-
急性肺损伤(ALI)是一种流行性炎症性肺疾病,是急性呼吸窘迫综合征(ARDS)的重要原因,其发病率和重症病人死亡率均很高。尽管在治疗策略方面已取得重大进展,但ARDS和ALI的年死亡率仍为40%,且医疗费用较高[1]。ALI作为一种进展性疾病,以促炎性因子的过度产生、炎症细胞浸润以及肺泡上皮细胞的凋亡为主要特征[2]。因此,有效地控制肺泡上皮细胞炎症和凋亡对改善病人预后意义重大。长链非编码RNA(lncRNA)通过染色体重塑、转录和转录后加工等多种方式参与调节基因表达,在广泛的生物学领域具有重要功能[3-4]。研究[5]证实,在低氧诱导的心肌细胞损伤中lncRNA嗜酸性粒细胞转录因子(EGOT)低表达,脑钠肽能通过促进lncRNA EGOT表达,抑制促凋亡蛋白表达从而缓解低氧诱导的心肌细胞损伤。lncRNABase网站在线预测显示,EGOT与miR-320a之间存在结合位点。既往研究[6]显示,高氧刺激后肺泡上皮细胞微泡中miR-320a表达增加促进巨噬细胞介导的肺部炎性反应。基于以上研究,本研究探讨lncRNA EGOT、miR-320a在ALI/ARDS中的作用及可能机制,以期为ALI/ARDS提供有效的临床治疗策略。
-
肺泡上皮细胞A549购自中科院上海细胞库;RPMI-1640、胎牛血清购自北京索莱宝生物;脂多糖(LPS)购自美国Sigma公司;miR-320a抑制物(anti-miR-320a)及其阴性对照(anti-miR-NC)、EGOT过表达质粒(pcDNA-EGOT)、质粒空载体(pcDNA)、miR-320a模拟物(miR-320a mimics)及其阴性对照(miR-NC)、EGOT小干扰RNA(si-EGOT)及其阴性对照(si-NC)、双荧光素酶报告基因载体购自广州锐博生物;白细胞介素(IL)-6、IL-1β酶连免疫吸附测定(ELISA)试剂盒购自北京博凌科为生物;Annexin V-FITC/PI凋亡检测试剂盒购自南京凯基生物;兔抗人B淋巴瘤-2(Bcl-2)抗体、兔抗人Bcl相关X蛋白(Bax)抗体、兔抗人甘油醛-3-磷酸脱氢酶(GAPDH)抗体、羊抗兔IgG购自上海艾博抗生物公司。
-
A549细胞采用含10%胎牛血清的RPMI-1640培养基置于含5% CO2、37 ℃培养箱中培养。将对数期A549按照2×105个/孔接种24孔板,1 h后加入LPS使其终浓度为10 mg/L孵育细胞24 h,记为LPS组[7],同时设置不加LPS对照(Con)组,收集细胞进行后续试验。为证实lncRNA EGOT、miR-320a在LPS诱导的A549细胞炎症反应和凋亡中的作用,利用脂质体转染法将pcDNA、pcDNA-EGOT、anti-miR-NC、anti-miR-320a、pcDNA-EGOT+miR-NC、pcDNA-EGOT+miR-320a mimics分别转染A549细胞,用含10 mg/L LPS的细胞培养液处理转染成功的细胞24 h,依次记为LPS+pcDNA组、LPS+pcDNA-EGOT组、LPS+anti-miR-NC组、LPS+anti-miR-320a组、LPS+pcDNA-EGOT+miR-NC组、LPS+ pcDNA-EGOT+miR-320a组,收集细胞进行后续试验。
-
使用Trizol试剂盒提取总RNA含量。PrimeScript RT试剂盒将总RNA反转录为cDNA,应用SYBR ® Premix Ex TaqTM Ⅱ试剂盒通过ABI7500系统进行RT-qPCR。U6作为miR-320a的内参,GAPDH作为lncRNA EGOT内参,2-△△CT法计算lncRNA EGOT和miR-320a相对表达量。U6引物F:5′-GTG ATC ACT CCC TGC CTG AG-3′,R:5′-GGA CTT CAC TGG ACC AGA CG-3′;GAPDH引物F:5′-CCG CAT CTT CTT GTG CAG TG-3′,R:5′-CCC AAT ACG GCC AAA TCC GT-3′;miR-320a引物F:5′-AAG GGA TCG CGG GCG-3′,R:5′-TGC GTG TCG TGG AGT C-3′;EGOT引物F:5′-CAC TGC ACA GGG AAA CAC AAA-3′,R:5′-ACC CTG TTC ATA AGC CCT GAT G-3′。
-
将未转染或转染后A549细胞调整为4×105个/毫升,取500 μL细胞悬液加至24孔板中,1 h后,按照LPS终浓度为10 mg/L加入LPS再培养24 h。1 500 r/min离心10 min,收集上清液参照ELISA试剂盒说明书检测IL-6和IL-1β水平。
-
采用结合缓冲液将LPS处理24 h的各组细胞调整为1×105个/毫升单细胞悬液。取100 μL细胞悬液加入到流式管,按照凋亡检测试剂盒说明书加入Annexin V-FITC和PI试剂进行染色,避光反应15 min,1 h内上级检测细胞凋亡。
-
用细胞裂解液提取各组细胞总蛋白。定量后,将30 μg蛋白质上样到聚丙烯酰胺电泳凝胶上,恒流转移至聚偏氟乙烯膜。将膜用含5%脱脂牛奶封闭液封闭1 h,并与稀释的兔抗Bcl-2、兔抗Bax抗体4 ℃孵育过夜。将膜在洗膜缓冲液中洗涤2次后,与HRP偶联的羊抗兔二抗在室温下孵育2 h。加入化学发光试剂显色、定影,图像处理软件分析各条带灰度值,Bcl-2和Bax表达相对表达量以其灰度值与内参灰度值比值表示。
-
应用lncRNABase在线预测显示lncRNA EGOT与miR-320a存在结合位点。基于预测结合位点合成EGOT野生型(WT)和突变型(MUT)序列,将上述序列分别克隆pmirGLO荧光素酶载体构建WT-EGOT、MUT-EGOT。将miR-320a mimics、miR-NC与WT-EGOT或MUT-EGOT分别共转染至A549细胞48 h后,双荧光素酶报告基因检测试剂盒测定相对荧光素酶活性。将pcDNA-EGOT、pcDNA、si-EGOT、si-NC分别转染A549细胞,转染48 h RT-qPCR法检测miR-320a表达水平。
-
采用t检验、方差分析和q检验。
-
与Con组相比,LPS组A549细胞中lncRNA EGOT表达明显降低(P<0.01),miR-320a表达明显升高(P<0.01)(见表 1)。
分组 EGOT miR-320a Con 1.00±0.07 1.00±0.08 LPS 0.39±0.03 2.61±0.23 t 24.03 19.83 P <0.01 <0.01 表 1 lncRNA EGOT和miR-320a在LPS刺激的肺上皮细胞中的表达(ni=9;x±s)
-
EGOT过表达对LPS诱导的肺泡上皮细胞炎症因子表达的影响与Con组相比,LPS组A549细胞中EGOT表达降低(P<0.05),培养液中IL-6和IL-1β水平均升高(P<0.05);与LPS+pcDNA组相比,LPS+pcDNA-EGOT组A549细胞中EGOT表达升高(P<0.05),培养液中IL-6和IL-1β水平均降低(P<0.05)(见表 2)。
分组 EGOT IL-6/(pg/mL) IL-1β/(pg/mL) Con 1.00±0.05 106.48±10.42 46.17±4.94 LPS 0.43±0.04* 353.51±25.90* 180.73±12.98* LPS+pcDNA 0.41±0.04* 349.84±19.43* 177.28±14.47* LPS+pcDNA-EGOT 2.31±0.22*#▲ 139.69±12.53*#▲ 64.96±6.26*#▲ F 528.68 482.33 419.16 P <0.01 <0.01 <0.01 MS组内 0.014 328.478 110.363 q检验:与Con组比较*P<0.05;与LPS组比较#P<0.05;与LPS+pcDNA组比较▲P<0.05 表 2 lncRNA EGOT过表达对LPS诱导的肺泡上皮细胞炎症因子表达的影响(ni=9;x±s)
-
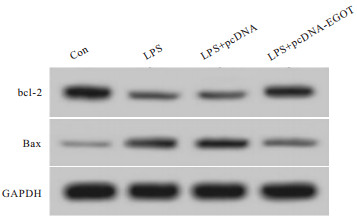
与Con组相比,LPS组A549细胞凋亡率、Bax蛋白表达均升高(P<0.05),Bcl-2蛋白表达降低(P<0.05);与LPS+pcDNA组相比,LPS+pcDNA-EGOT组A549细胞凋亡率、Bax蛋白表达均降低(P<0.05),Bcl-2蛋白表达升高(P<0.05)(见图 1~2、表 3)。
分组 凋亡率/% Bcl-2蛋白 Bax蛋白 Con 6.52±0.60 0.73±0.07 0.17±0.02 LPS 25.45±2.28* 0.28±0.03* 0.55±0.04* LPS+pcDNA 26.34±2.81* 0.26±0.02* 0.58±0.05* LPS+pcDNA-EGOT 10.59±1.01*#▲ 0.62±0.05*#▲ 0.24±0.02*#▲ F 256.47 234.86 324.49 P <0.01 <0.01 <0.01 MS组内 3.619 0.002 0.001 q检验:与Con组比较*P<0.05;与LPS组比较#P<0.05;与LPS+pcDNA组比较▲P<0.05 表 3 lncRNA EGOT过表达对LPS诱导的肺泡上皮细胞凋亡的影响(ni=9;x±s)
-
LncBase Predicted v.2在线预测分析显示,miR-320a与EGOT之间存在结合位点(见图 3)。双荧光素酶报告实验显示,与miR-NC和WT-EGOT共转染组相比,miR-320a mimics和WT-EGOT共转染组A549细胞相对荧光素酶活性降低(P<0.05);与miR-NC和MUT-EGOT共转染组相比,miR-320a mimics和MUT-EGOT共转染组A549细胞相对荧光素酶活性变化差异无统计学意义(P>0.05)(见表 4)。与pcDNA组相比,pcDNA-EGOT组A549细胞miR-320a表达水平降低(P<0.05);与si-NC组相比,si-EGOT组A549细胞miR-320a表达水平升高(P<0.05)(见表 5)。
分组 WT-EGOT MUT-EGOT miR-NC 0.99±0.05 0.97±0.06 miR-320a 0.38±0.04 1.00±0.07 t 28.58 0.98 P <0.01 >0.05 表 4 双荧光素酶报告实验结果(ni=9;x±s)
分组 miR-320a F P MS组内 pcDNA 1.00±0.05 579.59 <0.01 0.018 pcDNA-EGOT si-NC 0.50±0.05* 1.03±0.08# si-EGOT 2.97±0.25*#▲ q检验:与pcDNA组比较*P<0.05;与pcDNA-EGOT组比较#P<0.05;与si-NC组比较▲P<0.05 表 5 lncRNA EGOT靶向调控miR-320a的表达(ni=9;x±s)
-
与LPS+anti-miR-NC组比较,LPS+anti-miR-320a组A549细胞miR-320a表达、Bax蛋白表达、细胞凋亡率均明显降低(P<0.01),Bcl-2蛋白表达明显升高(P<0.01),培养液中IL-6和IL-1β水平均明显降低(P<0.01)(见图 4~5、表 6)。
分组 miR-320a IL-6/(pg/mL) IL-1β/(pg/mL) 凋亡率/% Bcl-2蛋白 Bax蛋白 LPS+anti-miR-NC 1.00±0.06 348.12±16.39 181.36±14.88 27.92±2.51 0.27±0.03 0.60±0.04 LPS+anti-miR-320a 0.54±0.05 152.01±16.50 78.06±7.03 12.56±1.18 0.58±0.04 0.30±0.03 t 17.67 25.30 188.83 16.61 18.60 14.40 P <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 表 6 抑制miR-320a表达对LPS诱导的肺泡上皮细胞炎症因子和细胞凋亡的影响(ni=9;x±s)
-
EGOT过表达对LPS诱导的肺泡上皮细胞炎症因子和细胞凋亡的作用与LPS+pcDNA-EGOT+miR-NC组比较,LPS+pcDNA-EGOT+miR-320a组A549细胞miR-320a表达、Bax蛋白表达、细胞凋亡率均明显升高(P<0.01),Bcl-2蛋白表达明显降低(P<0.01),培养液中IL-6和IL-1β水平均明显升高(P<0.01)(见图 6~7、表 7)。
分组 miR-320a IL-6/(pg/mL) IL-1β/(pg/mL) 凋亡率/% Bcl-2蛋白 Bax蛋白 LPS+pcDNA-EGOT+miR-NC 1.00±0.06 137.82±11.14 63.34±6.47 11.79±1.02 0.64±0.06 0.22±0.02 LPS+pcDNA-EGOT+miR-320a 2.53±0.23 311.34±28.45 145.64±12.94 20.51±1.84 0.32±0.03 0.45±0.03 t 19.31 17.04 17.07 12.44 14.31 19.14 P <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 表 7 miR-320a过表达逆转lncRNA EGOT过表达对LPS诱导的肺泡上皮细胞炎症因子和细胞凋亡的作用(ni=9;x±s)
-
革兰阴性菌引起的感染是ALI的重要病因,其可诱导剧烈的炎症反应和凋亡,导致呼吸膜的形态和功能损伤。本研究以脂多糖诱导肺泡上皮细胞A549损伤模拟ALI,探讨与ALI进展有关的基因和可能机制,以期为基于对抗肺泡上皮细胞损伤提供有效策略。
研究[8]显示,人脑利钠肽可提高缺氧条件下心肌细胞活力、减轻细胞凋亡和自噬进而改善缺氧诱导的心机细胞损伤,其机制与促进lncRNA EGOT表达有关。lncRNA EGOT通过调控基因表达还可抑制缺氧诱导肾小管的自噬防止急性损伤[9]。本研究显示,LPS诱导后A549细胞凋亡率、培养液中IL-6和IL-1β水平均明显升高,而lncRNA EGOT表达显著降低,提示lncRNA EGOT低表达可能与LPS诱导的A549炎症反应和凋亡有关。一般认为ALI的实质为炎症介质的释放导致肺部过度的炎症反应,鸢尾素通过抑制IL-6和IL-1β等炎性介质的产生可减轻肺部炎症损伤[10]。Bcl-2家族蛋白在细胞凋亡的信号转导中起着重要调节作用,Bax/Bcl-2比例失衡可导致细胞中线粒体膜电位的变化,促进线粒体细胞色素C释放到细胞质,然后激活caspase-3,最终导致细胞凋亡[11-13]。本研究显示,转染pcDNA-EGOT上调EGOT表达后,LPS诱导的A549中促凋亡蛋白Bax表达、凋亡率降低,抗凋亡蛋白Bcl-2表达增加,培养液中IL-6和IL-1β水平降低,说明lncRNA EGOT减轻LPS诱导的A549炎症反应和凋亡,是ALI临床治疗的潜在靶标。
生物信息学在线分析显示,lncRNA EGOT与miR-320a之间存在结合位点。miRNA是基因表达的重要调节剂,其在细胞增殖、代谢、凋亡等生物学过程中具有重要作用[14-15]。研究[16-17]显示,miR-320a高表达参与糖尿病肾病肾功能紊乱、骨关节炎进展。在阿霉素诱导的心脏损伤中miR-320a高表达,抑制miR-320a具有缓解阿霉素心脏损伤的作用[18]。此外,lncRNA MALAT1通过下调miR-320a表达还可减轻缺氧复氧诱导的血管内皮细胞损伤[19]。本研究显示,LPS诱导的A549细胞miR-320a表达显著升高,转染miR-320a抑制物下调miR-320a表达可减轻LPS诱导的A549细胞炎症反应和凋亡,与前人报道miR-320a的促炎结论类似[5]。此外,本研究发现lncRNA EGOT对miR-320a具有靶向负调控作用,且上调miR-320a表达还可逆转lncRNA OIP5-AS1对LPS诱导的A549细胞炎症反应和凋亡的影响,这为lncRNA OIP5-AS1/miR-320a分子轴参与LPS诱导的A549细胞炎症反应和凋亡提供了重要证据。
综上所述,lncRNA OIP5-AS1通过靶向miR-320a可减轻LPS诱导肺泡上皮细胞炎症反应和凋亡,进而可能减轻ALI严重程度。因此,lncRNA OIP5-AS1/miR-320a分子轴有望成为ALI的新型治疗靶点。
lncRNA EGOT靶向miR-320a对LPS诱导肺泡上皮细胞炎症反应和细胞凋亡的影响
Effect of lncRNA EGOT on LPS-induced inflammation and apoptosis of alveolar epithelial cells by targeting miR-320a
-
摘要:
目的探讨长链非编码RNA (lncRNA)嗜酸性粒细胞转录因子(EGOT)对脂多糖(LPS)诱导的肺泡上皮细胞A549凋亡、炎症反应的影响及可能机制。 方法将A549细胞分为对照组(Con)、LPS组、LPS+pcDNA组、LPS+pcDNA-EGOT组、LPS+anti-miR-NC组、LPS+anti-miR-320a组、LPS+pcDNA-EGOT+miR-NC组、LPS+pcDNA-EGOT+miR-320a组。实时荧光定量PCR (RT-qPCR)检测EGOT和miR-320a表达水平;流式细胞术检测细胞凋亡;试剂盒检测白细胞介素(IL)-6、IL-1β水平。双荧光素酶报告基因实验和RT-qPCR确定EGOT和miR-320a之间靶向作用。 结果与Con组比较,LPS组A549细胞凋亡率、IL-6和IL-1β水平均升高(P < 0.05),EGOT表达水平降低(P < 0.01),miR-320a表达水平明显升高(P < 0.01)。与LPS+pcDNA组比较,LPS+pcDNA-EGOT组A549细胞凋亡率、Bax蛋白表达、IL-6和IL-1β水平均降低(P < 0.05),Bcl-2蛋白表达升高(P < 0.05)。与LPS+anti-miR-NC组比较,LPS+anti-miR-320a组A549细胞凋亡率、Bax蛋白表达、IL-6和IL-1β水平均明显降低(P < 0.01),Bcl-2蛋白表达明显升高(P < 0.01)。与LPS+pcDNA-EGOT+miR-NC组比较,LPS+pcDNA-EGOT+miR-320a组A549细胞凋亡率、Bax蛋白表达、IL-6和IL-1β水平均明显升高(P < 0.01),Bcl-2蛋白表达明显降低(P < 0.01)。双荧光素酶报告实验显示,EGOT靶向负性调控miR-320a表达。 结论lncRNA EGOT通过靶向miR-320a可减轻LPS诱导肺泡上皮细胞炎症反应和细胞凋亡。 Abstract:ObjectiveTo explore the effect of long-chain non-coding RNA eosinophilic transcription factor (EGOT) on lipopolysaccharide (LPS) -induced apoptosis and inflammation of alveolar epithelial A549 cells and its possible mechanism. MethodsA549 cells were divided into control (Con) group, LPS group, LPS+pcDNA group, LPS+pcDNA-EGOT group, LPS+anti-miR-NC group, LPS+anti-miR-320a group, LPS+pcDNA-EGOT+miR-NC group, and LPS+pcDNA-EGOT+miR-320a group.Real-time quantitative PCR (RT-qPCR) was used to detect the expression levels of EGOT and miR-320a, flow cytometry was applied to detect apoptosis, and the kit was used to detect the levels of interleukin (IL) -6 and IL-1β.Dual luciferase reporter gene assay and RT-qPCR was employed to confirm the targeting relationship between EGOT and miR-320a. ResultsCompared with the Con group, the apoptosis rate, IL-6 and IL-1β levels of A549 cells in the LPS group were increased(P < 0.01), the EGOT expression level was reduced(P < 0.05), and the miR-320a expression level was significantly increased (P < 0.01).Compared with the LPS+pcDNA group, the apoptosis rate, Bax protein expression, IL-6 and IL-1β levels of A549 cells in the LPS+pcDNA-EGOT group were reduced(P < 0.05), and the Bcl-2 protein expression were increased(P < 0.05).Compared with the LPS+anti-miR-NC group, the apoptosis rate, Bax protein expression, IL-6 and IL-1β levels of A549 cells in the LPS+anti-miR-320a group were significantly reduced(P < 0.01), and the Bcl-2 protein expression were significantly increased(P < 0.01).Compared with the LPS+pcDNA-EGOT+miR-NC group, the apoptosis rate, Bax protein expression, IL-6 and IL-1β levels of A549 cells in the LPS+pcDNA-EGOT+miR-320a group were significantly increased (P < 0.01), and the Bcl-2 protein expression were significantly reduced (P < 0.01).The results of dual luciferase reporter gene assay showed that EGOT targeted and negatively regulated the miR-320a expression. ConclusionsLncRNA EGOT can alleviate LPS-induced inflammation and apoptosis of alveolar epithelial cells by targeting miR-320a. -
表 1 lncRNA EGOT和miR-320a在LPS刺激的肺上皮细胞中的表达(ni=9;x±s)
分组 EGOT miR-320a Con 1.00±0.07 1.00±0.08 LPS 0.39±0.03 2.61±0.23 t 24.03 19.83 P <0.01 <0.01 表 2 lncRNA EGOT过表达对LPS诱导的肺泡上皮细胞炎症因子表达的影响(ni=9;x±s)
分组 EGOT IL-6/(pg/mL) IL-1β/(pg/mL) Con 1.00±0.05 106.48±10.42 46.17±4.94 LPS 0.43±0.04* 353.51±25.90* 180.73±12.98* LPS+pcDNA 0.41±0.04* 349.84±19.43* 177.28±14.47* LPS+pcDNA-EGOT 2.31±0.22*#▲ 139.69±12.53*#▲ 64.96±6.26*#▲ F 528.68 482.33 419.16 P <0.01 <0.01 <0.01 MS组内 0.014 328.478 110.363 q检验:与Con组比较*P<0.05;与LPS组比较#P<0.05;与LPS+pcDNA组比较▲P<0.05 表 3 lncRNA EGOT过表达对LPS诱导的肺泡上皮细胞凋亡的影响(ni=9;x±s)
分组 凋亡率/% Bcl-2蛋白 Bax蛋白 Con 6.52±0.60 0.73±0.07 0.17±0.02 LPS 25.45±2.28* 0.28±0.03* 0.55±0.04* LPS+pcDNA 26.34±2.81* 0.26±0.02* 0.58±0.05* LPS+pcDNA-EGOT 10.59±1.01*#▲ 0.62±0.05*#▲ 0.24±0.02*#▲ F 256.47 234.86 324.49 P <0.01 <0.01 <0.01 MS组内 3.619 0.002 0.001 q检验:与Con组比较*P<0.05;与LPS组比较#P<0.05;与LPS+pcDNA组比较▲P<0.05 表 4 双荧光素酶报告实验结果(ni=9;x±s)
分组 WT-EGOT MUT-EGOT miR-NC 0.99±0.05 0.97±0.06 miR-320a 0.38±0.04 1.00±0.07 t 28.58 0.98 P <0.01 >0.05 表 5 lncRNA EGOT靶向调控miR-320a的表达(ni=9;x±s)
分组 miR-320a F P MS组内 pcDNA 1.00±0.05 579.59 <0.01 0.018 pcDNA-EGOT si-NC 0.50±0.05* 1.03±0.08# si-EGOT 2.97±0.25*#▲ q检验:与pcDNA组比较*P<0.05;与pcDNA-EGOT组比较#P<0.05;与si-NC组比较▲P<0.05 表 6 抑制miR-320a表达对LPS诱导的肺泡上皮细胞炎症因子和细胞凋亡的影响(ni=9;x±s)
分组 miR-320a IL-6/(pg/mL) IL-1β/(pg/mL) 凋亡率/% Bcl-2蛋白 Bax蛋白 LPS+anti-miR-NC 1.00±0.06 348.12±16.39 181.36±14.88 27.92±2.51 0.27±0.03 0.60±0.04 LPS+anti-miR-320a 0.54±0.05 152.01±16.50 78.06±7.03 12.56±1.18 0.58±0.04 0.30±0.03 t 17.67 25.30 188.83 16.61 18.60 14.40 P <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 表 7 miR-320a过表达逆转lncRNA EGOT过表达对LPS诱导的肺泡上皮细胞炎症因子和细胞凋亡的作用(ni=9;x±s)
分组 miR-320a IL-6/(pg/mL) IL-1β/(pg/mL) 凋亡率/% Bcl-2蛋白 Bax蛋白 LPS+pcDNA-EGOT+miR-NC 1.00±0.06 137.82±11.14 63.34±6.47 11.79±1.02 0.64±0.06 0.22±0.02 LPS+pcDNA-EGOT+miR-320a 2.53±0.23 311.34±28.45 145.64±12.94 20.51±1.84 0.32±0.03 0.45±0.03 t 19.31 17.04 17.07 12.44 14.31 19.14 P <0.01 <0.01 <0.01 <0.01 <0.01 <0.01 -
[1] BOCHAROW AV, WU T, BARANOVA IN, et al. Synthetic amphipathic helical peptides targeting cd36 attenuate lipopolysaccharide-induced inflammation and acute lung injury[J]. J Immunol, 2016, 197(2): 611. doi: 10.4049/jimmunol.1401028 [2] HEROLD S, GABRIELLI NM, VADÁSZ I. Novel concepts of acute lung injury and alveolar-capillary barrier dysfunction[J]. Am J Physiol Lung Cell Mol Physiol, 2013, 305(10): L665. doi: 10.1152/ajplung.00232.2013 [3] SCHMITZ SU, GROTE P, HERRMANN BG. Mechanisms of long noncoding RNA function in development and disease[J]. Cell Mol Life Sci, 2016, 73(13): 2491. doi: 10.1007/s00018-016-2174-5 [4] 马媛, 张大鹏, 王想, 等. lncRNA TUG1对高糖诱导的小鼠足细胞MPC5凋亡的影响[J]. 郑州大学学报(医学版), 2019, 54(6): 863. [5] 吴彩霞, 张小兰, 郭梦. 脑钠肽通过调控lncRNA EGOT缓解低氧诱导的大鼠心肌细胞损伤[J]. 临床和实验医学杂志, 2019, 18(19): 2020. doi: 10.3969/j.issn.1671-4695.2019.19.002 [6] LEE H, ZHANG D, ZHU Z, et al. Epithelial cell-derived microvesicles activate macrophages and promote inflammation via microvesicle-containing microRNAs[J]. Sci Rep, 2016, 6(1): 35250. doi: 10.1038/srep35250 [7] 罗超, 吴伟斌, 周瑾, 等. 奥拉帕尼通过PARP-1通路调控脂多糖诱导的A549细胞炎症反应[J]. 中国药理学与毒理学杂志, 2019, 33(3): 193. doi: 10.3867/j.issn.1000-3002.2019.03.004 [8] ZHANG C, PAN S, AISHA A, et al. Recombinant human brain natriuretic peptide regulates PI3K/AKT/mTOR pathway through lncRNA EGOT to attenuate hypoxia-induced injury in H9c2 cardiomyocytes[J]. Biochem Biophys Res Commun. 2018, 503(3): 1186. doi: 10.1016/j.bbrc.2018.07.023 [9] WANG IK, PALANISAMY K, SUN KT, et al. The functional interplay of lncRNA EGOT and HuR regulates hypoxia-induced autophagy in renal tubular cells[J]. J Cell Biochem, 2020, 121(11): 4522. doi: 10.1002/jcb.29669 [10] SHAO L, MENG D, YANG F, et al. Irisin-mediated protective effect on LPS-induced acute lung injury via suppressing inflammation and apoptosis of alveolar epithelial cells[J]. Biochem Biophys Res Commun, 2017, 487(2): 194. doi: 10.1016/j.bbrc.2017.04.020 [11] WANG L, TANG L, WANG Y, et al. Exendin-4 protects HUVECs from t-BHP-induced apoptosis via PI3K/Akt-Bcl-2-caspase-3 signaling[J]. Endocr Res, 2016, 41(3): 229. doi: 10.3109/07435800.2015.1110162 [12] 余鑫, 米智华, 高巨. 自噬在机械通气致大鼠脑组织神经元凋亡中的作用[J]. 实用临床医药杂志, 2019, 23(8): 18. [13] 王荣俊, 童斯浩. 姜黄素联合紫杉醇对肺癌细胞PC9凋亡的影响[J]. 蚌埠医学院学报, 2016, 41(12): 1549. [14] 赵华洲, 陈凛. 胃癌相关MicroRNA的研究进展[J]. 实用临床医药杂志, 2017, 21(13): 232. doi: 10.7619/jcmp.201713085 [15] LIN K, SU HY, JIANG LF, et al. Influences of miR-320a on proliferation and apoptosis of fibroblast-like synoviocytes in rheumatoid arthritis through targeting MAPK-ERK1/2[J]. Eur Rev Med Pharmacol Sci, 2019, 23(5): 1907. [16] JIN Y, CHEN X, GAO ZY, et al. The role of miR-320a and IL-1β in human chondrocyte degradation[J]. Bone Joint Res, 2017, 6(4): 196. doi: 10.1302/2046-3758.64.BJR-2016-0224.R1 [17] HE M, WANG J, YIN Z, et al. MiR-320a induces diabetic nephropathy via inhibiting MafB[J]. Aging (Albany NY), 2019, 11(10): 3055. [18] 何梦颖, 殷中伟, 赵艳茹, 等. MiR-320a通过靶向抑制VEGF信号通路介导阿霉素心脏损伤[J]. 中国分子心脏病学杂志, 2018, 18(5): 2594. [19] ZHU R, HU X, XU W, et al. LncRNA MALAT1 inhibits hypoxia/reoxygenation-induced human umbilical vein endothelial cell injury via targeting the microRNA-320a/RAC1 axis[J]. Biol Chem, 2020, 401(3): 349. doi: 10.1515/hsz-2019-0316 -





 下载:
下载: