8+比值降低,差异均有统计学意义(P < 0.05~P < 0.01);治疗30 d后,PCI组与药物组相比,淋巴细胞亚群表达结果与之相反,差异均有统计学意义(P < 0.05~P < 0.01);Pearson相关性分析显示,CD4+/CD8+比值与cTnI呈负相关关系(r=-0.53,P < 0.05)。 结论淋巴细胞亚群参与AMI免疫炎性反应,PCI治疗使AMI病人心肌免疫炎性损伤得到不同程度修复。CD4+/CD8+比值与cTnI呈负相关,CD4+/CD8+比值越低,cTnI值越高,提示心肌损伤越严重,预后越差。 Abstract:ObjectiveTo compare the expression changes of lymphocyte subsets in acute myocardial infarction (AMI) patients treated with between emergency percutaneous coronary intervention (PCI) and drug, and explore the immune defence mechanism of AMI occurrence. MethodsSixty AMI patients were set as the observation group[including 30 cases treatment with PCI combined with medication (PCI group) and 30 cases treatment with conventional medication (medication group), and 30 healthy people treated without special treatment were set as the control group.The peripheral blood of two groups were collected on the day of admission, the peripheral blood in the observation group were collected after 30 days of following-up.The expression of lymphocyte subsets was determined by flow cytometry, and the content of troponin Ⅰ (cTnI) in peripheral blood of the observation group was also detected. ResultsCompared with the control group, the percentages of CD3+CD8+ and CD19+ B lymphocytes increased, the percentages of CD3+CD4+ and CD4+/CD8+ ratio decreased in the observation group, and the differences of whose were statistically significant (P < 0.05 to P < 0.01).After 30 days of treatment, the expression of lymphocyte subsets in the PCI group was contrary to that in the drug group, and the difference of which was statistically significant (P < 0.05 to P < 0.01).The results of Pearson correlation analysis showed that the CD4+/CD8+ ratio was negatively correlated with cTnI (r=-0.53, P < 0.05). ConclusionsLymphocyte subsets are involved in the inflammatory response of AMI.The PCI therapy can repair myocardial immune inflammatory injury in AMI patients to different degrees.The CD4+/CD8+ ratio is negatively correlated with cTnI.The lower the CD4+/CD8+ ratio is, the higher the cTnI is, and which suggests that the more severe the myocardial damage is, the worse the prognosis is. -
表 1 PCI组与药物组一般资料比较[n;百分率(%)]
分组 n 年龄/岁 男 女 吸烟 高血压 糖尿病 体质量指数/(kg/m2) 谷丙转氨酶/(IU/L) 肌酐/(μmol/L) 低密度脂蛋白/(mmol/L) 高密度脂蛋白/(mmol/L) 三酰甘油/(mmol/L) 胆固醇/(mmol/L) PCI组 30 63.63±12.58 25 5 17(57) 21(70) 13(43) 21.46±2.62 37.93±20.10 84.33±31.56 2.75±0.72 1.37±0.43 1.30±0.71 4.32±0.87 药物组 30 63.73±14.41 23 7 16(60) 20(67) 20(67) 22.12±2.16 32.60±11.10 72.00±16.71 2.40±0.81 1.36±0.44 1.48±0.62 3.90±1.05 t — 0.03 0.42 0.07 0.08 3.30 1.06 1.27* 1.89* 1.77 0.09 1.05 1.69 P — >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 >0.05 *示t′值 表 2 观察组与对照组淋巴细胞亚群比较(x±s)
分组 n CD3+/% CD3+CD4+/% CD3+CD8+/% CD4+/CD8+ CD19+B/% 观察组 60 58.75±5.06 28.93±4.68 26.58±4.53 1.15±0.39 14.00±3.01 对照组 30 57.42±5.18 31.40±3.06 23.81±3.84 1.35±0.25 12.78±1.39 t — 1.17 3.00* 2.87 2.94* 2.63 P — >0.05 < 0.01 < 0.01 < 0.01 < 0.05 *示t′值 表 3 入组时PCI组与药物组淋巴细胞亚群比较(x±s)
分组 n CD3+/% CD3+CD4+/% CD3+CD8+/% CD4+/CD8+ CD19+B/% PCI组 30 57.59±4.67 28.77±4.96 26.04±4.89 1.17±0.39 13.55±3.46 药物组 30 59.91±5.24 29.09±4.47 27.11±4.16 1.13±0.40 14.38±2.46 t — 1.81 0.26 0.91 0.39 1.07 P — >0.05 >0.05 >0.05 >0.05 >0.05 表 4 治疗30 d后PCI组与药物组淋巴细胞亚群比较(x±s)
分组 n CD3+/% CD3+CD4+/% CD3+CD8+/% CD4+/CD8+ CD19+B% PCI组 30 59.12±6.12 36.15±4.49 21.90±4.73 1.72±0.39 11.51±2.25 药物组 30 57.72±4.23 32.87±5.32 24.99±6.25 1.38±0.39 12.96±2.00 t — 1.03 2.58 2.16 3.38 2.64 P — 0.31 < 0.05 < 0.05 < 0.01 < 0.05 -
[1] WATANABE H, MORIMOTO T, SHIOMI H, et al.Chronic total occlusion in non-infarct-related artery is associated with increased short-and long-term mortality in patients with ST-segment elevation acute myocardial infarction complicated by cardiogenic shock (from the CREDO-Kyoto AMI registry)[J].Catheter Cardiovasc Interv, 2018, 92(3):455. doi: 10.1002/ccd.27330 [2] SCHAFTENAAR F, FRODERMANN V, KUIPER J, et al.Atherosclerosis:the interplay between lipids and immune cells[J].Curr Opin Lipidol, 2016, 27(3):209. doi: 10.1097/MOL.0000000000000302 [3] DE PALMA R, CIRILLO P, CICCARELLI G, et al.Expression of functional tissue factor in activated T-lymphocytes in vitro and in vivo:A possible contribution of immunity to thrombosis?[J].Int J Cardiol, 2016, 218:188. doi: 10.1016/j.ijcard.2016.04.177 [4] MRÁZ M, CINKAJZLOVÁ A, KLOUČKOVÁ J, et al.Coronary artery disease is associated with an increased amount of T lymphocytes in human epicardial adipose tissue[J].Mediators Inflamm, 2019, 2019:4075086. [5] BENAGIANO M, BORGHI MO, ROMAGNOLI J, et al.Interleukin-17/Interleukin-21 and Interferon-γ producing T cells specific for β2 Glycoprotein I in atherosclerosis inflammation of systemic lupus erythematosus patients with antiphospholipid syndrome[J].Haematologica, 2019, 104(12):2519. doi: 10.3324/haematol.2018.209536 [6] FORMANOWICZ D, GUTOWSKA K, FORMANOWICZ P.Theoretical studies on the engagement of interleukin 18 in the immuno-inflammatory processes underlying atherosclerosis[J].Cell Metab, 2019, 29(2):475. [7] SHAW PX.Rethinking oxidized low-density lipoprotein, its role inatherogenesis and the immune responses associated with it[J].ArchImmunol Ther Exp, 2004, 5(2):225. [8] 黄立娟, 崔颖, 孙文英, 等.冠心病患者外周血CD4+CD28nullT淋巴细胞亚群变化及其临床意义[J].中华检验医学杂志, 2007, 30(4):4246. [9] 陈丹丹, 王敬萍.免疫功能失调与急性冠脉综合征发病关系的研究[J].中西医结合心脑血管病杂志, 2009, 7(4):405. doi: 10.3969/j.issn.1672-1349.2009.04.013 [10] TSIANTOULAS D, SAGE AP, GÖDERLE L, et al.B Cell-activating factor neutralization aggravates atherosclerosis.[J].Circulation, 2018, 138(20):2263. doi: 10.1161/CIRCULATIONAHA.117.032790 [11] SARKAR A, RAFIQ K.Humoral immunity in heart failure[J].Cardiovasc Hematol Disord Drug Targets, 2019, 19(1):14. doi: 10.2174/1871529X18666180518101527 [12] KONSTANTINOVA EV, KOCHETOV AG, SHOSTAK NA, et al.Characteristics of immune response and inflammatory reaction in atherothrombotic stroke and myocardial infarction[J].Zh Nevrol Psikhiatr Im S S Korsakova, 2015, 115(2):48. doi: 10.17116/jnevro20151152148-52 [13] AWAN MS, DAUD MY, KHAN M, et al.Usefulness of neutrophils to lymphocytes ratio for predicting troponin-i elevation in patients presenting with suspected nste-acute coronary syndrome[J].J Ayub Med Coll Abbottabad, 2019, 31(Suppl 1)(4):S674. [14] BRUNETTI ND, D'ANTUONO C, RANA M, et al.Lymphocyte subset characterization in patients with early clinical presentation of coronary heart disease[J].J Thromb Thrombolysis, 2012, 34(4):475. doi: 10.1007/s11239-012-0761-3 [15] DONG CH, WANG ZM, CHEN SY.Neutrophil to lymphocyte ratio predict mortality and major adverse cardiac events in acute coronary syndrome:A systematic review and meta-analysis[J].Clin Biochem, 2018, 52:131. doi: 10.1016/j.clinbiochem.2017.11.008 [16] XU N, TANG XF, YAO Y.Predictive value of neutrophil to lymphocyte ratio in long-term outcomes of left main and/or three-vessel disease in patients with acute myocardial infarction[J].Catheter Cardiovasc Interv, 2018, 91(S1):551. doi: 10.1002/ccd.27495 [17] WAN Q, LIU Z, YANG M, et al.Acceleratory effects of ambient fine particulate matter on the development and progression of atherosclerosis in apolipoprotein E knockout mice by down-regulating CD4+CD25+Foxp3+ regulatory T cells[J].Toxicol Lett, 2019, 316:27. doi: 10.1016/j.toxlet.2019.09.005 -





 下载:
下载: